Key Insights
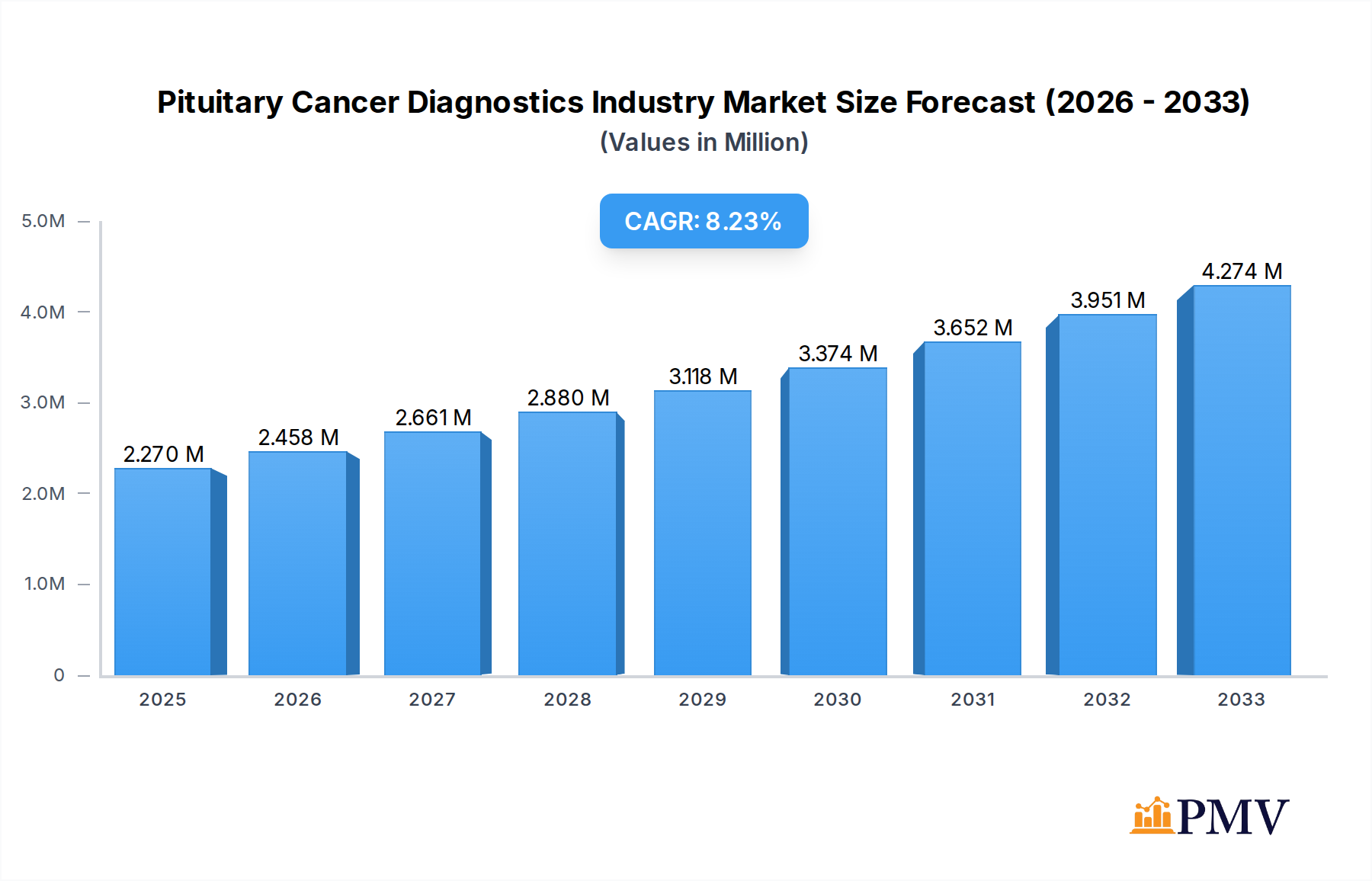
The global Pituitary Cancer Diagnostics market is poised for significant expansion, projected to reach USD 2.27 Million with a robust CAGR of 8.30% during the forecast period of 2025-2033. This growth is underpinned by escalating incidences of pituitary tumors, advancements in diagnostic technologies, and increased awareness among healthcare professionals and patients regarding early detection. The market is propelled by the critical need for accurate and timely diagnosis of pituitary adenomas and other malignant pituitary tumors, which can lead to severe hormonal imbalances and neurological complications if left untreated. Key drivers include the rising prevalence of endocrine disorders, coupled with a growing emphasis on precision medicine and personalized treatment approaches. The development of sophisticated imaging techniques like MRI and CT scans, alongside molecular testing and liquid biopsy, is revolutionizing diagnostic capabilities, offering higher accuracy and earlier identification of these rare but impactful cancers. This technological progression is crucial in improving patient outcomes and reducing the burden of the disease.

Pituitary Cancer Diagnostics Industry Market Size (In Million)

The market landscape is characterized by a competitive environment with key players like Siemens Healthineers, Hologic Inc., GE Healthcare, and Thermo Fisher Scientific, actively investing in research and development to enhance diagnostic platforms. The Diagnostic Type segment is dominated by Imaging Tests (MRI, CT Scan) due to their non-invasive nature and high diagnostic yield, followed by Molecular Testing which is gaining traction for its precision in identifying specific genetic mutations. Within Tumor Type, Metastatic and Glioblastoma segments are expected to show considerable growth due to their aggressive nature and the need for prompt diagnosis. The End User segment is largely driven by Hospitals, reflecting their role as primary healthcare providers equipped with advanced diagnostic infrastructure. Geographically, North America and Europe are anticipated to lead the market, owing to well-established healthcare systems and high adoption rates of advanced diagnostic technologies. The Asia Pacific region is emerging as a high-growth market due to increasing healthcare expenditure and a burgeoning patient population.
Pituitary Cancer Diagnostics Industry Company Market Share

Gain unparalleled insights into the dynamic pituitary cancer diagnostics market with this in-depth report. Covering the historical period of 2019–2024 and projecting growth through 2033, with a base and estimated year of 2025, this analysis delves deep into diagnostic technologies, tumor types, and end-user dynamics. Explore the latest pituitary adenoma diagnostics, neuroendocrine tumor testing, and brain tumor diagnostic solutions. Understand the competitive landscape, key market drivers, emerging trends, and strategic opportunities that will shape the future of pituitary cancer detection. This report is essential for stakeholders seeking to navigate the complexities of the global pituitary cancer diagnostics industry, including imaging tests, biopsy analysis, and molecular diagnostics.
Pituitary Cancer Diagnostics Industry Market Structure & Competitive Dynamics
The pituitary cancer diagnostics industry exhibits a moderately concentrated market structure, characterized by the presence of established multinational healthcare giants and specialized diagnostic solution providers. Innovation ecosystems are thriving, driven by advancements in molecular testing and advanced imaging techniques such as MRI and CT scans. Regulatory frameworks, overseen by bodies like the FDA and EMA, significantly influence product development and market entry, ensuring patient safety and diagnostic accuracy. Product substitutes, including less invasive screening methods and alternative diagnostic pathways, are emerging but currently hold a limited market share compared to established pituitary cancer diagnostic tests. End-user trends reveal a growing preference for minimally invasive and highly accurate diagnostic methods, pushing demand towards molecular diagnostics and advanced biopsy analysis. Mergers and acquisitions (M&A) activities are strategically shaping the competitive landscape, with recent transactions aimed at expanding product portfolios and geographic reach. For instance, the acquisition of smaller, innovative biotech firms by larger players has been observed, injecting fresh capital and technology into the market. Key companies like Siemens Healthineers, Hologic Inc., GE Healthcare, Hitachi Ltd., Koninklijke Philips N.V., Fujifilm Corporation, Thermo Fisher Scientific, and NantOmics are actively engaged in R&D and strategic partnerships to maintain their competitive edge. Market share distribution is influenced by the efficacy and cost-effectiveness of diagnostic solutions, with a notable emphasis on early detection of pituitary adenomas and metastatic brain tumors.
Pituitary Cancer Diagnostics Industry Industry Trends & Insights
The pituitary cancer diagnostics industry is poised for significant growth, projected to expand at a robust Compound Annual Growth Rate (CAGR) of approximately 7.5% between 2025 and 2033. This upward trajectory is fueled by several key market growth drivers. Firstly, the increasing global incidence of pituitary tumors, coupled with a heightened awareness among both healthcare professionals and patients regarding early detection, is a primary catalyst. Advancements in diagnostic imaging technologies, particularly high-resolution MRI and sophisticated CT scans, are enhancing the precision and speed of diagnosis, making them indispensable tools. Furthermore, the growing adoption of molecular testing for personalized medicine approaches is revolutionizing the field. These advanced tests can identify specific genetic mutations associated with pituitary tumors, aiding in prognosis and treatment planning. The integration of artificial intelligence (AI) and machine learning (ML) in image analysis and data interpretation is another significant technological disruption, promising to improve diagnostic accuracy and reduce turnaround times. Consumer preferences are shifting towards less invasive and more convenient diagnostic methods. This trend is driving innovation in liquid biopsy technologies and non-invasive screening approaches for pituitary adenoma diagnosis and other brain tumor detection. The competitive dynamics are intensifying, with companies investing heavily in research and development to launch novel diagnostic assays and platforms. Strategic collaborations between diagnostic manufacturers, research institutions, and pharmaceutical companies are becoming increasingly common to accelerate the development and commercialization of cutting-edge diagnostic solutions. The expanding healthcare infrastructure in emerging economies and rising healthcare expenditure are also contributing to market penetration, making advanced diagnostics more accessible to a wider population. The focus on improving patient outcomes and reducing the burden of pituitary cancer is a continuous underlying theme driving innovation and market expansion.
Dominant Markets & Segments in Pituitary Cancer Diagnostics Industry
North America currently stands as the dominant region in the pituitary cancer diagnostics industry, driven by its advanced healthcare infrastructure, high per capita healthcare spending, and significant investments in medical research and development. Within North America, the United States leads in market share due to the presence of leading diagnostic companies, extensive adoption of advanced technologies, and a robust regulatory environment that encourages innovation.
Dominant Segments:
Diagnostic Type:
- Imaging Test (MRI, CT Scan, Other): This segment holds the largest market share due to its non-invasive nature, high resolution, and established role in visualizing pituitary tumors and their extent. The ongoing technological advancements in MRI and CT technology, such as functional MRI (fMRI) and advanced spectral CT, further solidify its dominance.
- Key Drivers: High accuracy in tumor detection and localization, widespread availability of advanced imaging equipment, increasing incidence of incidentally discovered pituitary lesions, and insurance coverage for these procedures.
- Molecular Testing: This segment is experiencing the fastest growth, driven by the increasing demand for personalized medicine and targeted therapies. Genetic profiling helps in classifying tumor subtypes, predicting treatment response, and identifying potential therapeutic targets.
- Key Drivers: Growing understanding of the genetic basis of pituitary tumors, development of novel genetic markers, demand for precision oncology, and advancements in sequencing technologies.
- Biopsy: While invasive, biopsy remains crucial for definitive histopathological diagnosis and grading of pituitary tumors, especially for differentiating between benign adenomas and rarer malignant forms.
- Key Drivers: Need for definitive diagnosis in complex cases, characterization of tumor aggressiveness, and guidance for treatment selection.
- Lumbar Puncture & Others: These segments cater to specific diagnostic needs, such as assessing cerebrospinal fluid (CSF) for tumor markers or metastatic spread, but hold a smaller share compared to imaging and molecular tests.
- Imaging Test (MRI, CT Scan, Other): This segment holds the largest market share due to its non-invasive nature, high resolution, and established role in visualizing pituitary tumors and their extent. The ongoing technological advancements in MRI and CT technology, such as functional MRI (fMRI) and advanced spectral CT, further solidify its dominance.
Tumor Type:
- Pituitary Endoma: This category, encompassing pituitary adenomas, represents the most prevalent type of pituitary tumor, driving significant demand for diagnostic solutions.
- Key Drivers: High incidence rates, variety of hormonal and non-hormonal subtypes requiring specific diagnostic approaches, and continuous advancements in understanding their pathophysiology.
- Metastatic & Glioblastoma: While less common as primary pituitary tumors, metastatic lesions from other primary cancers and glioblastomas involving the pituitary region also contribute to the diagnostic market.
- Key Drivers: Increasing survival rates of other cancers leading to higher incidence of metastases, and the critical need for accurate diagnosis in complex neurological presentations.
- Meningioma & Others: These represent rarer types of tumors affecting the pituitary region, requiring specialized diagnostic expertise.
- Pituitary Endoma: This category, encompassing pituitary adenomas, represents the most prevalent type of pituitary tumor, driving significant demand for diagnostic solutions.
End User:
- Hospitals: Hospitals are the largest end-users, offering comprehensive diagnostic services, from imaging to pathology and molecular testing, under one roof.
- Key Drivers: Integrated healthcare delivery systems, access to advanced diagnostic equipment and specialized personnel, and the majority of complex pituitary cancer cases being managed in hospital settings.
- Diagnostics Centers: Independent diagnostic centers are growing in prominence, particularly for specialized imaging and molecular diagnostics, offering cost-effective and accessible solutions.
- Key Drivers: Focus on specific diagnostic services, competitive pricing, and increasing patient convenience.
- Others (Research Institutions, Academic Medical Centers): These entities contribute significantly to R&D and the development of new diagnostic methodologies.
- Hospitals: Hospitals are the largest end-users, offering comprehensive diagnostic services, from imaging to pathology and molecular testing, under one roof.
The interplay of these segments, driven by technological advancements and clinical needs, dictates the overall market dynamics and growth potential within the pituitary cancer diagnostics industry.
Pituitary Cancer Diagnostics Industry Product Innovations
Recent product innovations in the pituitary cancer diagnostics industry are heavily focused on enhancing accuracy, reducing invasiveness, and enabling personalized treatment strategies. Advances in high-resolution MRI and AI-powered image analysis software are improving the detection and characterization of subtle pituitary abnormalities. Furthermore, the development of novel molecular diagnostic assays for identifying specific genetic mutations and biomarkers associated with different pituitary tumor subtypes is a key trend. These innovations offer a competitive advantage by providing more precise diagnostic information, facilitating targeted therapy selection, and ultimately improving patient outcomes in the management of pituitary adenomas and other brain tumors.
Report Segmentation & Scope
This comprehensive report segments the pituitary cancer diagnostics market across several key dimensions to provide granular insights. The Diagnostic Type segmentation includes: Imaging Test (MRI, CT Scan, Other), Lumbar Puncture, Biopsy, Molecular Testing, and Others. Each sub-segment is analyzed for its market size, growth projections, and competitive dynamics, with Molecular Testing anticipated to show the highest growth rate. The Tumor Type segmentation covers: Metastatic, Meningioma, Glioblastoma, Pituitary Endoma, and Others. Pituitary Endoma is expected to represent the largest market share due to its prevalence. The End User segmentation focuses on Hospitals, Diagnostics Centers, and Others, with Hospitals projected to remain the dominant end-user segment throughout the forecast period. The scope of the report extends from 2019 to 2033, with detailed analysis for the historical, base, estimated, and forecast periods.
Key Drivers of Pituitary Cancer Diagnostics Industry Growth
Several pivotal factors are accelerating the growth of the pituitary cancer diagnostics industry. The increasing incidence of pituitary tumors, coupled with a growing emphasis on early detection and diagnosis, is a primary driver. Technological advancements in medical imaging (e.g., advanced MRI sequences) and molecular diagnostics are providing more accurate and faster diagnostic capabilities. Growing global healthcare expenditure and expanding access to diagnostic services, particularly in emerging economies, are also contributing significantly. Furthermore, the increasing understanding of the genetic underpinnings of pituitary tumors is fueling the demand for precise molecular testing for personalized treatment strategies.
Challenges in the Pituitary Cancer Diagnostics Industry Sector
Despite the promising growth, the pituitary cancer diagnostics industry faces several challenges. High costs associated with advanced diagnostic technologies and molecular testing can limit accessibility, especially in resource-constrained regions. Stringent regulatory approval processes for new diagnostic devices and assays can lead to extended market entry timelines. The shortage of skilled professionals in specialized areas like neuro-radiology and molecular pathology can also pose a restraint. Furthermore, the availability of less invasive but potentially less specific screening methods necessitates careful interpretation and validation to avoid misdiagnosis, impacting the overall diagnostic pathway for pituitary cancer.
Leading Players in the Pituitary Cancer Diagnostics Industry Market
- Siemens Healthineers
- Hologic Inc.
- GE Healthcare
- Hitachi Ltd.
- Koninklijke Philips N V
- Fujifilm Corporation
- Thermo Fisher Scientific
- NantOmics
Key Developments in Pituitary Cancer Diagnostics Industry Sector
- February 2023: Roche announced the launch of the IDH1 R132H (MRQ-67) Rabbit Monoclonal Primary Antibody and the ATRX Rabbit Polyclonal Antibody to identify mutation status in patients diagnosed with brain cancer. This development offers enhanced tools for brain tumor diagnostics and personalized treatment selection.
- January 2023: Datar Cancer Genetics Inc. received FDA breakthrough device designation for the TriNetra-Glio blood test to help in the diagnosis of brain tumors. This signifies a major advancement in non-invasive tumor detection and could revolutionize early pituitary cancer diagnosis.
Strategic Pituitary Cancer Diagnostics Industry Market Outlook
The strategic outlook for the pituitary cancer diagnostics industry is highly positive, characterized by continuous innovation and expanding market opportunities. The increasing demand for early and accurate diagnosis, coupled with the burgeoning field of personalized medicine, will drive further investment in advanced molecular diagnostic technologies and sophisticated imaging solutions. Collaborations between technology providers, healthcare institutions, and research organizations will be crucial for accelerating the development and adoption of novel diagnostic tools. The growing emphasis on minimally invasive techniques and liquid biopsy approaches presents significant potential for market expansion. Stakeholders focusing on developing cost-effective and accessible diagnostic solutions will be well-positioned to capitalize on the increasing global demand for effective pituitary cancer detection and management.
Pituitary Cancer Diagnostics Industry Segmentation
-
1. Diagnostic Type
-
1.1. Imaging Test
- 1.1.1. MRI
- 1.1.2. CT Scan
- 1.1.3. Other
- 1.2. Lumbar Puncture
- 1.3. Biopsy
- 1.4. Molecular Testing
- 1.5. Others
-
1.1. Imaging Test
-
2. Tumor Type
- 2.1. Metastatic
- 2.2. Meningioma
- 2.3. Glioblastoma
- 2.4. Pituitary Endoma
- 2.5. Others
-
3. End User
- 3.1. Hospitals
- 3.2. Diagnostics Centers
- 3.3. Others
Pituitary Cancer Diagnostics Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America

Pituitary Cancer Diagnostics Industry Regional Market Share

Geographic Coverage of Pituitary Cancer Diagnostics Industry
Pituitary Cancer Diagnostics Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.30% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.2.1. Increasing Prevalence of Neurological Disorders and Cancers; Rising Number of Smokers
- 3.3. Market Restrains
- 3.3.1. High Costs Associated with Diagnosis
- 3.4. Market Trends
- 3.4.1. MRI segment is Expected to Hold a Significant Share in the Brain Tumor Diagnostics Market
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 5.1.1. Imaging Test
- 5.1.1.1. MRI
- 5.1.1.2. CT Scan
- 5.1.1.3. Other
- 5.1.2. Lumbar Puncture
- 5.1.3. Biopsy
- 5.1.4. Molecular Testing
- 5.1.5. Others
- 5.1.1. Imaging Test
- 5.2. Market Analysis, Insights and Forecast - by Tumor Type
- 5.2.1. Metastatic
- 5.2.2. Meningioma
- 5.2.3. Glioblastoma
- 5.2.4. Pituitary Endoma
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by End User
- 5.3.1. Hospitals
- 5.3.2. Diagnostics Centers
- 5.3.3. Others
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 6. North America Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 6.1.1. Imaging Test
- 6.1.1.1. MRI
- 6.1.1.2. CT Scan
- 6.1.1.3. Other
- 6.1.2. Lumbar Puncture
- 6.1.3. Biopsy
- 6.1.4. Molecular Testing
- 6.1.5. Others
- 6.1.1. Imaging Test
- 6.2. Market Analysis, Insights and Forecast - by Tumor Type
- 6.2.1. Metastatic
- 6.2.2. Meningioma
- 6.2.3. Glioblastoma
- 6.2.4. Pituitary Endoma
- 6.2.5. Others
- 6.3. Market Analysis, Insights and Forecast - by End User
- 6.3.1. Hospitals
- 6.3.2. Diagnostics Centers
- 6.3.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 7. Europe Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 7.1.1. Imaging Test
- 7.1.1.1. MRI
- 7.1.1.2. CT Scan
- 7.1.1.3. Other
- 7.1.2. Lumbar Puncture
- 7.1.3. Biopsy
- 7.1.4. Molecular Testing
- 7.1.5. Others
- 7.1.1. Imaging Test
- 7.2. Market Analysis, Insights and Forecast - by Tumor Type
- 7.2.1. Metastatic
- 7.2.2. Meningioma
- 7.2.3. Glioblastoma
- 7.2.4. Pituitary Endoma
- 7.2.5. Others
- 7.3. Market Analysis, Insights and Forecast - by End User
- 7.3.1. Hospitals
- 7.3.2. Diagnostics Centers
- 7.3.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 8. Asia Pacific Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 8.1.1. Imaging Test
- 8.1.1.1. MRI
- 8.1.1.2. CT Scan
- 8.1.1.3. Other
- 8.1.2. Lumbar Puncture
- 8.1.3. Biopsy
- 8.1.4. Molecular Testing
- 8.1.5. Others
- 8.1.1. Imaging Test
- 8.2. Market Analysis, Insights and Forecast - by Tumor Type
- 8.2.1. Metastatic
- 8.2.2. Meningioma
- 8.2.3. Glioblastoma
- 8.2.4. Pituitary Endoma
- 8.2.5. Others
- 8.3. Market Analysis, Insights and Forecast - by End User
- 8.3.1. Hospitals
- 8.3.2. Diagnostics Centers
- 8.3.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 9. Middle East and Africa Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 9.1.1. Imaging Test
- 9.1.1.1. MRI
- 9.1.1.2. CT Scan
- 9.1.1.3. Other
- 9.1.2. Lumbar Puncture
- 9.1.3. Biopsy
- 9.1.4. Molecular Testing
- 9.1.5. Others
- 9.1.1. Imaging Test
- 9.2. Market Analysis, Insights and Forecast - by Tumor Type
- 9.2.1. Metastatic
- 9.2.2. Meningioma
- 9.2.3. Glioblastoma
- 9.2.4. Pituitary Endoma
- 9.2.5. Others
- 9.3. Market Analysis, Insights and Forecast - by End User
- 9.3.1. Hospitals
- 9.3.2. Diagnostics Centers
- 9.3.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 10. South America Pituitary Cancer Diagnostics Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 10.1.1. Imaging Test
- 10.1.1.1. MRI
- 10.1.1.2. CT Scan
- 10.1.1.3. Other
- 10.1.2. Lumbar Puncture
- 10.1.3. Biopsy
- 10.1.4. Molecular Testing
- 10.1.5. Others
- 10.1.1. Imaging Test
- 10.2. Market Analysis, Insights and Forecast - by Tumor Type
- 10.2.1. Metastatic
- 10.2.2. Meningioma
- 10.2.3. Glioblastoma
- 10.2.4. Pituitary Endoma
- 10.2.5. Others
- 10.3. Market Analysis, Insights and Forecast - by End User
- 10.3.1. Hospitals
- 10.3.2. Diagnostics Centers
- 10.3.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Diagnostic Type
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Siemens Healthineers
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Hologic Inc
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 GE Healthcare
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Hitachi Ltd
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Koninklijke Philips N V
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Fujifilm Corporation
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Thermo Fisher Scientific
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 NantOmics
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.1 Siemens Healthineers
List of Figures
- Figure 1: Global Pituitary Cancer Diagnostics Industry Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 3: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 4: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 5: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 6: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 7: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 8: North America Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 9: North America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 11: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 12: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 13: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 14: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 15: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 16: Europe Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 17: Europe Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 19: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 20: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 21: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 22: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 23: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 24: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 25: Asia Pacific Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 27: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 28: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 29: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 30: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 31: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 32: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 33: Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by Diagnostic Type 2025 & 2033
- Figure 35: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Diagnostic Type 2025 & 2033
- Figure 36: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by Tumor Type 2025 & 2033
- Figure 37: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Tumor Type 2025 & 2033
- Figure 38: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by End User 2025 & 2033
- Figure 39: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 40: South America Pituitary Cancer Diagnostics Industry Revenue (Million), by Country 2025 & 2033
- Figure 41: South America Pituitary Cancer Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 2: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 3: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 4: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 5: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 6: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 7: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 8: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 9: United States Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 10: Canada Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 11: Mexico Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 12: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 13: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 14: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 15: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Germany Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 17: United Kingdom Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 18: France Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 19: Italy Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 20: Spain Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 21: Rest of Europe Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 22: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 23: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 24: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 25: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 26: China Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 27: Japan Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 28: India Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 29: Australia Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 30: South Korea Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 31: Rest of Asia Pacific Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 32: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 33: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 34: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 35: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 36: GCC Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 37: South Africa Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 38: Rest of Middle East and Africa Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 39: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Diagnostic Type 2020 & 2033
- Table 40: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Tumor Type 2020 & 2033
- Table 41: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by End User 2020 & 2033
- Table 42: Global Pituitary Cancer Diagnostics Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 43: Brazil Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 44: Argentina Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 45: Rest of South America Pituitary Cancer Diagnostics Industry Revenue (Million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Pituitary Cancer Diagnostics Industry?
The projected CAGR is approximately 8.30%.
2. Which companies are prominent players in the Pituitary Cancer Diagnostics Industry?
Key companies in the market include Siemens Healthineers, Hologic Inc, GE Healthcare, Hitachi Ltd, Koninklijke Philips N V, Fujifilm Corporation, Thermo Fisher Scientific, NantOmics.
3. What are the main segments of the Pituitary Cancer Diagnostics Industry?
The market segments include Diagnostic Type, Tumor Type, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD 2.27 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Neurological Disorders and Cancers; Rising Number of Smokers.
6. What are the notable trends driving market growth?
MRI segment is Expected to Hold a Significant Share in the Brain Tumor Diagnostics Market.
7. Are there any restraints impacting market growth?
High Costs Associated with Diagnosis.
8. Can you provide examples of recent developments in the market?
February 2023: Roche announced the launch of the IDH1 R132H (MRQ-67) Rabbit Monoclonal Primary Antibody and the ATRX Rabbit Polyclonal Antibody to identify mutation status in patients diagnosed with brain cancer.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Pituitary Cancer Diagnostics Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Pituitary Cancer Diagnostics Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Pituitary Cancer Diagnostics Industry?
To stay informed about further developments, trends, and reports in the Pituitary Cancer Diagnostics Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database


Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


