Key Insights
The Brazilian respiratory devices market is poised for significant expansion, projected to reach an estimated USD 343.74 million by 2024, with a robust Compound Annual Growth Rate (CAGR) of 6.5% through 2033. This growth is primarily fueled by the increasing prevalence of respiratory diseases such as asthma, COPD, and sleep apnea, driven by factors like air pollution, smoking, and an aging population. The rising healthcare expenditure in Brazil, coupled with a growing awareness of respiratory health management, is further bolstering market demand. The diagnostic and monitoring segment, encompassing essential devices like spirometers and pulse oximeters, is expected to witness steady growth as early detection and continuous monitoring become paramount. Similarly, the therapeutic devices segment, including CPAP, BiPAP, and ventilators, is crucial for managing chronic respiratory conditions, contributing significantly to the overall market expansion. Furthermore, the increasing adoption of home healthcare solutions for respiratory management is a key trend shaping the market landscape.
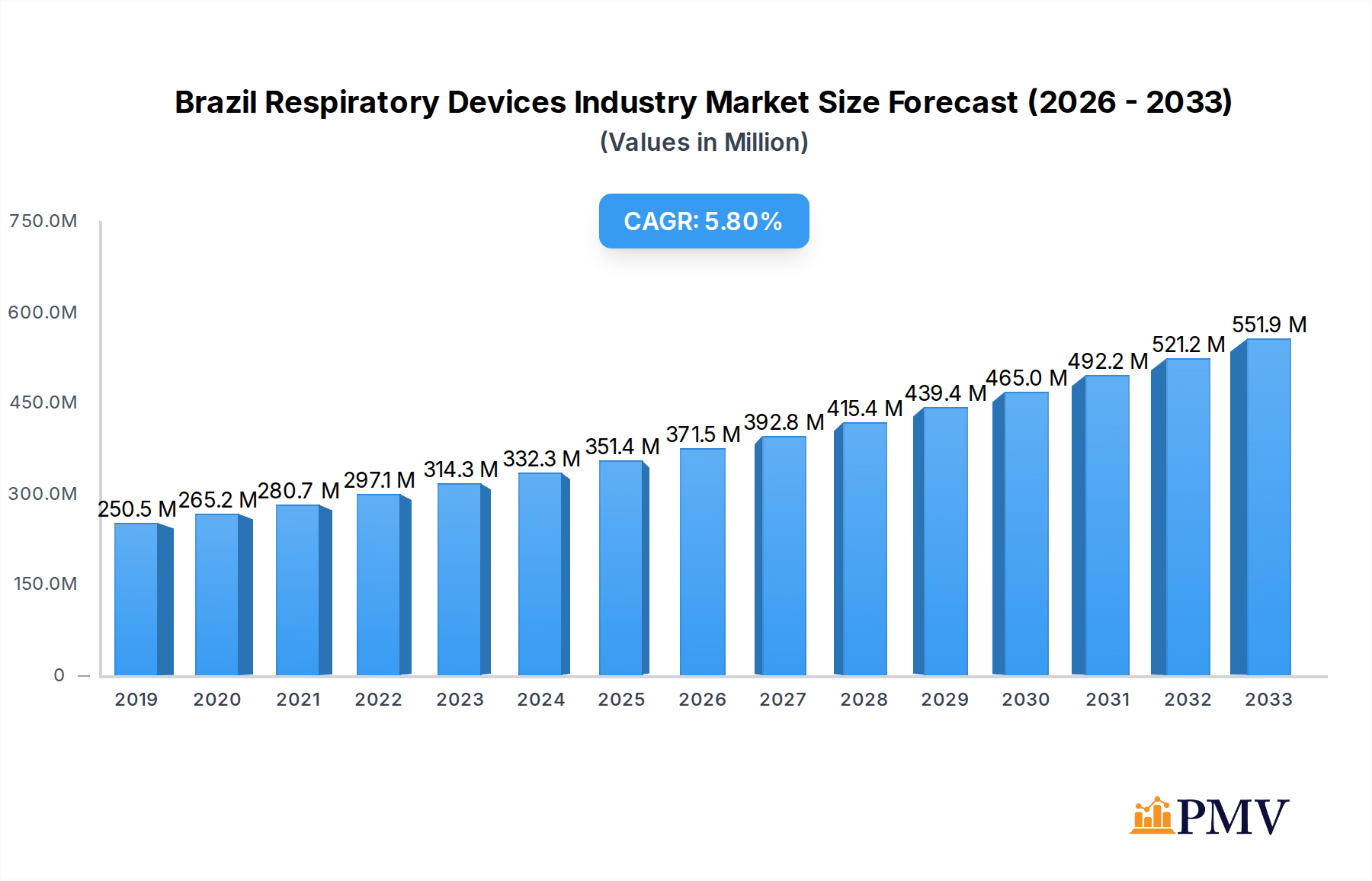
Brazil Respiratory Devices Industry Market Size (In Million)

The market's trajectory is also influenced by evolving healthcare infrastructure and technological advancements. Investments in advanced respiratory care technologies, including portable diagnostic tools and more sophisticated therapeutic devices, are expected to drive market penetration. While the market shows strong growth potential, certain restraints, such as the high cost of advanced medical devices and limited reimbursement policies in some segments, could pose challenges. However, the growing demand for advanced respiratory solutions and the increasing focus on preventive healthcare are anticipated to outweigh these constraints. The competitive landscape is characterized by the presence of both global and local players, fostering innovation and driving market competitiveness. Strategic collaborations and product launches are expected to be key strategies for companies operating in this dynamic market.
Brazil Respiratory Devices Industry Company Market Share

This comprehensive report delves into the dynamic Brazil Respiratory Devices Industry, providing in-depth analysis and actionable insights for stakeholders. Covering a study period from 2019 to 2033, with a base and estimated year of 2025, this report offers a detailed forecast for the period 2025–2033 and a retrospective view of the historical period 2019–2024. We examine market structure, competitive dynamics, key trends, dominant segments, product innovations, growth drivers, challenges, leading players, and crucial industry developments.
Brazil Respiratory Devices Industry Market Structure & Competitive Dynamics
The Brazil Respiratory Devices Industry is characterized by a moderate to high level of market concentration, with a few key global players holding significant market shares. Key companies like Hamilton Medical, Drägerwerk AG & Co KGaA, BD (Becton Dickinson and Company), Masimo, Medtronic, Fisher & Paykel Healthcare Limited, Koninklijke Philips NV, ResMed Inc, Chart Industries, and General Electric Company (GE HealthCare) are prominent. The innovation ecosystem is robust, driven by continuous advancements in medical technology, particularly in areas like artificial intelligence for diagnostics and miniaturization of therapeutic devices. Brazil's regulatory framework, spearheaded by ANVISA, plays a crucial role in shaping market entry and product approval. Recent updates to Brazilian Good Manufacturing Practices (BGMP) through RDC 665/2022 aim to enhance product quality and safety, impacting all manufacturers. Product substitutes, while present in some categories, are often limited by the specialized nature of respiratory care equipment. End-user trends are increasingly influenced by the rising prevalence of respiratory diseases, an aging population, and a growing demand for home healthcare solutions. Merger and acquisition (M&A) activities, while not extensively detailed here without specific deal values, are anticipated to continue as companies seek to expand their product portfolios and market reach within Brazil. Estimated market share shifts are carefully analyzed throughout the report to provide a clear competitive landscape.
Brazil Respiratory Devices Industry Industry Trends & Insights
The Brazil Respiratory Devices Industry is experiencing a significant growth trajectory, fueled by a confluence of compelling trends and insights. The increasing prevalence of chronic respiratory diseases such as COPD, asthma, and sleep apnea, coupled with a rapidly aging population, are primary market growth drivers. Furthermore, the growing awareness among healthcare professionals and patients about the benefits of early diagnosis and effective management of respiratory conditions is spurring demand for advanced diagnostic and therapeutic devices. Technological advancements are a cornerstone of this industry's evolution. Innovations in connected devices, wearable respiratory monitors, and AI-powered diagnostic tools are revolutionizing patient care and remote monitoring capabilities. The rise of telemedicine and home healthcare services further accentuates the demand for portable, user-friendly, and increasingly intelligent respiratory devices. Consumer preferences are shifting towards solutions that offer convenience, comfort, and improved quality of life, driving innovation in areas like quieter and more ergonomic CPAP machines and advanced nebulizer technologies. The competitive dynamics are characterized by intense innovation, strategic partnerships, and a focus on expanding distribution networks to reach a wider patient base across Brazil's vast geography. The projected Compound Annual Growth Rate (CAGR) for the Brazil Respiratory Devices Industry is robust, reflecting the sustained demand and market penetration of essential respiratory care solutions. Market penetration of advanced respiratory devices is on an upward trend as healthcare infrastructure continues to develop and accessibility improves. The industry is poised for sustained expansion, driven by these multifaceted trends.
Dominant Markets & Segments in Brazil Respiratory Devices Industry
The Brazil Respiratory Devices Industry exhibits distinct dominance across various segments, driven by specific market needs and regulatory influences.
Therapeutic Devices represent a particularly strong market segment.
- Ventilators: The demand for mechanical ventilators remains high, driven by the ongoing need for critical care in hospitals, particularly in light of public health emergencies and the management of severe respiratory illnesses. Economic policies that prioritize healthcare infrastructure development directly impact the procurement of these life-saving devices.
- CPAP Devices & BiPAP Devices: With the increasing diagnosis of sleep apnea and other sleep-disordered breathing conditions, the market for Continuous Positive Airway Pressure (CPAP) and Bilevel Positive Airway Pressure (BiPAP) devices is experiencing substantial growth. An aging population and a sedentary lifestyle contribute to the rising incidence of these conditions.
- Oxygen Concentrators: The expansion of home healthcare services and the growing need for supplemental oxygen therapy for conditions like COPD and chronic heart failure are propelling the growth of the oxygen concentrator market. Manufacturers are focusing on developing more efficient and portable models to cater to this demand.
Diagnostic and Monitoring Devices also hold significant sway.
- Pulse Oximeters: Their essential role in monitoring blood oxygen saturation levels, especially in home care settings and emergency response, makes them a continuously high-demand product. Their affordability and ease of use contribute to widespread adoption.
- Spirometers: These devices are crucial for diagnosing and monitoring lung function in clinical settings and are seeing increased adoption in primary care to facilitate early detection of respiratory diseases.
- Sleep Test Devices: The growing awareness of the health implications of sleep disorders is driving the market for home sleep apnea testing devices, offering a convenient alternative to in-lab polysomnography.
Disposables are fundamental to the sustained use of respiratory devices.
- Masks and Breathing Circuits: The consistent need for replacement masks, tubing, and filters for therapeutic devices ensures a steady and growing market for these consumables. The emphasis on hygiene and infection control further reinforces this segment.
The dominance within these segments is further influenced by Brazil's diverse economic landscape and regional healthcare infrastructure variations. Urban centers tend to have higher adoption rates for advanced technologies, while efforts are underway to expand access to essential respiratory care in remote areas.
Brazil Respiratory Devices Industry Product Innovations
Product innovations within the Brazil Respiratory Devices Industry are significantly enhancing patient care and market competitiveness. Advancements focus on miniaturization, connectivity, and user-friendliness, leading to more portable and integrated solutions. For instance, the development of smart nebulizers that connect to mobile apps for personalized treatment and adherence monitoring is gaining traction. Innovations in ventilator technology are leading to more adaptable modes and improved patient comfort, crucial for prolonged ventilation. The introduction of advanced filtration systems in CPAP masks and breathing circuits addresses hygiene and comfort concerns. These technological leaps are driven by a desire to improve therapeutic efficacy, reduce the burden of respiratory diseases, and enable seamless integration into evolving healthcare ecosystems, including telemedicine.
Report Segmentation & Scope
This report meticulously segments the Brazil Respiratory Devices Industry to provide a granular understanding of market dynamics. The scope encompasses:
- Diagnostic and Monitoring Devices: This broad category includes Spirometers for lung function testing, Sleep Test Devices for diagnosing sleep disorders, Peak Flow Meters for asthma management, Pulse Oximeters for oxygen saturation monitoring, Capnographs for end-tidal CO2 measurement, and Other Diagnostic and Monitoring Devices.
- Therapeutic Devices: This vital segment comprises CPAP Devices and BiPAP Devices for sleep apnea and other respiratory conditions, Humidifiers for adding moisture to inhaled air, Nebulizers for delivering medication directly to the lungs, Oxygen Concentrators for providing supplemental oxygen, Ventilators for mechanical breathing support, Inhalers for drug delivery, and Other Therapeutic Devices.
- Disposables: This segment includes essential consumables like Masks for CPAP/BiPAP devices and ventilators, Breathing Circuits for ventilator systems, and Other Disposables such as filters and tubing.
Each segment is analyzed with its current market size, projected growth rates, and the competitive landscape influencing its expansion.
Key Drivers of Brazil Respiratory Devices Industry Growth
Several pivotal factors are propelling the growth of the Brazil Respiratory Devices Industry. The escalating burden of respiratory diseases, including COPD and asthma, due to factors like air pollution and lifestyle changes, is a significant demand generator. An aging demographic further amplifies the need for respiratory care. Technological advancements, such as the integration of AI in diagnostics and the development of connected devices for remote patient monitoring, are creating new market opportunities. Government initiatives aimed at improving healthcare infrastructure and increasing access to medical devices, particularly in underserved regions, are crucial accelerators. The expanding adoption of home healthcare models, driven by patient preference and cost-effectiveness, also boosts demand for portable and user-friendly respiratory devices.
Challenges in the Brazil Respiratory Devices Industry Sector
Despite its growth, the Brazil Respiratory Devices Industry faces several significant challenges. Regulatory hurdles, including the rigorous approval processes by ANVISA and evolving compliance standards, can impact the speed of market entry for new products. Economic volatility and currency fluctuations can affect import costs and affordability for consumers and healthcare providers. Supply chain disruptions, exacerbated by global events, can lead to shortages and price increases for critical components and finished goods. Intense competition among global and local players can put pressure on pricing. Furthermore, disparities in healthcare access and infrastructure across Brazil's vast territory can limit the penetration of advanced respiratory devices in rural and remote areas.
Leading Players in the Brazil Respiratory Devices Industry Market
- Hamilton Medical
- Drägerwerk AG & Co KGaA
- BD (Becton Dickinson and Company)
- Masimo
- Medtronic
- Fisher & Paykel Healthcare Limited
- Koninklijke Philips NV
- ResMed Inc
- Chart Industries
- General Electric Company (GE HealthCare)
Key Developments in Brazil Respiratory Devices Industry Sector
- April 2022: ANVISA, Brazil's medical device market regulator, issued a new resolution updating Brazilian Good Manufacturing Practices (BGMP) for medical devices. ANVISA's RDC 665/2022 replaced previous regulations, including RDC 16/2013 and IN 08/2013, emphasizing stricter quality control and compliance for manufacturers.
- April 2022: Global oxygen equipment manufacturer CAIRE Inc. expanded its portfolio of solutions in Latin America by launching Companion 5 and NewLife Intensity 10 stationary oxygen concentrators in Brazil, following anticipated approval by the Brazilian Health Regulatory Agency (Anvisa), enhancing the availability of oxygen therapy options.
Strategic Brazil Respiratory Devices Industry Market Outlook
The strategic outlook for the Brazil Respiratory Devices Industry is overwhelmingly positive, marked by significant growth accelerators. The increasing focus on preventative healthcare and early diagnosis of respiratory conditions, coupled with the robust expansion of home healthcare services, presents a substantial opportunity for market players. Investments in digital health technologies, including connected devices and AI-driven analytics, will further enhance patient outcomes and operational efficiencies. Favorable regulatory reforms aimed at streamlining market access and promoting innovation are anticipated to encourage further investment. Strategic partnerships between technology providers and healthcare institutions will be crucial for developing integrated respiratory care solutions. The industry is poised for sustained expansion driven by unmet medical needs and a growing emphasis on improving the quality of life for individuals suffering from respiratory ailments.
Brazil Respiratory Devices Industry Segmentation
-
1. Diagnostic and Monitoring Devices
- 1.1. Spirometers
- 1.2. Sleep Test Devices
- 1.3. Peak Flow Meters
- 1.4. Pulse Oximeters
- 1.5. Capnographs
- 1.6. Other Diagnostic and Monitoring Devices
-
2. Therapeutic Devices
- 2.1. CPAP Devices
- 2.2. BiPAP Devices
- 2.3. Humidifiers
- 2.4. Nebulizers
- 2.5. Oxygen Concentrators
- 2.6. Ventilators
- 2.7. Inhalers
- 2.8. Other Therapeutic Devices
-
3. Disposables
- 3.1. Masks
- 3.2. Breathing Circuit
- 3.3. Other Disposables
Brazil Respiratory Devices Industry Segmentation By Geography
- 1. Brazil
Brazil Respiratory Devices Industry Regional Market Share

Geographic Coverage of Brazil Respiratory Devices Industry
Brazil Respiratory Devices Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic and Monitoring Devices
- 5.1.1. Spirometers
- 5.1.2. Sleep Test Devices
- 5.1.3. Peak Flow Meters
- 5.1.4. Pulse Oximeters
- 5.1.5. Capnographs
- 5.1.6. Other Diagnostic and Monitoring Devices
- 5.2. Market Analysis, Insights and Forecast - by Therapeutic Devices
- 5.2.1. CPAP Devices
- 5.2.2. BiPAP Devices
- 5.2.3. Humidifiers
- 5.2.4. Nebulizers
- 5.2.5. Oxygen Concentrators
- 5.2.6. Ventilators
- 5.2.7. Inhalers
- 5.2.8. Other Therapeutic Devices
- 5.3. Market Analysis, Insights and Forecast - by Disposables
- 5.3.1. Masks
- 5.3.2. Breathing Circuit
- 5.3.3. Other Disposables
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. Brazil
- 5.1. Market Analysis, Insights and Forecast - by Diagnostic and Monitoring Devices
- 6. Brazil Respiratory Devices Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Diagnostic and Monitoring Devices
- 6.1.1. Spirometers
- 6.1.2. Sleep Test Devices
- 6.1.3. Peak Flow Meters
- 6.1.4. Pulse Oximeters
- 6.1.5. Capnographs
- 6.1.6. Other Diagnostic and Monitoring Devices
- 6.2. Market Analysis, Insights and Forecast - by Therapeutic Devices
- 6.2.1. CPAP Devices
- 6.2.2. BiPAP Devices
- 6.2.3. Humidifiers
- 6.2.4. Nebulizers
- 6.2.5. Oxygen Concentrators
- 6.2.6. Ventilators
- 6.2.7. Inhalers
- 6.2.8. Other Therapeutic Devices
- 6.3. Market Analysis, Insights and Forecast - by Disposables
- 6.3.1. Masks
- 6.3.2. Breathing Circuit
- 6.3.3. Other Disposables
- 6.1. Market Analysis, Insights and Forecast - by Diagnostic and Monitoring Devices
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Hamilton Medical
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Drägerwerk AG & Co KGaA
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 BD (Becton Dickinson and Company)*List Not Exhaustive
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Masimo
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Medtronic
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Fisher & Paykel Healthcare Limited
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Koninklijke Philips NV
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 ResMed Inc
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Chart Industries
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 General Electric Company (GE HealthCare)
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.1 Hamilton Medical
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: Brazil Respiratory Devices Industry Revenue Breakdown (million, %) by Product 2025 & 2033
- Figure 2: Brazil Respiratory Devices Industry Share (%) by Company 2025
List of Tables
- Table 1: Brazil Respiratory Devices Industry Revenue million Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 2: Brazil Respiratory Devices Industry Volume K Units Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 3: Brazil Respiratory Devices Industry Revenue million Forecast, by Therapeutic Devices 2020 & 2033
- Table 4: Brazil Respiratory Devices Industry Volume K Units Forecast, by Therapeutic Devices 2020 & 2033
- Table 5: Brazil Respiratory Devices Industry Revenue million Forecast, by Disposables 2020 & 2033
- Table 6: Brazil Respiratory Devices Industry Volume K Units Forecast, by Disposables 2020 & 2033
- Table 7: Brazil Respiratory Devices Industry Revenue million Forecast, by Region 2020 & 2033
- Table 8: Brazil Respiratory Devices Industry Volume K Units Forecast, by Region 2020 & 2033
- Table 9: Brazil Respiratory Devices Industry Revenue million Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 10: Brazil Respiratory Devices Industry Volume K Units Forecast, by Diagnostic and Monitoring Devices 2020 & 2033
- Table 11: Brazil Respiratory Devices Industry Revenue million Forecast, by Therapeutic Devices 2020 & 2033
- Table 12: Brazil Respiratory Devices Industry Volume K Units Forecast, by Therapeutic Devices 2020 & 2033
- Table 13: Brazil Respiratory Devices Industry Revenue million Forecast, by Disposables 2020 & 2033
- Table 14: Brazil Respiratory Devices Industry Volume K Units Forecast, by Disposables 2020 & 2033
- Table 15: Brazil Respiratory Devices Industry Revenue million Forecast, by Country 2020 & 2033
- Table 16: Brazil Respiratory Devices Industry Volume K Units Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Brazil Respiratory Devices Industry?
The projected CAGR is approximately 6.5%.
2. Which companies are prominent players in the Brazil Respiratory Devices Industry?
Key companies in the market include Hamilton Medical, Drägerwerk AG & Co KGaA, BD (Becton Dickinson and Company)*List Not Exhaustive, Masimo, Medtronic, Fisher & Paykel Healthcare Limited, Koninklijke Philips NV, ResMed Inc, Chart Industries, General Electric Company (GE HealthCare).
3. What are the main segments of the Brazil Respiratory Devices Industry?
The market segments include Diagnostic and Monitoring Devices, Therapeutic Devices, Disposables.
4. Can you provide details about the market size?
The market size is estimated to be USD 343.74 million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Respiratory Disorders Such as COPD and TB; Technological Advancements and Increasing Applications in Homecare Setting.
6. What are the notable trends driving market growth?
Inhalers Expected to Grow Fastest During the Forecast Period.
7. Are there any restraints impacting market growth?
High Cost of Devices.
8. Can you provide examples of recent developments in the market?
April 2022: ANVISA, Brazil's medical device market regulator, issued a new resolution updating Brazilian Good Manufacturing Practices (BGMP) for medical devices. ANVISA's RDC 665/2022 replaced previous regulations, including RDC 16/2013 and IN 08/2013.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Brazil Respiratory Devices Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Brazil Respiratory Devices Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Brazil Respiratory Devices Industry?
To stay informed about further developments, trends, and reports in the Brazil Respiratory Devices Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


