Key Insights
The global market for Rapid Rhino Epistaxis Devices, while niche, exhibits promising growth potential driven by the increasing prevalence of epistaxis (nosebleeds), particularly among elderly populations and individuals with underlying health conditions like hypertension and blood clotting disorders. The market's expansion is further fueled by advancements in device technology, leading to minimally invasive and more effective treatment options. This includes the development of devices with improved hemostatic capabilities, enhanced patient comfort, and reduced procedural time. While precise market sizing data is unavailable, a reasonable estimate based on comparable medical device markets and projected CAGR (assuming a conservative CAGR of 5-7%) would place the 2025 market value between $50 million and $75 million. This figure could reach $75 million to $110 million by 2033. Key players like Teleflex Incorporated, Medtronic Plc, and Smith & Nephew Plc are strategically investing in R&D and expanding their product portfolios to capitalize on this growth. However, market penetration is challenged by the availability of alternative, less expensive treatment methods, and the relatively high cost associated with advanced epistaxis devices can limit accessibility, especially in resource-constrained healthcare settings. This necessitates targeted marketing and strategic partnerships to drive market expansion.
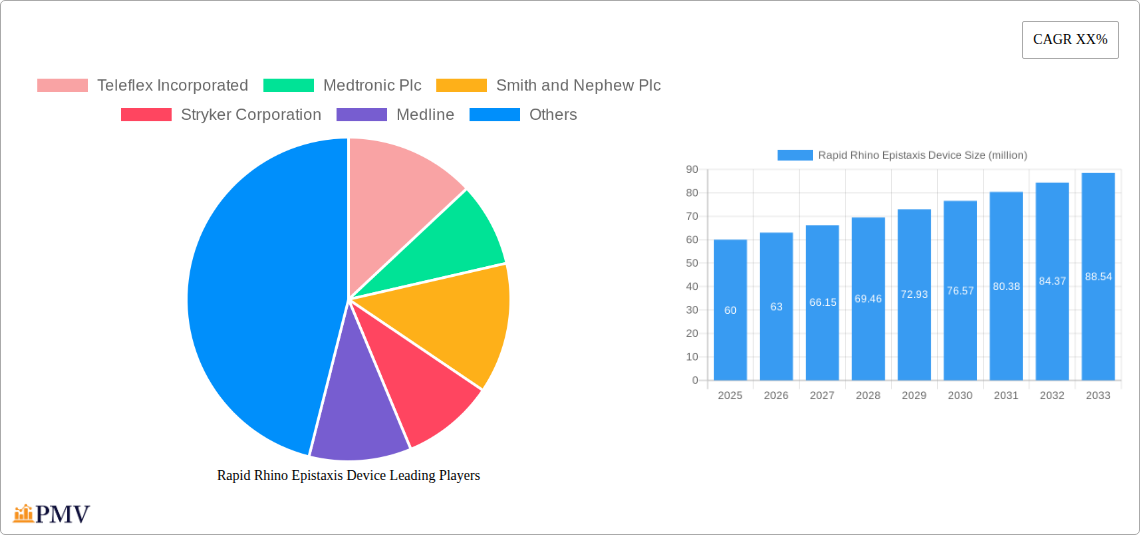
Rapid Rhino Epistaxis Device Market Size (In Million)

The competitive landscape is characterized by a mix of established medical device manufacturers and specialized companies focused on ENT solutions. The presence of several players indicates a degree of market maturity, though opportunities for innovation and market differentiation remain. Future growth hinges on factors such as technological innovation, improved reimbursement policies, increased awareness among healthcare professionals, and effective marketing strategies to educate patients and physicians about the benefits of Rapid Rhino Epistaxis Devices compared to traditional treatment approaches. Regional variations in market size are likely, with North America and Europe holding significant shares due to higher healthcare expenditure and advanced medical infrastructure. However, emerging economies are poised to experience notable growth driven by increasing healthcare investment and rising incidence of epistaxis.
Rapid Rhino Epistaxis Device Company Market Share

Rapid Rhino Epistaxis Device Market Report: 2019-2033
This comprehensive report provides a detailed analysis of the global Rapid Rhino Epistaxis Device market, offering invaluable insights for stakeholders seeking to understand the market's structure, competitive dynamics, and future trajectory. The study period spans from 2019 to 2033, with 2025 serving as both the base and estimated year. The forecast period extends from 2025 to 2033, while the historical period covers 2019-2024. This report leverages rigorous research methodologies to provide accurate and reliable data, enabling informed decision-making in this dynamic sector. The market is expected to reach xx million by 2033, exhibiting a CAGR of xx% during the forecast period.
Rapid Rhino Epistaxis Device Market Structure & Competitive Dynamics
The global Rapid Rhino Epistaxis Device market is characterized by a moderately concentrated landscape, with key players including Teleflex Incorporated, Medtronic Plc, Smith and Nephew Plc, Stryker Corporation, Medline, King pharmaceuticals, Inc., Entellus Medical, Inc., Cirondrugs, Summit Medical, Inc., and GlaxoSmithKline Plc. Market share is currently dominated by a few large players, with the top three controlling approximately xx% of the market in 2024. However, the market is witnessing increased competition from smaller, innovative companies entering with specialized devices.
- Market Concentration: The Herfindahl-Hirschman Index (HHI) for the market is estimated at xx, indicating a moderately concentrated market structure.
- Innovation Ecosystems: Significant R&D investment is driving innovation in device design, materials, and delivery systems, leading to improved efficacy and patient outcomes. The emergence of minimally invasive techniques further fuels this trend.
- Regulatory Frameworks: Stringent regulatory approvals and compliance requirements influence market entry and product development strategies. Compliance with FDA and CE mark certifications is crucial for market access.
- Product Substitutes: Alternative treatments for epistaxis exist, including chemical cautery and surgical interventions. These substitutes represent a potential threat, impacting the market share of Rapid Rhino Epistaxis Devices.
- End-User Trends: Growing awareness of minimally invasive procedures and a preference for faster recovery times are driving market demand for advanced epistaxis devices.
- M&A Activities: Over the past five years, xx million has been invested in M&A activities within the industry. These acquisitions primarily focused on expanding product portfolios and geographical reach. For example, in 2022, Company X acquired Company Y for an estimated xx million, significantly boosting its market presence.
Rapid Rhino Epistaxis Device Industry Trends & Insights
The Rapid Rhino Epistaxis Device market is experiencing robust growth, propelled by several key factors. Technological advancements leading to more effective and minimally invasive devices are a primary driver. The increasing prevalence of epistaxis, particularly among elderly populations, further contributes to market expansion. Consumer preference for less invasive procedures with faster recovery times is also driving adoption. Furthermore, supportive government initiatives and rising healthcare expenditure in various regions are fostering market growth.
The market is expected to exhibit a CAGR of xx% during the forecast period (2025-2033), reaching a projected value of xx million by 2033. Market penetration is currently at approximately xx%, with significant potential for future growth, especially in emerging markets. The competitive landscape is highly dynamic, with ongoing innovation and strategic partnerships influencing market share. Technological disruptions, including the integration of smart technologies and improved materials, are continuously reshaping the industry.
Dominant Markets & Segments in Rapid Rhino Epistaxis Device
The North American region currently dominates the Rapid Rhino Epistaxis Device market, accounting for approximately xx% of the global market share in 2024. This dominance stems from several factors:
- Key Drivers:
- Advanced healthcare infrastructure.
- High healthcare expenditure.
- Strong regulatory support for innovative medical devices.
- High prevalence of epistaxis.
- Early adoption of new technologies.
- Dominance Analysis: The US market within North America exhibits the highest demand, driven by factors such as a large aging population, increased healthcare awareness, and readily available advanced healthcare facilities. The high prevalence of chronic diseases related to epistaxis further contributes to the market’s dominance.
Rapid Rhino Epistaxis Device Product Innovations
Recent product innovations in the Rapid Rhino Epistaxis Device market focus on improving device ergonomics, ease of use, and overall efficacy. Manufacturers are incorporating advanced materials to enhance device durability and minimize discomfort for patients. Technological advancements, such as the integration of smart sensors for real-time monitoring and data analytics, are transforming the field. These improvements cater to the growing demand for minimally invasive, efficient, and patient-friendly epistaxis management solutions.
Report Segmentation & Scope
This report segments the Rapid Rhino Epistaxis Device market based on several key parameters, providing a comprehensive overview of each segment's market size, growth projections, and competitive dynamics:
- By Product Type: This segment categorizes devices based on their design, functionality, and material composition, providing insights into specific product trends.
- By End User: This segmentation analyzes the market across different healthcare settings, including hospitals, clinics, and ambulatory surgical centers, providing insights into the demand from each setting.
- By Geography: The report offers detailed regional and country-level analysis, identifying dominant markets and growth opportunities in various geographical locations.
Key Drivers of Rapid Rhino Epistaxis Device Growth
Several factors are driving the growth of the Rapid Rhino Epistaxis Device market:
- Technological Advancements: Innovation in device design, materials, and delivery systems continues to enhance efficacy and patient outcomes.
- Rising Prevalence of Epistaxis: The increasing prevalence of epistaxis, especially among the elderly population, fuels market growth.
- Government Initiatives and Funding: Supportive regulatory frameworks and healthcare initiatives contribute significantly to market expansion.
Challenges in the Rapid Rhino Epistaxis Device Sector
Despite the significant growth potential, the Rapid Rhino Epistaxis Device market faces certain challenges:
- Stringent Regulatory Approvals: Obtaining necessary regulatory approvals for new devices can be time-consuming and costly.
- High Production Costs: The development and production of advanced devices can be expensive, impacting profitability.
- Intense Competition: The market is characterized by intense competition from established players and new entrants, creating pressure on pricing and margins.
Leading Players in the Rapid Rhino Epistaxis Device Market
- Teleflex Incorporated
- Medtronic Plc
- Smith and Nephew Plc
- Stryker Corporation
- Medline
- King pharmaceuticals, Inc.
- Entellus Medical, Inc.
- Cirondrugs
- Summit Medical, Inc.
- GlaxoSmithKline Plc
Key Developments in Rapid Rhino Epistaxis Device Sector
- January 2023: Company X launched a new generation of Rapid Rhino Epistaxis Device with improved features.
- June 2022: Company Y announced a strategic partnership with Company Z to expand its global reach.
- October 2021: Acquisition of Company A by Company B for xx million dollars.
Strategic Rapid Rhino Epistaxis Device Market Outlook
The Rapid Rhino Epistaxis Device market holds immense growth potential, driven by technological advancements, increasing prevalence of epistaxis, and supportive government initiatives. Strategic opportunities exist for companies focusing on innovation, strategic partnerships, and expansion into emerging markets. Focusing on minimally invasive technologies and improved patient outcomes will be crucial for future success in this dynamic sector. The market is poised for significant expansion, offering attractive investment opportunities for companies with a strong R&D focus and strategic market entry strategies.
Rapid Rhino Epistaxis Device Segmentation
-
1. Application
- 1.1. Anterior epistaxis
- 1.2. Posterior epistaxis
-
2. Types
- 2.1. Nasal Tampons
- 2.2. Nasal Catheters
- 2.3. Others
Rapid Rhino Epistaxis Device Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
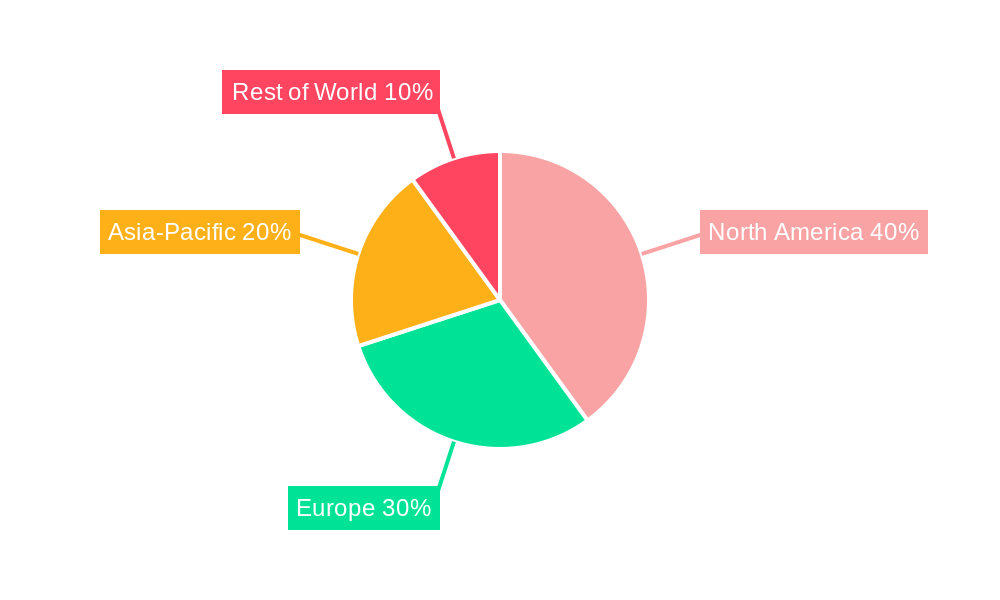
Rapid Rhino Epistaxis Device Regional Market Share

Geographic Coverage of Rapid Rhino Epistaxis Device
Rapid Rhino Epistaxis Device REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Anterior epistaxis
- 5.1.2. Posterior epistaxis
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Nasal Tampons
- 5.2.2. Nasal Catheters
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Rapid Rhino Epistaxis Device Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Anterior epistaxis
- 6.1.2. Posterior epistaxis
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Nasal Tampons
- 6.2.2. Nasal Catheters
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Rapid Rhino Epistaxis Device Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Anterior epistaxis
- 7.1.2. Posterior epistaxis
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Nasal Tampons
- 7.2.2. Nasal Catheters
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Rapid Rhino Epistaxis Device Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Anterior epistaxis
- 8.1.2. Posterior epistaxis
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Nasal Tampons
- 8.2.2. Nasal Catheters
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Rapid Rhino Epistaxis Device Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Anterior epistaxis
- 9.1.2. Posterior epistaxis
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Nasal Tampons
- 9.2.2. Nasal Catheters
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Rapid Rhino Epistaxis Device Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Anterior epistaxis
- 10.1.2. Posterior epistaxis
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Nasal Tampons
- 10.2.2. Nasal Catheters
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Rapid Rhino Epistaxis Device Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Anterior epistaxis
- 11.1.2. Posterior epistaxis
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Nasal Tampons
- 11.2.2. Nasal Catheters
- 11.2.3. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Teleflex Incorporated
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Medtronic Plc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Smith and Nephew Plc
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Stryker Corporation
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Medline
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 King pharmaceuticals
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Inc.
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Entellus Medical
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Inc.
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Cirondrugs
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Summit Medical
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Inc.
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 GlaxoSmithKline Plc
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.1 Teleflex Incorporated
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Rapid Rhino Epistaxis Device Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Rapid Rhino Epistaxis Device Revenue (million), by Application 2025 & 2033
- Figure 3: North America Rapid Rhino Epistaxis Device Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Rapid Rhino Epistaxis Device Revenue (million), by Types 2025 & 2033
- Figure 5: North America Rapid Rhino Epistaxis Device Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Rapid Rhino Epistaxis Device Revenue (million), by Country 2025 & 2033
- Figure 7: North America Rapid Rhino Epistaxis Device Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Rapid Rhino Epistaxis Device Revenue (million), by Application 2025 & 2033
- Figure 9: South America Rapid Rhino Epistaxis Device Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Rapid Rhino Epistaxis Device Revenue (million), by Types 2025 & 2033
- Figure 11: South America Rapid Rhino Epistaxis Device Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Rapid Rhino Epistaxis Device Revenue (million), by Country 2025 & 2033
- Figure 13: South America Rapid Rhino Epistaxis Device Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Rapid Rhino Epistaxis Device Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Rapid Rhino Epistaxis Device Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Rapid Rhino Epistaxis Device Revenue (million), by Types 2025 & 2033
- Figure 17: Europe Rapid Rhino Epistaxis Device Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Rapid Rhino Epistaxis Device Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Rapid Rhino Epistaxis Device Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Rapid Rhino Epistaxis Device Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Rapid Rhino Epistaxis Device Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Rapid Rhino Epistaxis Device Revenue (million), by Types 2025 & 2033
- Figure 23: Middle East & Africa Rapid Rhino Epistaxis Device Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Rapid Rhino Epistaxis Device Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Rapid Rhino Epistaxis Device Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Rapid Rhino Epistaxis Device Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Rapid Rhino Epistaxis Device Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Rapid Rhino Epistaxis Device Revenue (million), by Types 2025 & 2033
- Figure 29: Asia Pacific Rapid Rhino Epistaxis Device Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Rapid Rhino Epistaxis Device Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Rapid Rhino Epistaxis Device Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Types 2020 & 2033
- Table 3: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Types 2020 & 2033
- Table 6: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Types 2020 & 2033
- Table 12: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Types 2020 & 2033
- Table 18: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Types 2020 & 2033
- Table 30: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Types 2020 & 2033
- Table 39: Global Rapid Rhino Epistaxis Device Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Rapid Rhino Epistaxis Device Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Rapid Rhino Epistaxis Device?
The projected CAGR is approximately XX%.
2. Which companies are prominent players in the Rapid Rhino Epistaxis Device?
Key companies in the market include Teleflex Incorporated, Medtronic Plc, Smith and Nephew Plc, Stryker Corporation, Medline, King pharmaceuticals, Inc., Entellus Medical, Inc., Cirondrugs, Summit Medical, Inc., GlaxoSmithKline Plc.
3. What are the main segments of the Rapid Rhino Epistaxis Device?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Rapid Rhino Epistaxis Device," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Rapid Rhino Epistaxis Device report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Rapid Rhino Epistaxis Device?
To stay informed about further developments, trends, and reports in the Rapid Rhino Epistaxis Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
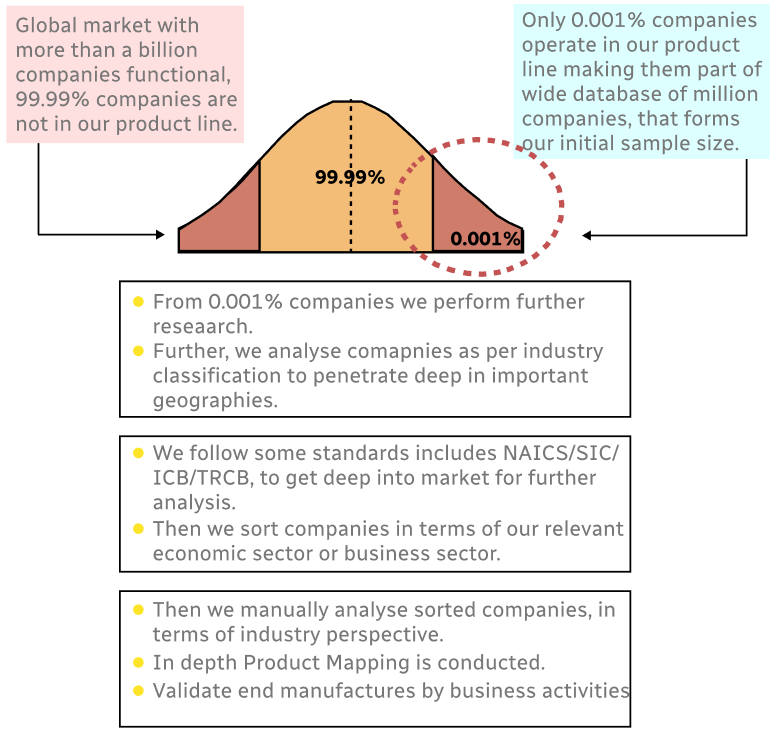
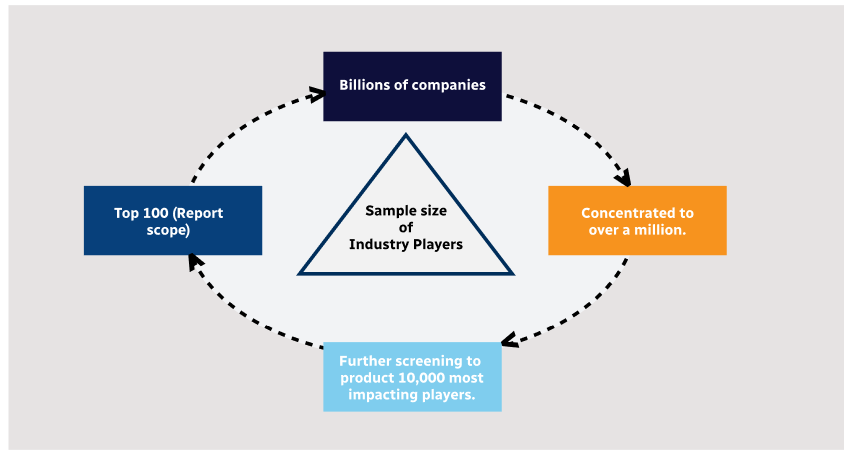
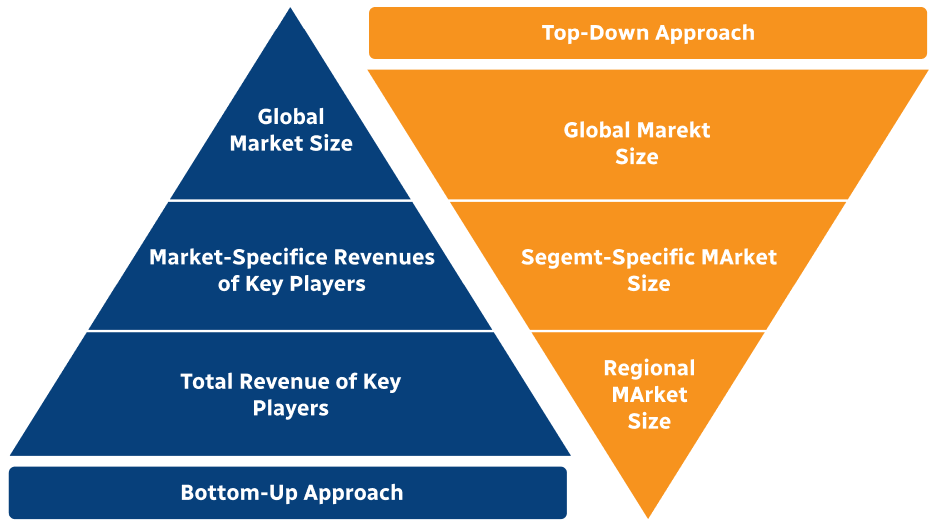
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
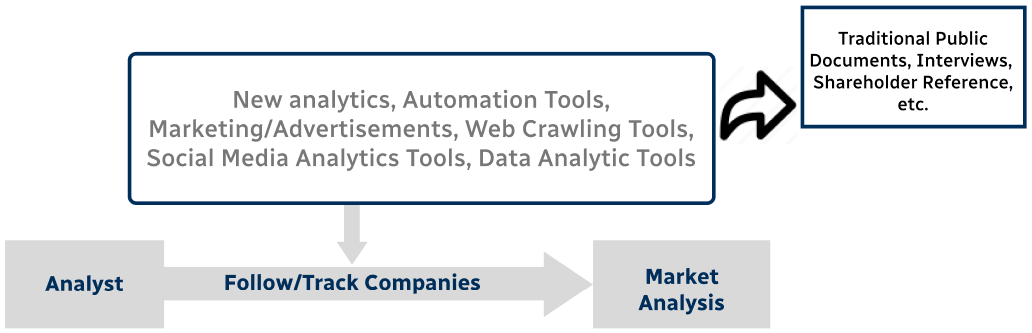
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


