Key Insights
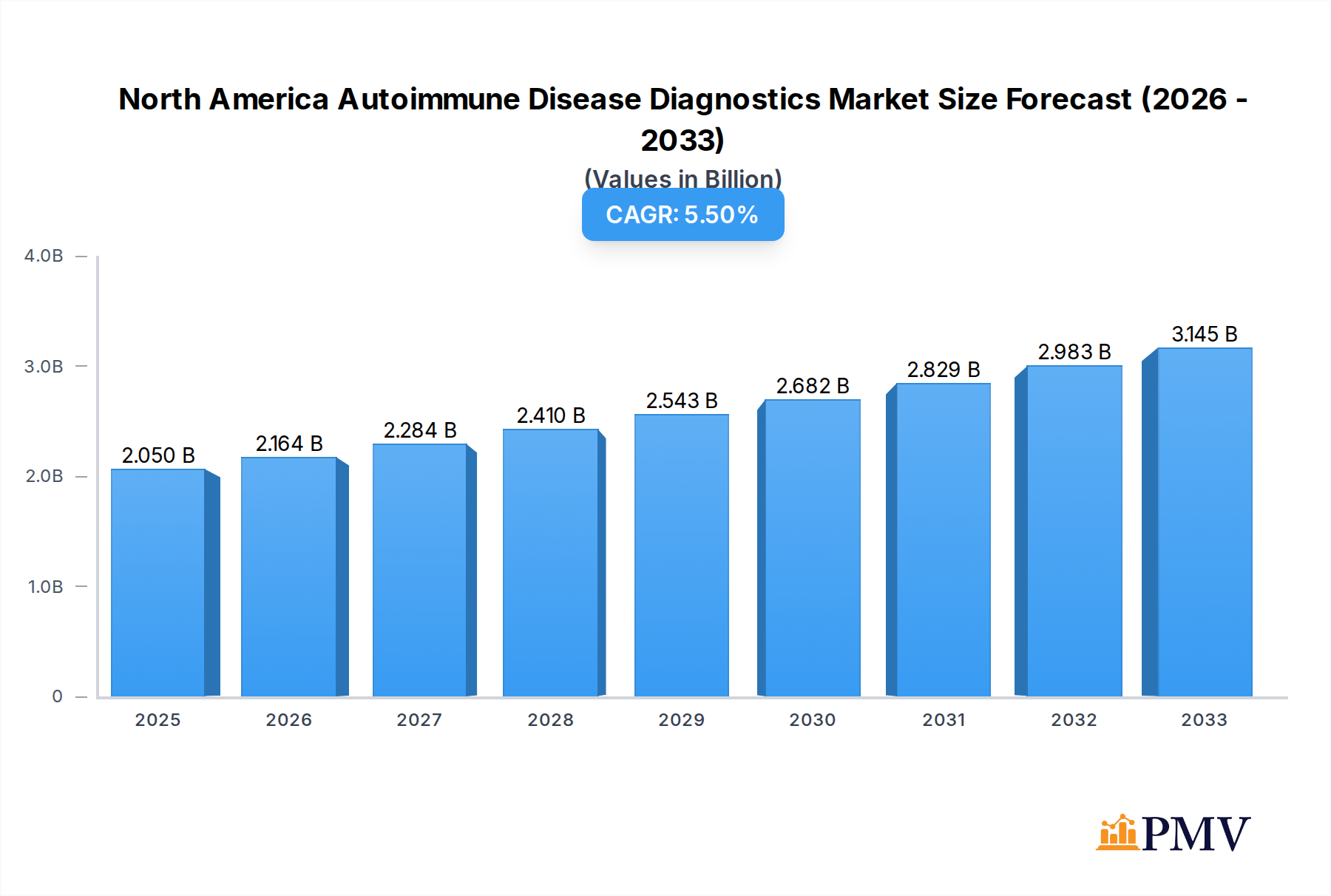
The North America autoimmune disease diagnostics market is poised for significant expansion, projected to reach USD 2.05 billion by 2025, with a robust Compound Annual Growth Rate (CAGR) of 5.53%. This growth is fueled by an increasing prevalence of autoimmune conditions, a rising awareness among both patients and healthcare professionals, and advancements in diagnostic technologies. The market is segmented into disease types, including Systemic Autoimmune Diseases like Rheumatoid Arthritis, Psoriasis, and Systemic Lupus Erythematosus (SLE), and Localized Autoimmune Diseases such as Inflammatory Bowel Disease and Type 1 Diabetes. The growing understanding of the complex interplay of genetic and environmental factors contributing to these diseases is driving the demand for more sophisticated and accurate diagnostic tools. Furthermore, the development of highly sensitive and specific assays, including antibody tests and inflammatory markers, is enabling earlier and more precise diagnoses, which is crucial for effective disease management and improved patient outcomes.
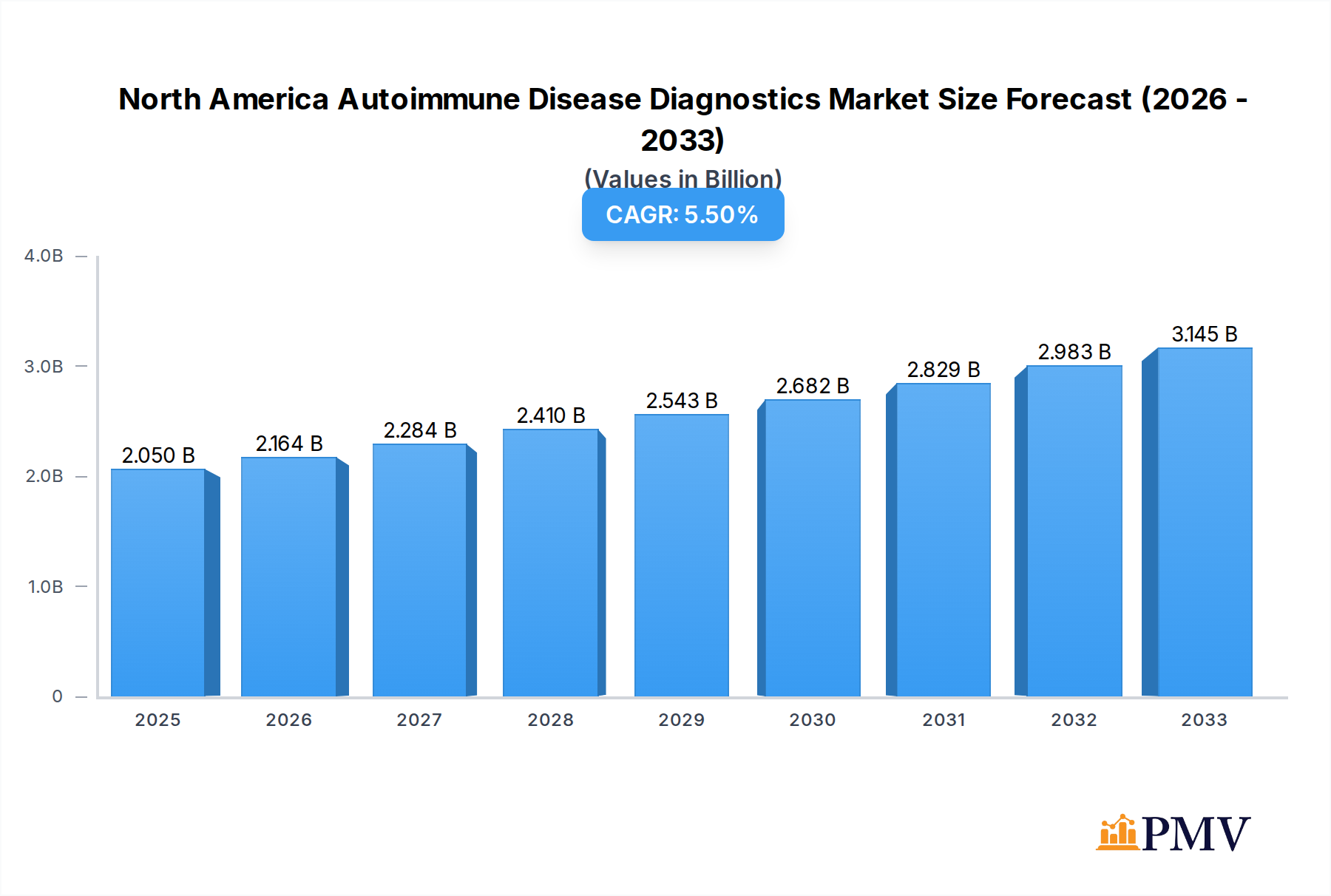
North America Autoimmune Disease Diagnostics Market Market Size (In Billion)

Key drivers for this market's ascent include the rising incidence of chronic autoimmune disorders, escalating healthcare expenditure, and continuous innovation in diagnostic methodologies. Trends such as the increasing adoption of multiplex assays for simultaneous detection of multiple autoantibodies, the integration of artificial intelligence in diagnostic interpretation, and a greater focus on personalized medicine are shaping the market landscape. However, challenges such as the high cost of advanced diagnostic tests and the need for standardization across different regions could pose some restraints. Despite these, the strong emphasis on early detection and proactive management of autoimmune diseases, coupled with the expanding product portfolios of leading market players like Abbott Laboratories, Siemens Healthineers Inc., and Thermo Fisher Scientific, are expected to propel the market forward. The United States is anticipated to be the dominant region, owing to its advanced healthcare infrastructure and high prevalence rates.
North America Autoimmune Disease Diagnostics Market Company Market Share

This detailed report delves into the dynamic North America Autoimmune Disease Diagnostics Market, offering in-depth analysis and actionable insights for stakeholders. Leveraging a robust study period of 2019–2033, with a base year of 2025, the report provides critical information on market structure, trends, dominant segments, product innovations, and future outlook.
North America Autoimmune Disease Diagnostics Market Market Structure & Competitive Dynamics
The North America Autoimmune Disease Diagnostics Market is characterized by a moderate to high level of concentration, driven by the presence of established global players and the increasing complexity of diagnostic technologies. Innovation ecosystems are vibrant, fueled by continuous research and development in areas like biomarker discovery and advanced immunoassay techniques. Regulatory frameworks, primarily governed by bodies like the U.S. Food and Drug Administration (FDA) and Health Canada, play a crucial role in market entry and product approval. The report analyzes potential product substitutes, such as advanced imaging techniques and clinical assessment tools, while also highlighting the rising influence of end-user trends, including patient demand for personalized diagnostics and early disease detection.
- Market Concentration: Key players hold significant market share, but the landscape is evolving with the emergence of specialized diagnostic companies.
- Innovation Ecosystems: Driven by academic research, biotechnology advancements, and venture capital funding.
- Regulatory Frameworks: Strict but essential for ensuring diagnostic accuracy and patient safety.
- Product Substitutes: While diagnostic tests remain central, integrated healthcare approaches are gaining traction.
- End-User Trends: Growing emphasis on proactive health management and direct-to-consumer testing for certain autoimmune markers.
- M&A Activities: Strategic acquisitions and partnerships are evident as companies seek to expand their product portfolios and market reach. For instance, the acquisition of Euroimmun AG by PerkinElmer Inc. exemplifies consolidation in the diagnostics sector. While specific M&A deal values are proprietary, they are estimated to be in the hundreds of millions of dollars, reflecting the strategic importance of autoimmune diagnostic capabilities.
North America Autoimmune Disease Diagnostics Market Industry Trends & Insights
The North America Autoimmune Disease Diagnostics Market is poised for significant growth, driven by a confluence of factors including rising prevalence of autoimmune disorders, increasing healthcare expenditure, and rapid technological advancements. The market is projected to witness a Compound Annual Growth Rate (CAGR) of approximately 7.5% during the forecast period. This growth is fueled by a greater understanding of the pathogenesis of autoimmune diseases and the subsequent demand for more accurate and timely diagnostic solutions. Technological disruptions, such as the advent of next-generation sequencing (NGS) for genetic predisposition analysis and the refinement of multiplex immunoassays, are revolutionizing disease detection. Consumer preferences are increasingly shifting towards personalized medicine, with patients seeking diagnostic tests that can predict disease risk and guide treatment decisions. Competitive dynamics are intensifying, with companies investing heavily in R&D to develop novel biomarkers and more sensitive diagnostic platforms. The penetration of advanced diagnostic techniques in primary care settings is also expanding, further contributing to market growth.
- Market Growth Drivers: Increased prevalence of autoimmune diseases, growing awareness, and advancements in diagnostic technologies.
- Technological Disruptions: Development of highly sensitive immunoassay platforms, automation in laboratories, and integration of AI for data analysis.
- Consumer Preferences: Demand for early detection, risk assessment, and personalized treatment strategies.
- Competitive Dynamics: Fierce competition characterized by strategic partnerships, product launches, and acquisitions.
- Market Penetration: Expanding adoption of advanced diagnostic tests across various healthcare settings, including specialized clinics and research institutions.
- Estimated Market Size: The North America Autoimmune Disease Diagnostics Market is estimated to reach approximately $6,500 Million by 2025, with strong growth projected through 2033.
Dominant Markets & Segments in North America Autoimmune Disease Diagnostics Market
The United States dominates the North America Autoimmune Disease Diagnostics Market, accounting for over 70% of the total market share. This dominance is attributed to factors such as a robust healthcare infrastructure, high healthcare spending, a large patient population, and a strong presence of leading diagnostic companies. Canada and Mexico represent significant, albeit smaller, markets with growing potential.
Within the Disease Type segmentation, Systemic Autoimmune Diseases command the largest market share. This segment includes:
- Rheumatoid Arthritis (RA): Characterized by a high prevalence and significant demand for diagnostic tests to monitor disease activity and treatment efficacy.
- Psoriasis: A chronic inflammatory skin condition with increasing diagnostic needs, especially for differentiating it from other dermatological disorders.
- Systemic Lupus Erythematosus (SLE): A complex autoimmune disease requiring sophisticated diagnostic panels.
- Multiple Sclerosis (MS): A neurological disorder with a growing focus on early and accurate diagnosis to improve patient outcomes.
- Other Disease Types: Encompasses a range of other systemic autoimmune conditions.
The Localized Autoimmune Diseases segment is also a critical area of growth, with notable sub-segments including:
- Inflammatory Bowel Disease (IBD): Including Crohn's disease and ulcerative colitis, which require specialized diagnostic markers.
- Type 1 Diabetes: A metabolic autoimmune disorder with established diagnostic protocols.
- Thyroid Autoimmune Diseases: Such as Hashimoto's thyroiditis and Graves' disease, which are prevalent and require specific antibody testing.
- Other Localized Autoimmune Diseases: Covering a spectrum of conditions affecting specific organs.
In terms of Diagnostic Tests, Immunologic Assays hold the largest market share due to their accuracy and versatility in detecting autoantibodies. This segment includes:
Antibody Tests: The cornerstone of autoimmune disease diagnosis, identifying specific autoantibodies associated with different conditions.
Inflammatory Markers: Such as C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR), which indicate inflammation but are not specific to autoimmune diseases.
Regular Laboratory Tests: Including complete blood counts (CBCs) and basic metabolic panels, providing essential baseline information.
Other Tests: Encompassing emerging diagnostic technologies and specialized panels.
Dominant Region: United States, driven by advanced healthcare infrastructure and high R&D investment.
Leading Disease Type: Systemic Autoimmune Diseases, owing to their widespread prevalence and diagnostic complexity.
Key Diagnostic Test Segment: Immunologic Assays, due to their specificity and efficacy in detecting autoantibodies.
Market Drivers for Dominance:
- United States: High per capita healthcare spending, early adoption of new technologies, and a large patient pool.
- Systemic Autoimmune Diseases: Rising incidence rates and the need for comprehensive diagnostic panels.
- Immunologic Assays: Development of highly sensitive and specific antibody detection methods.
North America Autoimmune Disease Diagnostics Market Product Innovations
Product innovations in the North America Autoimmune Disease Diagnostics Market are centered on enhancing sensitivity, specificity, and efficiency. Companies are developing novel multiplex assays capable of detecting multiple autoantibodies simultaneously, thereby streamlining the diagnostic process and reducing turnaround times. Advances in automation and digital integration are transforming laboratory workflows, enabling higher throughput and improved data management. The development of point-of-care diagnostic solutions for certain autoimmune markers is also an emerging trend, promising faster results and improved patient access. These innovations are crucial for early disease detection, differential diagnosis, and personalized treatment monitoring.
Report Segmentation & Scope
This report provides a comprehensive segmentation of the North America Autoimmune Disease Diagnostics Market. The analysis is meticulously categorized by Disease Type, encompassing both Systemic Autoimmune Disease (including Rheumatoid Arthritis, Psoriasis, Systemic Lupus Erythematosus (SLE), Multiple Sclerosis, and Other Disease Types) and Localized Autoimmune Disease (including Inflammatory Bowel disease, Type 1 Diabetes, Thyroid, and Other Localized Autoimmune Diseases). Further segmentation is provided by Diagnostic Test, covering Regular Laboratory Tests, Inflammatory Markers, Immunologic Assays, Antibody Tests, and Other Tests. Geographically, the market is analyzed across the United States, Canada, and Mexico. Growth projections and market sizes are detailed for each segment, providing a granular understanding of the competitive landscape and future opportunities.
Key Drivers of North America Autoimmune Disease Diagnostics Market Growth
The North America Autoimmune Disease Diagnostics Market is propelled by several key drivers. The increasing prevalence of autoimmune diseases, partly due to environmental factors and lifestyle changes, is a primary growth accelerator. Technological advancements in diagnostic platforms, such as enhanced immunoassay sensitivity and the development of novel biomarkers, are crucial. Growing awareness among the public and healthcare professionals regarding autoimmune conditions leads to increased demand for diagnostic testing. Furthermore, favorable reimbursement policies for diagnostic procedures and supportive government initiatives aimed at improving healthcare access contribute significantly to market expansion. The ongoing pursuit of personalized medicine also fuels the demand for more precise and individualized diagnostic solutions.
Challenges in the North America Autoimmune Disease Diagnostics Market Sector
Despite its robust growth, the North America Autoimmune Disease Diagnostics Market faces several challenges. High development costs for novel diagnostic tests, coupled with stringent regulatory approval processes, can hinder market entry for new products. Reimbursement complexities and variations across different healthcare systems can impact the adoption rates of advanced diagnostics. The availability of skilled laboratory professionals and the need for specialized training can also pose operational challenges. Furthermore, competition from established players and the threat of pricing pressures can impact profitability. Supply chain disruptions, as witnessed in recent global events, can also affect the availability of critical reagents and components, impacting the overall diagnostic workflow.
Leading Players in the North America Autoimmune Disease Diagnostics Market Market
- Oncimmune
- HYCOR Biomedical
- Grifols SA
- Werfen Group
- Abbott Laboratories
- Siemens Healthineers Inc
- Bio-rad Laboratories
- Myriad Genetics
- Euroimmun AG (Perkinelmer Inc )
- F Hoffmann-la Roche
- Thermo Fisher Scientific
- Trinity Biotech
Key Developments in North America Autoimmune Disease Diagnostics Market Sector
- February 2023: Edesa Biotech received approval from Health Canada for a phase II clinical trial of its EB06 monoclonal antibody candidate to treat vitiligo, a life-altering autoimmune disease. This development highlights advancements in therapeutic targets and the interconnectedness of diagnostic and therapeutic research.
- June 2022: Thermo Scientific received United States FDA clearance for the EliA RNA Pol III and EliA Rib-P tests for aiding in the diagnosis of systemic sclerosis and systemic lupus erythematosus (SLE). This clearance signifies the introduction of more specific and sensitive diagnostic tools for complex autoimmune conditions, enhancing diagnostic accuracy.
Strategic North America Autoimmune Disease Diagnostics Market Market Outlook
The strategic outlook for the North America Autoimmune Disease Diagnostics Market is exceptionally promising. Continued investment in research and development for novel biomarkers and advanced diagnostic technologies will be a key growth accelerator. The increasing adoption of integrated diagnostics, which combine multiple testing modalities, will offer more comprehensive patient insights. The growing emphasis on early disease detection and risk stratification will drive demand for predictive and prognostic tests. Strategic partnerships and collaborations between diagnostic companies, pharmaceutical firms, and academic institutions are expected to foster innovation and accelerate product development. The expansion of telemedicine and remote diagnostic capabilities will also play a significant role in improving accessibility and patient convenience, further solidifying the market's upward trajectory.
North America Autoimmune Disease Diagnostics Market Segmentation
-
1. Disease Type
-
1.1. Systemic Autoimmune Disease
- 1.1.1. Rheumatoid Arthritis
- 1.1.2. Psoriasis
- 1.1.3. Systemic Lupus Erythematosus (SLE)
- 1.1.4. Multiple Sclerosis
- 1.1.5. Other Disease Types
-
1.2. Localized Autoimmune Disease
- 1.2.1. Inflammatory Bowel disease
- 1.2.2. Type 1 Diabetes
- 1.2.3. Thyroid
- 1.2.4. Other Localized Autoimmune Diseases
-
1.1. Systemic Autoimmune Disease
-
2. Diagnostic Test
- 2.1. Regular Laboratory Tests
- 2.2. Inflammatory Markers
- 2.3. Immunologic Assays
- 2.4. Antibody Tests
- 2.5. Other Tests
-
3. Geography
- 3.1. United States
- 3.2. Canada
- 3.3. Mexico
North America Autoimmune Disease Diagnostics Market Segmentation By Geography
- 1. United States
- 2. Canada
- 3. Mexico
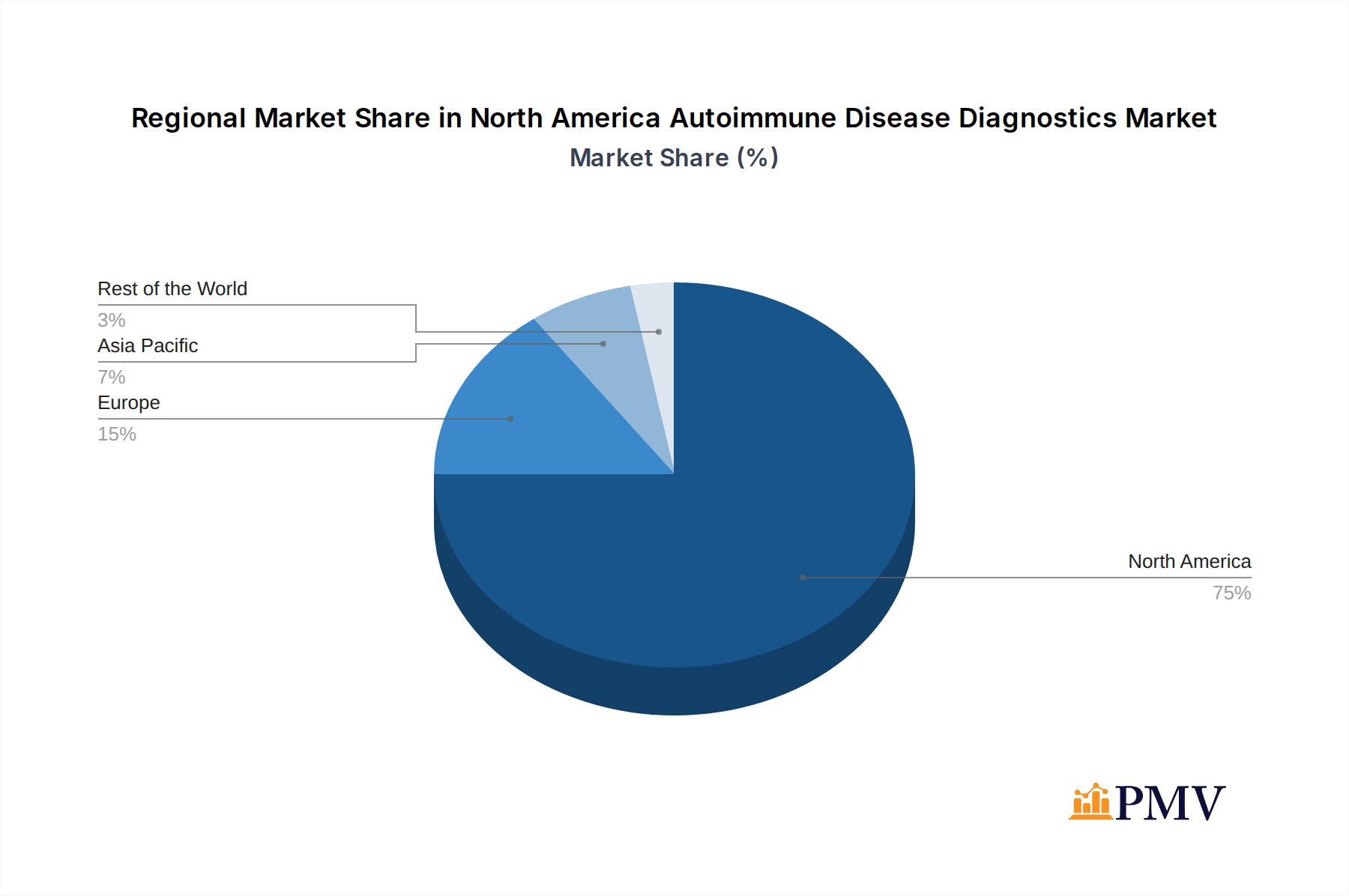
North America Autoimmune Disease Diagnostics Market Regional Market Share

Geographic Coverage of North America Autoimmune Disease Diagnostics Market
North America Autoimmune Disease Diagnostics Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.53% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.2.1. Increasing Prevalence and Growing Public Awareness of Autoimmune Diseases; Technological Advancements in Autoimmune Disease Diagnostics
- 3.3. Market Restrains
- 3.3.1. Slow Turnaround Time for Autoimmune Disease Diagnostic Test Results; High Frequency of False Positive Result
- 3.4. Market Trends
- 3.4.1. Immunologic Assays Segment is Expected to Hold a Significant Market Share Over the Forecast Period
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Disease Type
- 5.1.1. Systemic Autoimmune Disease
- 5.1.1.1. Rheumatoid Arthritis
- 5.1.1.2. Psoriasis
- 5.1.1.3. Systemic Lupus Erythematosus (SLE)
- 5.1.1.4. Multiple Sclerosis
- 5.1.1.5. Other Disease Types
- 5.1.2. Localized Autoimmune Disease
- 5.1.2.1. Inflammatory Bowel disease
- 5.1.2.2. Type 1 Diabetes
- 5.1.2.3. Thyroid
- 5.1.2.4. Other Localized Autoimmune Diseases
- 5.1.1. Systemic Autoimmune Disease
- 5.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 5.2.1. Regular Laboratory Tests
- 5.2.2. Inflammatory Markers
- 5.2.3. Immunologic Assays
- 5.2.4. Antibody Tests
- 5.2.5. Other Tests
- 5.3. Market Analysis, Insights and Forecast - by Geography
- 5.3.1. United States
- 5.3.2. Canada
- 5.3.3. Mexico
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. United States
- 5.4.2. Canada
- 5.4.3. Mexico
- 5.1. Market Analysis, Insights and Forecast - by Disease Type
- 6. United States North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Disease Type
- 6.1.1. Systemic Autoimmune Disease
- 6.1.1.1. Rheumatoid Arthritis
- 6.1.1.2. Psoriasis
- 6.1.1.3. Systemic Lupus Erythematosus (SLE)
- 6.1.1.4. Multiple Sclerosis
- 6.1.1.5. Other Disease Types
- 6.1.2. Localized Autoimmune Disease
- 6.1.2.1. Inflammatory Bowel disease
- 6.1.2.2. Type 1 Diabetes
- 6.1.2.3. Thyroid
- 6.1.2.4. Other Localized Autoimmune Diseases
- 6.1.1. Systemic Autoimmune Disease
- 6.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 6.2.1. Regular Laboratory Tests
- 6.2.2. Inflammatory Markers
- 6.2.3. Immunologic Assays
- 6.2.4. Antibody Tests
- 6.2.5. Other Tests
- 6.3. Market Analysis, Insights and Forecast - by Geography
- 6.3.1. United States
- 6.3.2. Canada
- 6.3.3. Mexico
- 6.1. Market Analysis, Insights and Forecast - by Disease Type
- 7. Canada North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Disease Type
- 7.1.1. Systemic Autoimmune Disease
- 7.1.1.1. Rheumatoid Arthritis
- 7.1.1.2. Psoriasis
- 7.1.1.3. Systemic Lupus Erythematosus (SLE)
- 7.1.1.4. Multiple Sclerosis
- 7.1.1.5. Other Disease Types
- 7.1.2. Localized Autoimmune Disease
- 7.1.2.1. Inflammatory Bowel disease
- 7.1.2.2. Type 1 Diabetes
- 7.1.2.3. Thyroid
- 7.1.2.4. Other Localized Autoimmune Diseases
- 7.1.1. Systemic Autoimmune Disease
- 7.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 7.2.1. Regular Laboratory Tests
- 7.2.2. Inflammatory Markers
- 7.2.3. Immunologic Assays
- 7.2.4. Antibody Tests
- 7.2.5. Other Tests
- 7.3. Market Analysis, Insights and Forecast - by Geography
- 7.3.1. United States
- 7.3.2. Canada
- 7.3.3. Mexico
- 7.1. Market Analysis, Insights and Forecast - by Disease Type
- 8. Mexico North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Disease Type
- 8.1.1. Systemic Autoimmune Disease
- 8.1.1.1. Rheumatoid Arthritis
- 8.1.1.2. Psoriasis
- 8.1.1.3. Systemic Lupus Erythematosus (SLE)
- 8.1.1.4. Multiple Sclerosis
- 8.1.1.5. Other Disease Types
- 8.1.2. Localized Autoimmune Disease
- 8.1.2.1. Inflammatory Bowel disease
- 8.1.2.2. Type 1 Diabetes
- 8.1.2.3. Thyroid
- 8.1.2.4. Other Localized Autoimmune Diseases
- 8.1.1. Systemic Autoimmune Disease
- 8.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 8.2.1. Regular Laboratory Tests
- 8.2.2. Inflammatory Markers
- 8.2.3. Immunologic Assays
- 8.2.4. Antibody Tests
- 8.2.5. Other Tests
- 8.3. Market Analysis, Insights and Forecast - by Geography
- 8.3.1. United States
- 8.3.2. Canada
- 8.3.3. Mexico
- 8.1. Market Analysis, Insights and Forecast - by Disease Type
- 9. Competitive Analysis
- 9.1. Market Share Analysis 2025
- 9.2. Company Profiles
- 9.2.1 Oncimmune
- 9.2.1.1. Overview
- 9.2.1.2. Products
- 9.2.1.3. SWOT Analysis
- 9.2.1.4. Recent Developments
- 9.2.1.5. Financials (Based on Availability)
- 9.2.2 HYCOR Biomedical
- 9.2.2.1. Overview
- 9.2.2.2. Products
- 9.2.2.3. SWOT Analysis
- 9.2.2.4. Recent Developments
- 9.2.2.5. Financials (Based on Availability)
- 9.2.3 Grifols SA
- 9.2.3.1. Overview
- 9.2.3.2. Products
- 9.2.3.3. SWOT Analysis
- 9.2.3.4. Recent Developments
- 9.2.3.5. Financials (Based on Availability)
- 9.2.4 Werfen Group
- 9.2.4.1. Overview
- 9.2.4.2. Products
- 9.2.4.3. SWOT Analysis
- 9.2.4.4. Recent Developments
- 9.2.4.5. Financials (Based on Availability)
- 9.2.5 Abbott Laboratories
- 9.2.5.1. Overview
- 9.2.5.2. Products
- 9.2.5.3. SWOT Analysis
- 9.2.5.4. Recent Developments
- 9.2.5.5. Financials (Based on Availability)
- 9.2.6 Siemens Healthineers Inc
- 9.2.6.1. Overview
- 9.2.6.2. Products
- 9.2.6.3. SWOT Analysis
- 9.2.6.4. Recent Developments
- 9.2.6.5. Financials (Based on Availability)
- 9.2.7 Bio-rad Laboratories
- 9.2.7.1. Overview
- 9.2.7.2. Products
- 9.2.7.3. SWOT Analysis
- 9.2.7.4. Recent Developments
- 9.2.7.5. Financials (Based on Availability)
- 9.2.8 Myriad Genetics
- 9.2.8.1. Overview
- 9.2.8.2. Products
- 9.2.8.3. SWOT Analysis
- 9.2.8.4. Recent Developments
- 9.2.8.5. Financials (Based on Availability)
- 9.2.9 Euroimmun AG (Perkinelmer Inc )
- 9.2.9.1. Overview
- 9.2.9.2. Products
- 9.2.9.3. SWOT Analysis
- 9.2.9.4. Recent Developments
- 9.2.9.5. Financials (Based on Availability)
- 9.2.10 F Hoffmann-la Roche
- 9.2.10.1. Overview
- 9.2.10.2. Products
- 9.2.10.3. SWOT Analysis
- 9.2.10.4. Recent Developments
- 9.2.10.5. Financials (Based on Availability)
- 9.2.11 Thermo Fisher Scientific
- 9.2.11.1. Overview
- 9.2.11.2. Products
- 9.2.11.3. SWOT Analysis
- 9.2.11.4. Recent Developments
- 9.2.11.5. Financials (Based on Availability)
- 9.2.12 Trinity Biotech
- 9.2.12.1. Overview
- 9.2.12.2. Products
- 9.2.12.3. SWOT Analysis
- 9.2.12.4. Recent Developments
- 9.2.12.5. Financials (Based on Availability)
- 9.2.1 Oncimmune
List of Figures
- Figure 1: North America Autoimmune Disease Diagnostics Market Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: North America Autoimmune Disease Diagnostics Market Share (%) by Company 2025
List of Tables
- Table 1: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 2: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 3: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 4: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 5: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 6: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 7: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Region 2020 & 2033
- Table 8: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 9: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 10: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 11: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 12: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 13: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 14: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 15: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Country 2020 & 2033
- Table 16: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 17: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 18: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 19: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 20: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 21: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 22: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 23: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Country 2020 & 2033
- Table 24: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 26: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 27: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 28: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 29: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 30: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 31: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Country 2020 & 2033
- Table 32: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the North America Autoimmune Disease Diagnostics Market?
The projected CAGR is approximately 5.53%.
2. Which companies are prominent players in the North America Autoimmune Disease Diagnostics Market?
Key companies in the market include Oncimmune, HYCOR Biomedical, Grifols SA, Werfen Group, Abbott Laboratories, Siemens Healthineers Inc, Bio-rad Laboratories, Myriad Genetics, Euroimmun AG (Perkinelmer Inc ), F Hoffmann-la Roche, Thermo Fisher Scientific, Trinity Biotech.
3. What are the main segments of the North America Autoimmune Disease Diagnostics Market?
The market segments include Disease Type, Diagnostic Test, Geography.
4. Can you provide details about the market size?
The market size is estimated to be USD 2.05 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence and Growing Public Awareness of Autoimmune Diseases; Technological Advancements in Autoimmune Disease Diagnostics.
6. What are the notable trends driving market growth?
Immunologic Assays Segment is Expected to Hold a Significant Market Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Slow Turnaround Time for Autoimmune Disease Diagnostic Test Results; High Frequency of False Positive Result.
8. Can you provide examples of recent developments in the market?
February 2023: Edesa Biotech received approval from Health Canada for a phase II clinical trial of its EB06 monoclonal antibody candidate to treat vitiligo, a life-altering autoimmune disease.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "North America Autoimmune Disease Diagnostics Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the North America Autoimmune Disease Diagnostics Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the North America Autoimmune Disease Diagnostics Market?
To stay informed about further developments, trends, and reports in the North America Autoimmune Disease Diagnostics Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
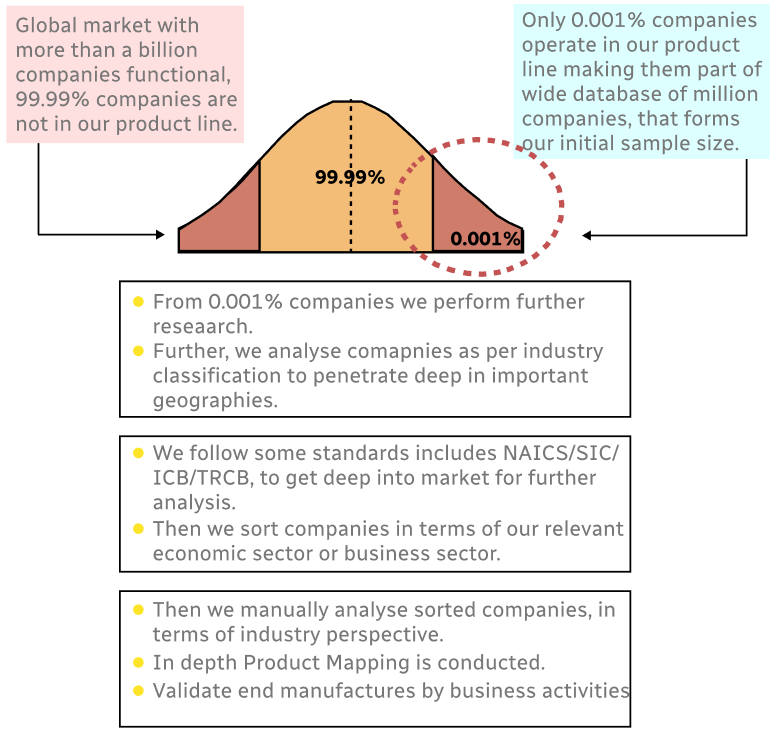
Step 1 - Identification of Relevant Samples Size from Population Database
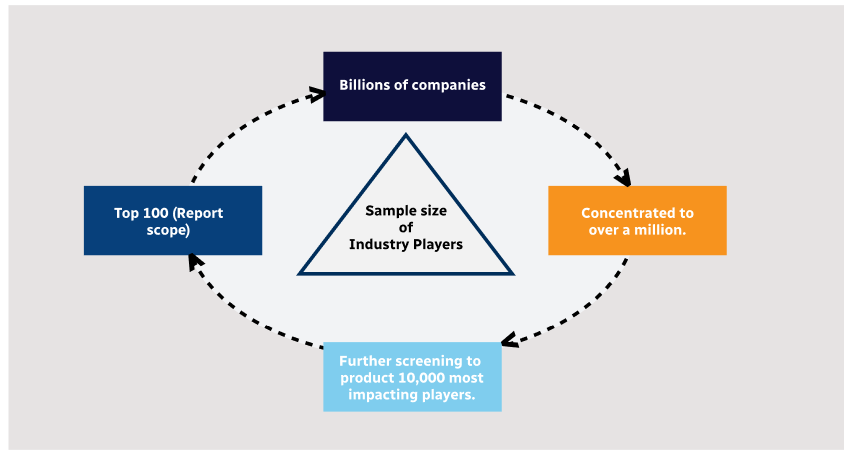
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
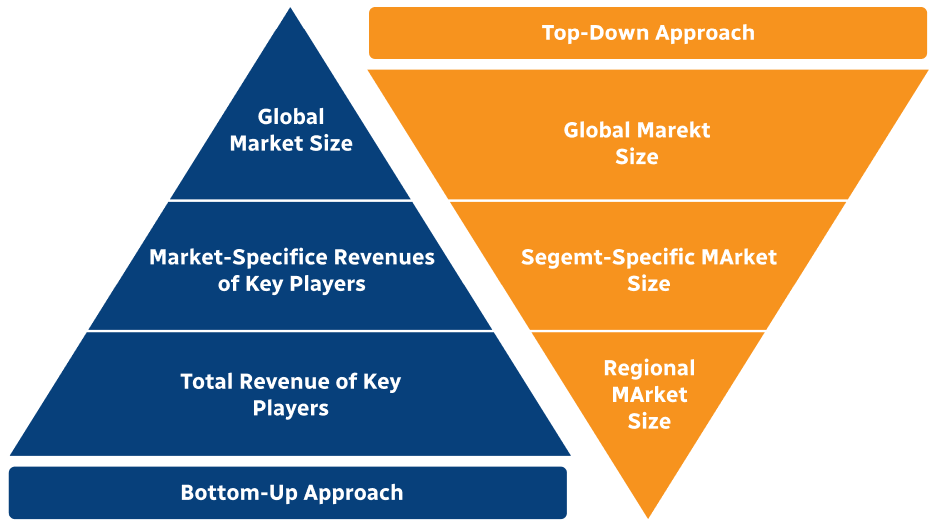
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
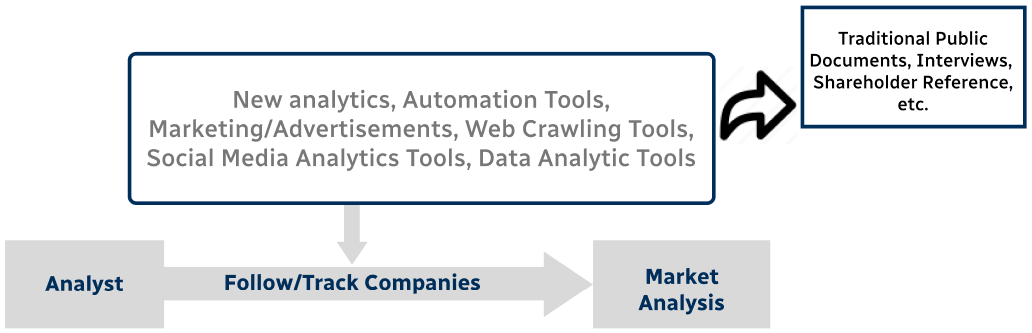
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


