Key Insights
The MiRNA Sequencing and Assay Industry is poised for robust growth, with an estimated market size of USD 382.71 million in 2025, driven by an impressive Compound Annual Growth Rate (CAGR) of 11.51% through 2033. This expansion is fueled by the increasing adoption of miRNA sequencing for diagnostic purposes in clinical settings and its growing application in life sciences research for understanding gene regulation and disease mechanisms. Advancements in sequencing technologies, particularly the development of more cost-effective and higher-throughput platforms, are making miRNA analysis more accessible. Furthermore, the rising prevalence of complex diseases such as cancer, cardiovascular disorders, and neurological conditions, where miRNAs play a crucial regulatory role, is a significant catalyst for market growth. The demand for personalized medicine and the increasing focus on biomarker discovery further bolster the utilization of miRNA sequencing and assay solutions.
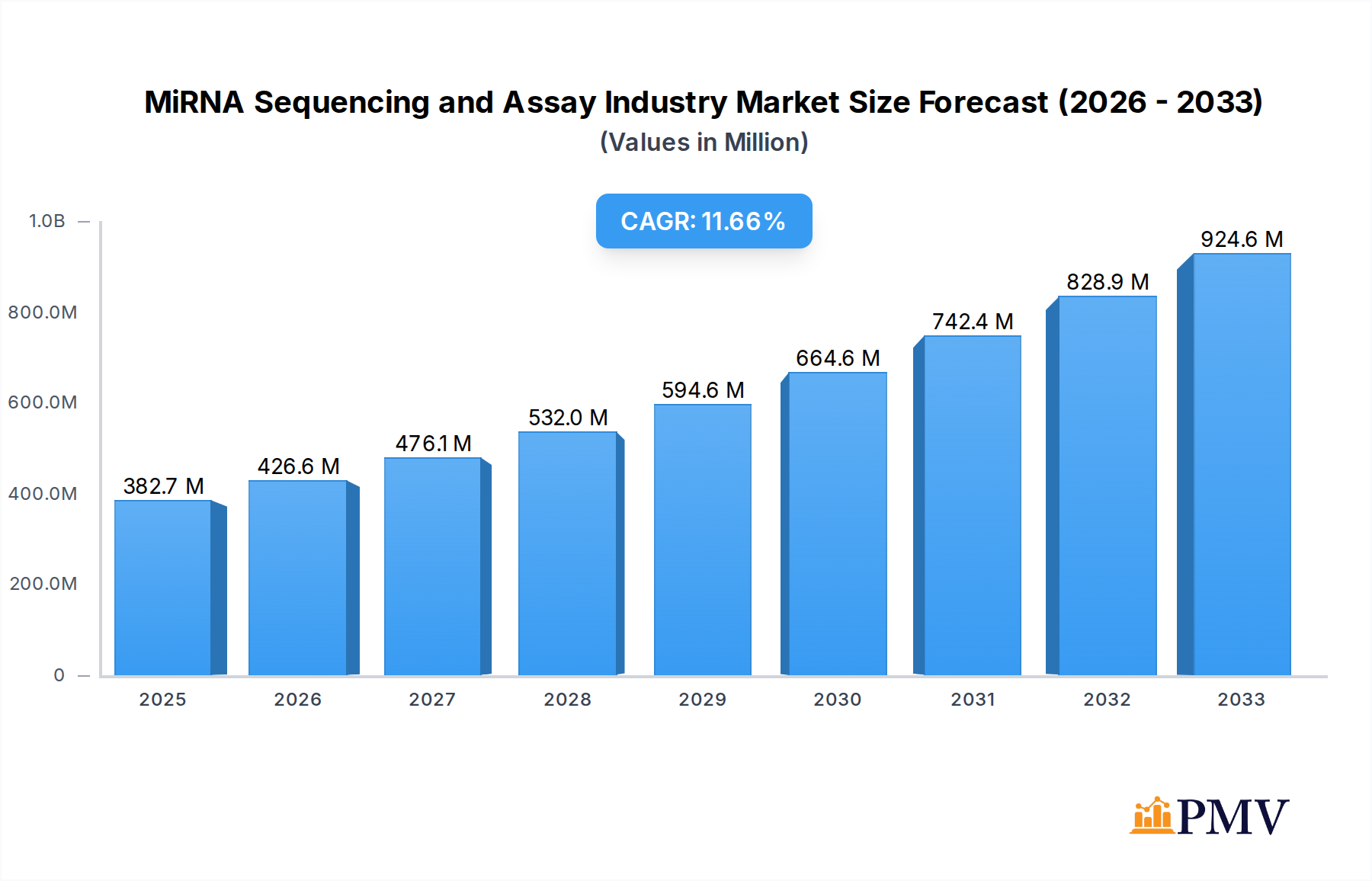
MiRNA Sequencing and Assay Industry Market Size (In Million)

The market is segmented across various product categories, including Sequencing Consumables and Library Preparation Kits, with significant contributions from both. Key technologies like Sequencing by Synthesis and Nanopore Sequencing are leading the technological advancements, offering improved accuracy and efficiency. The end-user landscape is dominated by Clinical Laboratories and Life Science Organizations, reflecting the dual utility of miRNA analysis in both diagnostics and research. Geographically, North America is expected to lead the market due to substantial investments in genomics research and healthcare infrastructure, followed closely by Europe and the rapidly expanding Asia Pacific region, which benefits from growing healthcare expenditure and increasing research activities. While market growth is strong, potential challenges such as the high initial cost of advanced sequencing equipment and the need for standardized protocols could influence the pace of adoption in certain segments.
MiRNA Sequencing and Assay Industry Company Market Share

This in-depth industry report provides an exhaustive analysis of the global MiRNA Sequencing and Assay market, projected to reach significant valuation by 2033. Leveraging a detailed study period from 2019 to 2033, with a base year of 2025 and a forecast period from 2025 to 2033, this report offers unparalleled insights into market dynamics, growth drivers, emerging trends, and competitive landscapes. We delve into the intricate ecosystem of miRNA profiling, encompassing crucial segments like Sequencing Consumables and Library Preparation Kits, and explore the impact of leading technologies such as Sequencing by Synthesis, Ion Semiconductor, SOLiD, and Nanopore Sequencing. The report meticulously examines end-user adoption across Clinical Laboratories, Life Science Organizations, and Other End Users, providing actionable intelligence for stakeholders.
MiRNA Sequencing and Assay Industry Market Structure & Competitive Dynamics
The MiRNA Sequencing and Assay market exhibits a dynamic structure characterized by moderate market concentration and a vibrant innovation ecosystem. Key players are strategically investing in research and development to enhance assay sensitivity, specificity, and throughput, driving innovation in areas like single-cell miRNA analysis and liquid biopsy applications. Regulatory frameworks, particularly those governing diagnostic assays and personalized medicine, play a crucial role in shaping market entry and product development. Product substitutes, while present in broader gene expression analysis, are increasingly being differentiated by the specific applications and analytical power offered by dedicated miRNA sequencing and assay solutions. End-user trends reveal a growing demand for more precise and less invasive diagnostic tools, fueling the adoption of miRNA-based biomarkers. Mergers and acquisition (M&A) activities are anticipated to intensify as larger players seek to consolidate market share and acquire novel technologies. For instance, M&A deal values are projected to reach several hundred million dollars annually, driven by the pursuit of intellectual property and expanded market reach. Market share distribution is expected to see shifts, with leaders focusing on expanding their product portfolios and geographic presence.
MiRNA Sequencing and Assay Industry Industry Trends & Insights
The MiRNA Sequencing and Assay industry is poised for robust growth, driven by an escalating demand for advanced diagnostics, personalized medicine, and a deeper understanding of biological processes at the microRNA level. The estimated Compound Annual Growth Rate (CAGR) for the forecast period is projected to be substantial, exceeding xx% annually. This growth is underpinned by significant technological disruptions. Innovations in next-generation sequencing (NGS) technologies have dramatically reduced costs and increased throughput, making miRNA sequencing more accessible for a wider range of research and clinical applications. The development of highly sensitive and specific miRNA assay platforms is also a major catalyst, enabling the detection of circulating miRNAs in biofluids for non-invasive diagnostics. Consumer preferences are increasingly shifting towards targeted therapies and predictive diagnostics, where miRNA profiling plays a pivotal role in disease identification, prognosis, and treatment response prediction across various therapeutic areas, including oncology, cardiology, and neurology. Competitive dynamics are intensifying, with companies focusing on developing integrated solutions that encompass sample preparation, sequencing, data analysis, and interpretation. Market penetration of advanced miRNA sequencing and assay technologies is steadily increasing, particularly in developed regions with strong healthcare infrastructure and a high propensity for adopting cutting-edge biomedical solutions. The growing awareness of the therapeutic and diagnostic potential of microRNAs is fueling investments in research and commercialization efforts, further accelerating market expansion.
Dominant Markets & Segments in MiRNA Sequencing and Assay Industry
The global MiRNA Sequencing and Assay market is characterized by several dominant markets and segments, each contributing significantly to overall growth.
- Leading Region: North America currently dominates the market, driven by substantial investments in life sciences research, a high prevalence of chronic diseases, and a robust healthcare infrastructure that readily adopts new diagnostic technologies. The United States, in particular, leads in research funding and clinical trial activity related to miRNA biomarkers.
- Leading Technology: Sequencing by Synthesis remains the dominant technology due to its high throughput, accuracy, and established infrastructure. However, Nanopore Sequencing is rapidly gaining traction due to its real-time data generation capabilities and potential for portable, point-of-care applications.
- Sequencing by Synthesis: Its widespread adoption in academic and clinical settings, coupled with continuous technological advancements by leading manufacturers, solidifies its market leadership.
- Nanopore Sequencing: Emerging applications in rapid pathogen identification and field-based diagnostics are expanding its market share.
- Dominant Product Segment: Library Preparation Kits are experiencing significant growth. The efficiency and accuracy of these kits are critical for the success of downstream sequencing and assay workflows, making them a vital component of the market.
- Library Preparation Kits: Development of kits optimized for low-input samples and diverse sample types (e.g., FFPE, plasma, urine) is a key driver for this segment.
- Dominant End User: Life Science Organizations represent the largest end-user segment, actively utilizing miRNA sequencing and assays for fundamental research, drug discovery, and biomarker identification.
- Life Science Organizations: Academic institutions and pharmaceutical companies are key consumers, driving innovation and demand for advanced genomic tools.
- Emerging End User: The Clinical Laboratory segment is experiencing rapid expansion as miRNA-based diagnostics move from research to clinical practice.
- Clinical Laboratory: Increased regulatory approvals for miRNA-based diagnostic tests will further propel growth in this segment, leading to widespread adoption in routine clinical diagnostics.
Economic policies supporting biotechnology research and development, along with advanced healthcare infrastructure, are crucial drivers for regional dominance. The ongoing expansion of genomic sequencing capabilities within clinical settings is a significant factor driving the growth of the clinical laboratory segment.
MiRNA Sequencing and Assay Industry Product Innovations
Product innovations in the MiRNA Sequencing and Assay industry are primarily focused on enhancing assay sensitivity, reducing turnaround times, and improving data analysis capabilities. Companies are developing novel library preparation kits that accommodate smaller sample volumes and diverse biological matrices, enabling the analysis of challenging samples like liquid biopsies. Advancements in sequencing platforms, such as increased read lengths and reduced error rates with Nanopore sequencing, are opening new avenues for miRNA research and diagnostics. Furthermore, the integration of bioinformatics tools and artificial intelligence for miRNA data interpretation is enhancing the clinical utility of these technologies. These innovations provide competitive advantages by enabling more accurate disease detection, improved patient stratification, and the identification of novel therapeutic targets.
Report Segmentation & Scope
This comprehensive report segments the MiRNA Sequencing and Assay market across several key dimensions. The Product segmentation includes Sequencing Consumables and Library Preparation Kits. The Technology segmentation encompasses Sequencing by Synthesis, Ion Semiconductor, SOLiD, and Nanopore Sequencing. The End User segmentation covers Clinical Laboratory, Life Science Organization, and Other End Users.
- Sequencing Consumables: This segment includes reagents, enzymes, and other consumables essential for the sequencing workflow. Growth projections for this segment are substantial, driven by the increasing volume of sequencing performed globally.
- Library Preparation Kits: This segment is critical for preparing samples for sequencing. We anticipate significant growth due to the development of specialized kits for various applications and sample types.
- Sequencing by Synthesis: This mature technology segment is expected to maintain its market leadership, with steady growth driven by ongoing platform improvements and widespread adoption.
- Ion Semiconductor: This segment offers a cost-effective alternative for certain applications and is projected to witness moderate growth.
- SOLiD: While a legacy technology, it continues to hold a niche in specific research applications, with a stable market presence.
- Nanopore Sequencing: This segment is poised for rapid growth due to its emerging applications and technological advancements.
- Clinical Laboratory: This segment is projected to experience the highest growth rate as miRNA-based diagnostics become increasingly integrated into clinical practice.
- Life Science Organization: This segment, encompassing academic research and pharmaceutical R&D, will continue to be a major consumer of miRNA sequencing and assay technologies, exhibiting steady growth.
- Other End Users: This segment includes diverse applications such as agricultural research and environmental monitoring, contributing to the overall market expansion.
Key Drivers of MiRNA Sequencing and Assay Industry Growth
The growth of the MiRNA Sequencing and Assay industry is propelled by several key factors. Technologically, advancements in next-generation sequencing (NGS) and the development of highly sensitive assay platforms are making miRNA analysis more accessible and informative. Economically, increased funding for biomedical research and the growing adoption of precision medicine are creating substantial market opportunities. Regulatory bodies are increasingly recognizing the potential of miRNA biomarkers for diagnostic and therapeutic applications, facilitating market entry for innovative products. The expanding research into the role of miRNAs in various diseases, from cancer to neurological disorders, is a primary driver, fueling the demand for comprehensive sequencing and assay solutions. The development of non-invasive diagnostic methods, such as liquid biopsies utilizing circulating miRNAs, is also a significant growth accelerator.
Challenges in the MiRNA Sequencing and Assay Industry Sector
Despite its promising outlook, the MiRNA Sequencing and Assay industry faces several challenges. Regulatory hurdles for the approval of novel miRNA-based diagnostic tests can be lengthy and complex, potentially delaying market entry. Supply chain issues, particularly for specialized reagents and consumables, can impact manufacturing and availability. Intense competitive pressures among established players and emerging startups necessitate continuous innovation and cost optimization. Furthermore, standardization of miRNA profiling protocols and data analysis pipelines remains an ongoing challenge, impacting comparability across studies and laboratories. The high initial cost of advanced sequencing equipment can also be a barrier for smaller research institutions and clinical laboratories, although this is steadily decreasing.
Leading Players in the MiRNA Sequencing and Assay Industry Market
The MiRNA Sequencing and Assay industry is home to several innovative companies driving its advancement. Among the leading players are:
- Norgen Biotek Corporation
- Trilink Biotechnologies
- Takara Bio Inc
- Qiagen N V
- Lexogen
- Oxford Nanopore Technologies
- New England Biolabs Inc
- PerkinElmer Inc
- Illumina Inc
- Thermo Fisher Scientific
Key Developments in MiRNA Sequencing and Assay Industry Sector
- April 2022: HTC Molecular Diagnostics Inc. unveiled new features of its proprietary HTG EdgeSeq technology. The company has improved its technology, and now its new sample preparation harmonization enables miRNA and mRNA profiling without the need for additional samples.
- January 2022: Allogene Therapeutics announced a collaboration with Antion Biosciences to advance multiplex gene silencing for the development of next-generation allogeneic CAR T products. As part of the collaboration, Antion's miRNA technology (miCAR) will be used as an additional tool to enhance the efficacy and safety of allogeneic CAR T therapies.
Strategic MiRNA Sequencing and Assay Industry Market Outlook
The strategic outlook for the MiRNA Sequencing and Assay industry is highly positive, driven by ongoing technological advancements and increasing clinical adoption. Future growth will be accelerated by the expanding applications of miRNA biomarkers in oncology, infectious diseases, and cardiovascular conditions. The development of more integrated and user-friendly platforms, coupled with robust bioinformatics support, will further democratize access to miRNA profiling. Strategic opportunities lie in the expansion of liquid biopsy applications, the development of companion diagnostics, and the exploration of miRNA-based therapeutic interventions. Collaboration between technology providers, research institutions, and pharmaceutical companies will be crucial for translating research discoveries into impactful clinical solutions, solidifying the industry's role in the future of personalized healthcare.
MiRNA Sequencing and Assay Industry Segmentation
-
1. Product
- 1.1. Sequencing Consumables
- 1.2. Library Preparation Kits
-
2. Technology
- 2.1. Sequencing by Synthesis
- 2.2. Ion Semiconductor
- 2.3. SOLiD
- 2.4. Nanopore Sequencing
-
3. End User
- 3.1. Clinical Laboratory
- 3.2. Life Science Organization
- 3.3. Other End Users
MiRNA Sequencing and Assay Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
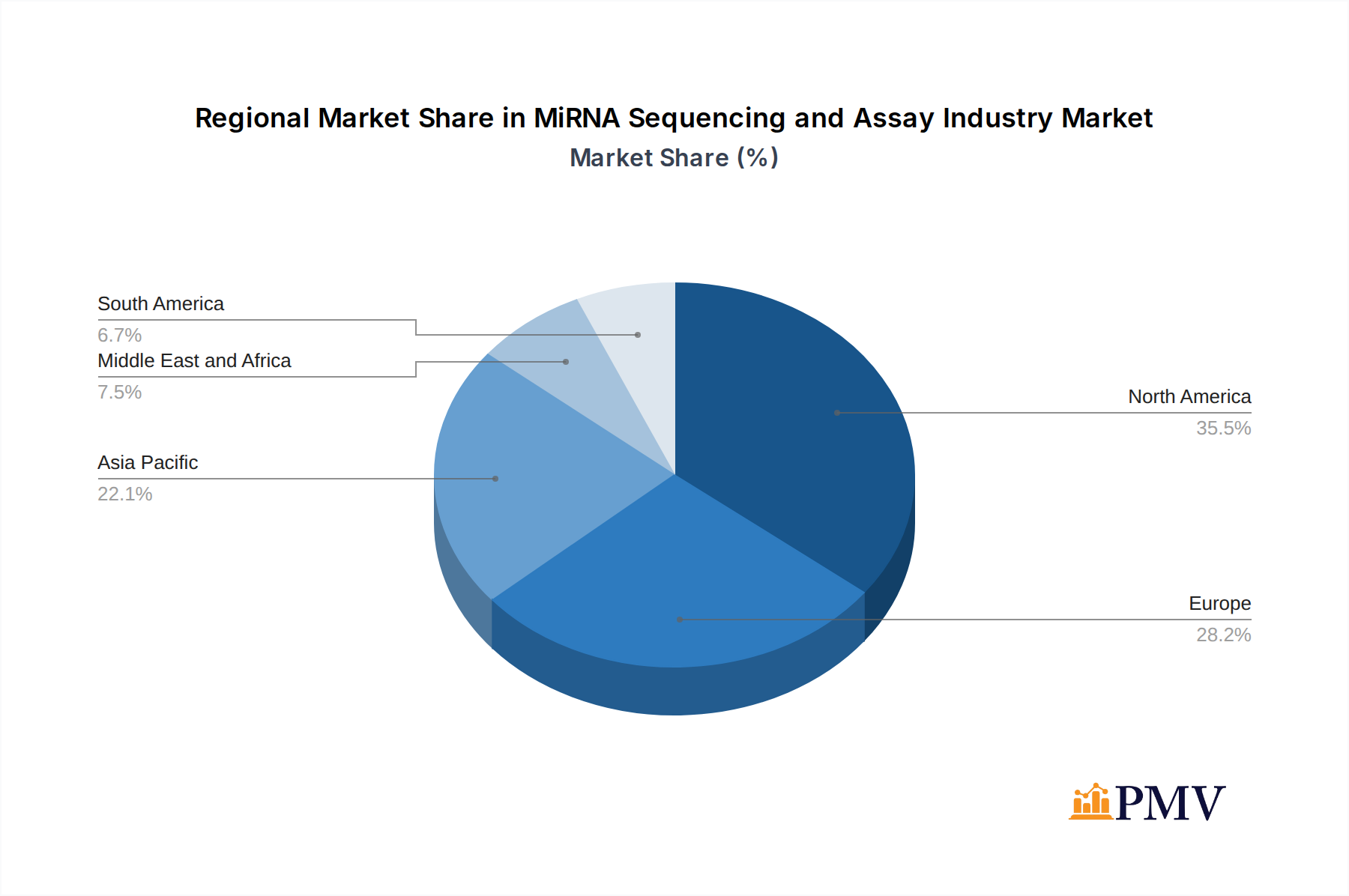
MiRNA Sequencing and Assay Industry Regional Market Share

Geographic Coverage of MiRNA Sequencing and Assay Industry
MiRNA Sequencing and Assay Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.51% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Sequencing Consumables
- 5.1.2. Library Preparation Kits
- 5.2. Market Analysis, Insights and Forecast - by Technology
- 5.2.1. Sequencing by Synthesis
- 5.2.2. Ion Semiconductor
- 5.2.3. SOLiD
- 5.2.4. Nanopore Sequencing
- 5.3. Market Analysis, Insights and Forecast - by End User
- 5.3.1. Clinical Laboratory
- 5.3.2. Life Science Organization
- 5.3.3. Other End Users
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. Global MiRNA Sequencing and Assay Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Sequencing Consumables
- 6.1.2. Library Preparation Kits
- 6.2. Market Analysis, Insights and Forecast - by Technology
- 6.2.1. Sequencing by Synthesis
- 6.2.2. Ion Semiconductor
- 6.2.3. SOLiD
- 6.2.4. Nanopore Sequencing
- 6.3. Market Analysis, Insights and Forecast - by End User
- 6.3.1. Clinical Laboratory
- 6.3.2. Life Science Organization
- 6.3.3. Other End Users
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. North America MiRNA Sequencing and Assay Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Sequencing Consumables
- 7.1.2. Library Preparation Kits
- 7.2. Market Analysis, Insights and Forecast - by Technology
- 7.2.1. Sequencing by Synthesis
- 7.2.2. Ion Semiconductor
- 7.2.3. SOLiD
- 7.2.4. Nanopore Sequencing
- 7.3. Market Analysis, Insights and Forecast - by End User
- 7.3.1. Clinical Laboratory
- 7.3.2. Life Science Organization
- 7.3.3. Other End Users
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Europe MiRNA Sequencing and Assay Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Sequencing Consumables
- 8.1.2. Library Preparation Kits
- 8.2. Market Analysis, Insights and Forecast - by Technology
- 8.2.1. Sequencing by Synthesis
- 8.2.2. Ion Semiconductor
- 8.2.3. SOLiD
- 8.2.4. Nanopore Sequencing
- 8.3. Market Analysis, Insights and Forecast - by End User
- 8.3.1. Clinical Laboratory
- 8.3.2. Life Science Organization
- 8.3.3. Other End Users
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Asia Pacific MiRNA Sequencing and Assay Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Sequencing Consumables
- 9.1.2. Library Preparation Kits
- 9.2. Market Analysis, Insights and Forecast - by Technology
- 9.2.1. Sequencing by Synthesis
- 9.2.2. Ion Semiconductor
- 9.2.3. SOLiD
- 9.2.4. Nanopore Sequencing
- 9.3. Market Analysis, Insights and Forecast - by End User
- 9.3.1. Clinical Laboratory
- 9.3.2. Life Science Organization
- 9.3.3. Other End Users
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Middle East and Africa MiRNA Sequencing and Assay Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product
- 10.1.1. Sequencing Consumables
- 10.1.2. Library Preparation Kits
- 10.2. Market Analysis, Insights and Forecast - by Technology
- 10.2.1. Sequencing by Synthesis
- 10.2.2. Ion Semiconductor
- 10.2.3. SOLiD
- 10.2.4. Nanopore Sequencing
- 10.3. Market Analysis, Insights and Forecast - by End User
- 10.3.1. Clinical Laboratory
- 10.3.2. Life Science Organization
- 10.3.3. Other End Users
- 10.1. Market Analysis, Insights and Forecast - by Product
- 11. South America MiRNA Sequencing and Assay Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product
- 11.1.1. Sequencing Consumables
- 11.1.2. Library Preparation Kits
- 11.2. Market Analysis, Insights and Forecast - by Technology
- 11.2.1. Sequencing by Synthesis
- 11.2.2. Ion Semiconductor
- 11.2.3. SOLiD
- 11.2.4. Nanopore Sequencing
- 11.3. Market Analysis, Insights and Forecast - by End User
- 11.3.1. Clinical Laboratory
- 11.3.2. Life Science Organization
- 11.3.3. Other End Users
- 11.1. Market Analysis, Insights and Forecast - by Product
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Norgen Biotek Corporation
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Trilink Biotechnologies
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Takara Bio Inc
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Qiagen N V
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Lexogen
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Oxford Nanopore Technologies*List Not Exhaustive
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 New England Biolabs Inc
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 PerkinElmer Inc
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Illumina Inc
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Thermo Fisher Scientific
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.1 Norgen Biotek Corporation
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global MiRNA Sequencing and Assay Industry Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America MiRNA Sequencing and Assay Industry Revenue (million), by Product 2025 & 2033
- Figure 3: North America MiRNA Sequencing and Assay Industry Revenue Share (%), by Product 2025 & 2033
- Figure 4: North America MiRNA Sequencing and Assay Industry Revenue (million), by Technology 2025 & 2033
- Figure 5: North America MiRNA Sequencing and Assay Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 6: North America MiRNA Sequencing and Assay Industry Revenue (million), by End User 2025 & 2033
- Figure 7: North America MiRNA Sequencing and Assay Industry Revenue Share (%), by End User 2025 & 2033
- Figure 8: North America MiRNA Sequencing and Assay Industry Revenue (million), by Country 2025 & 2033
- Figure 9: North America MiRNA Sequencing and Assay Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: Europe MiRNA Sequencing and Assay Industry Revenue (million), by Product 2025 & 2033
- Figure 11: Europe MiRNA Sequencing and Assay Industry Revenue Share (%), by Product 2025 & 2033
- Figure 12: Europe MiRNA Sequencing and Assay Industry Revenue (million), by Technology 2025 & 2033
- Figure 13: Europe MiRNA Sequencing and Assay Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 14: Europe MiRNA Sequencing and Assay Industry Revenue (million), by End User 2025 & 2033
- Figure 15: Europe MiRNA Sequencing and Assay Industry Revenue Share (%), by End User 2025 & 2033
- Figure 16: Europe MiRNA Sequencing and Assay Industry Revenue (million), by Country 2025 & 2033
- Figure 17: Europe MiRNA Sequencing and Assay Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: Asia Pacific MiRNA Sequencing and Assay Industry Revenue (million), by Product 2025 & 2033
- Figure 19: Asia Pacific MiRNA Sequencing and Assay Industry Revenue Share (%), by Product 2025 & 2033
- Figure 20: Asia Pacific MiRNA Sequencing and Assay Industry Revenue (million), by Technology 2025 & 2033
- Figure 21: Asia Pacific MiRNA Sequencing and Assay Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 22: Asia Pacific MiRNA Sequencing and Assay Industry Revenue (million), by End User 2025 & 2033
- Figure 23: Asia Pacific MiRNA Sequencing and Assay Industry Revenue Share (%), by End User 2025 & 2033
- Figure 24: Asia Pacific MiRNA Sequencing and Assay Industry Revenue (million), by Country 2025 & 2033
- Figure 25: Asia Pacific MiRNA Sequencing and Assay Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue (million), by Product 2025 & 2033
- Figure 27: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue Share (%), by Product 2025 & 2033
- Figure 28: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue (million), by Technology 2025 & 2033
- Figure 29: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 30: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue (million), by End User 2025 & 2033
- Figure 31: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue Share (%), by End User 2025 & 2033
- Figure 32: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue (million), by Country 2025 & 2033
- Figure 33: Middle East and Africa MiRNA Sequencing and Assay Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: South America MiRNA Sequencing and Assay Industry Revenue (million), by Product 2025 & 2033
- Figure 35: South America MiRNA Sequencing and Assay Industry Revenue Share (%), by Product 2025 & 2033
- Figure 36: South America MiRNA Sequencing and Assay Industry Revenue (million), by Technology 2025 & 2033
- Figure 37: South America MiRNA Sequencing and Assay Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 38: South America MiRNA Sequencing and Assay Industry Revenue (million), by End User 2025 & 2033
- Figure 39: South America MiRNA Sequencing and Assay Industry Revenue Share (%), by End User 2025 & 2033
- Figure 40: South America MiRNA Sequencing and Assay Industry Revenue (million), by Country 2025 & 2033
- Figure 41: South America MiRNA Sequencing and Assay Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Product 2020 & 2033
- Table 2: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 3: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by End User 2020 & 2033
- Table 4: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Region 2020 & 2033
- Table 5: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Product 2020 & 2033
- Table 6: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 7: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by End User 2020 & 2033
- Table 8: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Country 2020 & 2033
- Table 9: United States MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Canada MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 11: Mexico MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 12: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Product 2020 & 2033
- Table 13: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 14: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by End User 2020 & 2033
- Table 15: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Country 2020 & 2033
- Table 16: Germany MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 17: United Kingdom MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 18: France MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 19: Italy MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Spain MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: Rest of Europe MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Product 2020 & 2033
- Table 23: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 24: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by End User 2020 & 2033
- Table 25: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Country 2020 & 2033
- Table 26: China MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Japan MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: India MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 29: Australia MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 30: South Korea MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 31: Rest of Asia Pacific MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Product 2020 & 2033
- Table 33: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 34: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by End User 2020 & 2033
- Table 35: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Country 2020 & 2033
- Table 36: GCC MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: South Africa MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 38: Rest of Middle East and Africa MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 39: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Product 2020 & 2033
- Table 40: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Technology 2020 & 2033
- Table 41: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by End User 2020 & 2033
- Table 42: Global MiRNA Sequencing and Assay Industry Revenue million Forecast, by Country 2020 & 2033
- Table 43: Brazil MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: Argentina MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Rest of South America MiRNA Sequencing and Assay Industry Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the MiRNA Sequencing and Assay Industry?
The projected CAGR is approximately 11.51%.
2. Which companies are prominent players in the MiRNA Sequencing and Assay Industry?
Key companies in the market include Norgen Biotek Corporation, Trilink Biotechnologies, Takara Bio Inc, Qiagen N V, Lexogen, Oxford Nanopore Technologies*List Not Exhaustive, New England Biolabs Inc, PerkinElmer Inc, Illumina Inc, Thermo Fisher Scientific.
3. What are the main segments of the MiRNA Sequencing and Assay Industry?
The market segments include Product, Technology, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD 382.71 million as of 2022.
5. What are some drivers contributing to market growth?
MiRNA Sequencing and Assay Sequencing Supremacy Over Other Methods; Increased Research in the Field of Genomics; Reduced Sequencing Costs.
6. What are the notable trends driving market growth?
Library Preparation Kits are Expected to Hold the Largest Market Share in the miRNA Sequencing and Assay Market.
7. Are there any restraints impacting market growth?
Paucity of Expert and Need for Computing Infrastructure.
8. Can you provide examples of recent developments in the market?
April 2022: HTC Molecular Diagnostics Inc. unveiled new features of its proprietary HTG EdgeSeq technology. The company has improved its technology, and now its new sample preparation harmonization enables miRNA and mRNA profiling without the need for additional samples.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "MiRNA Sequencing and Assay Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the MiRNA Sequencing and Assay Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the MiRNA Sequencing and Assay Industry?
To stay informed about further developments, trends, and reports in the MiRNA Sequencing and Assay Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
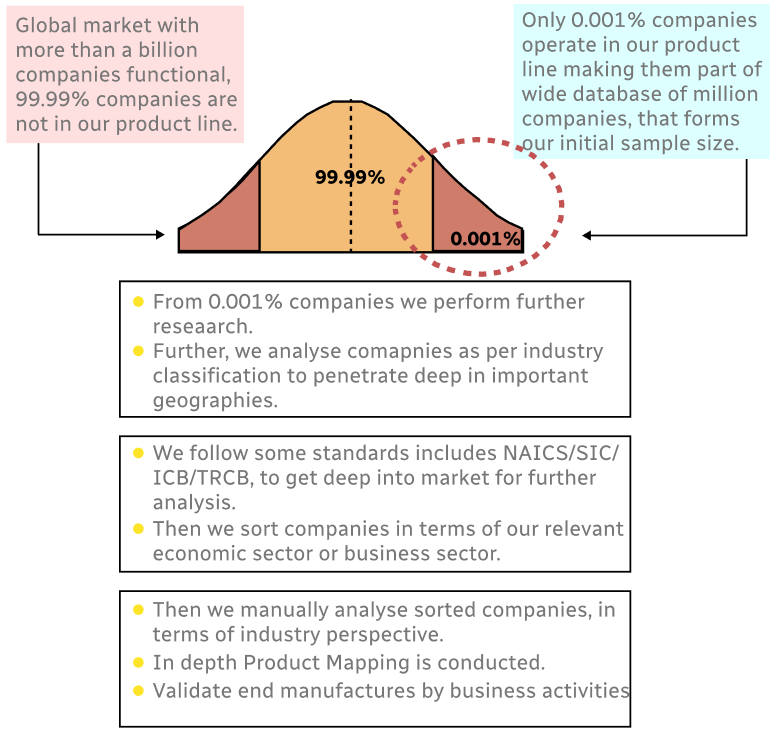
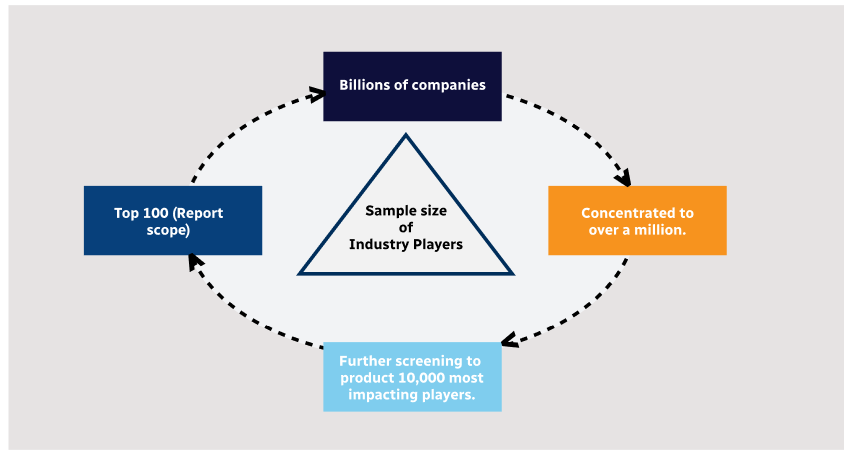
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
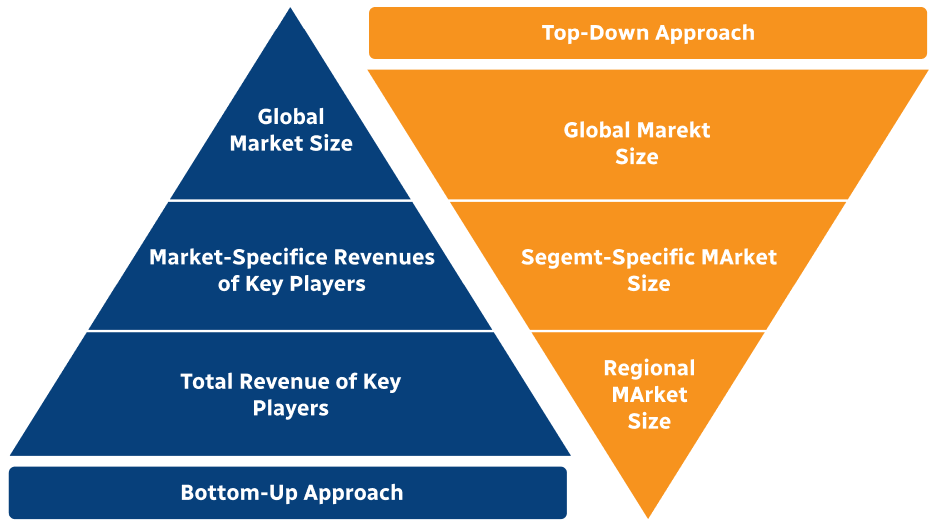
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
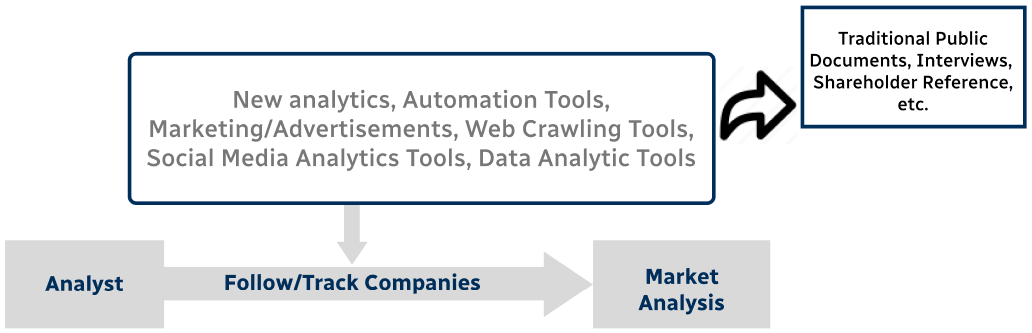
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


