Key Insights
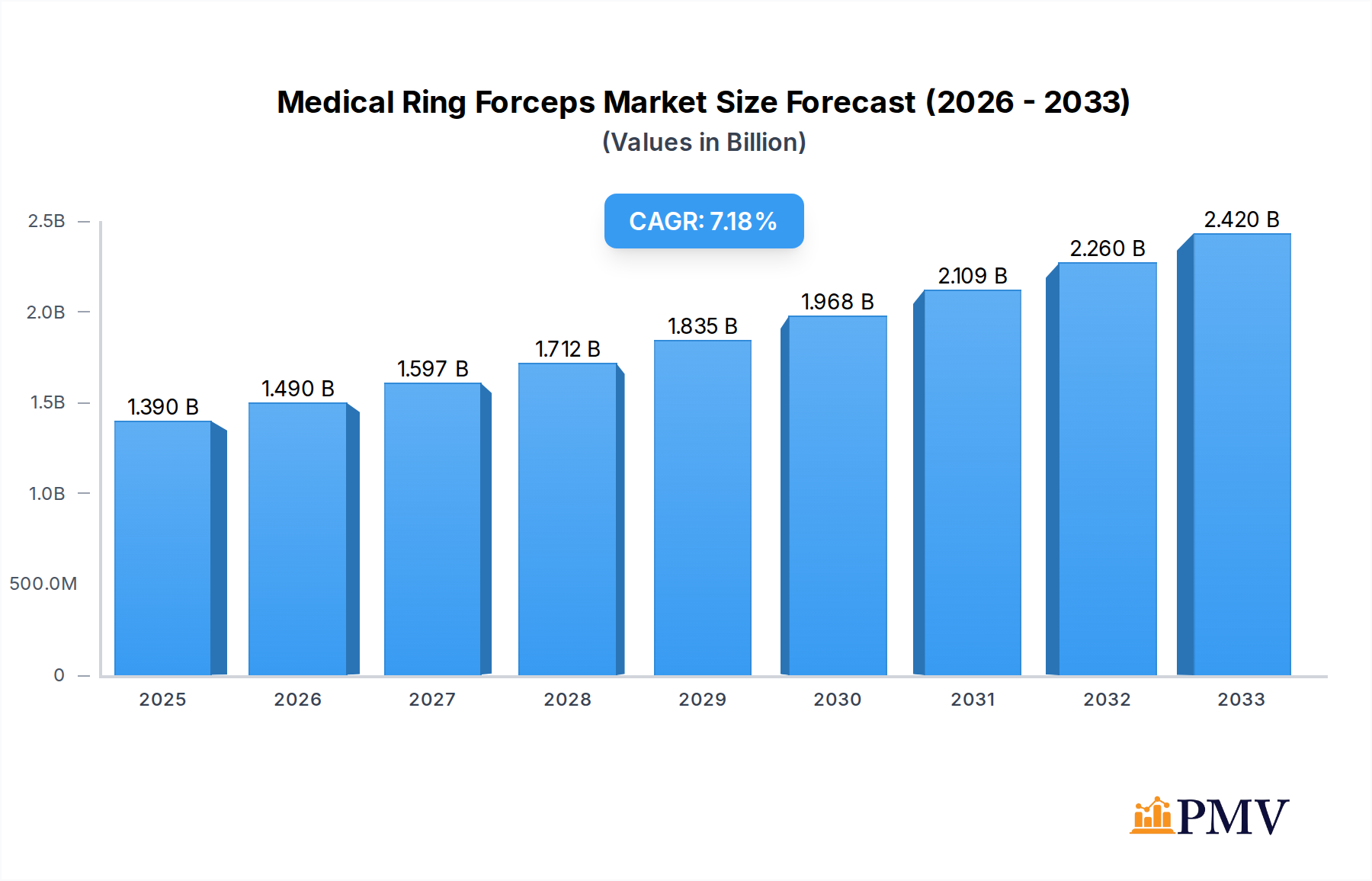
The global Medical Ring Forceps market is poised for robust expansion, projected to reach USD 1.39 billion in 2025, with a compelling Compound Annual Growth Rate (CAGR) of 7.3% anticipated throughout the forecast period from 2025 to 2033. This significant growth is underpinned by several key drivers, including the increasing prevalence of surgical procedures across various medical specialties and a rising demand for minimally invasive interventions, which often rely on specialized forceps. Advancements in material science, particularly the development of lighter and more durable titanium alloys, are also contributing to market dynamism by offering enhanced performance and patient safety. Furthermore, the growing global healthcare infrastructure, especially in emerging economies, coupled with increased healthcare expenditure, is creating fertile ground for market penetration and sustained growth. The market is witnessing a discernible trend towards the adoption of advanced, ergonomic designs that improve surgical precision and reduce user fatigue, aligning with the broader push for innovation in surgical instrumentation.
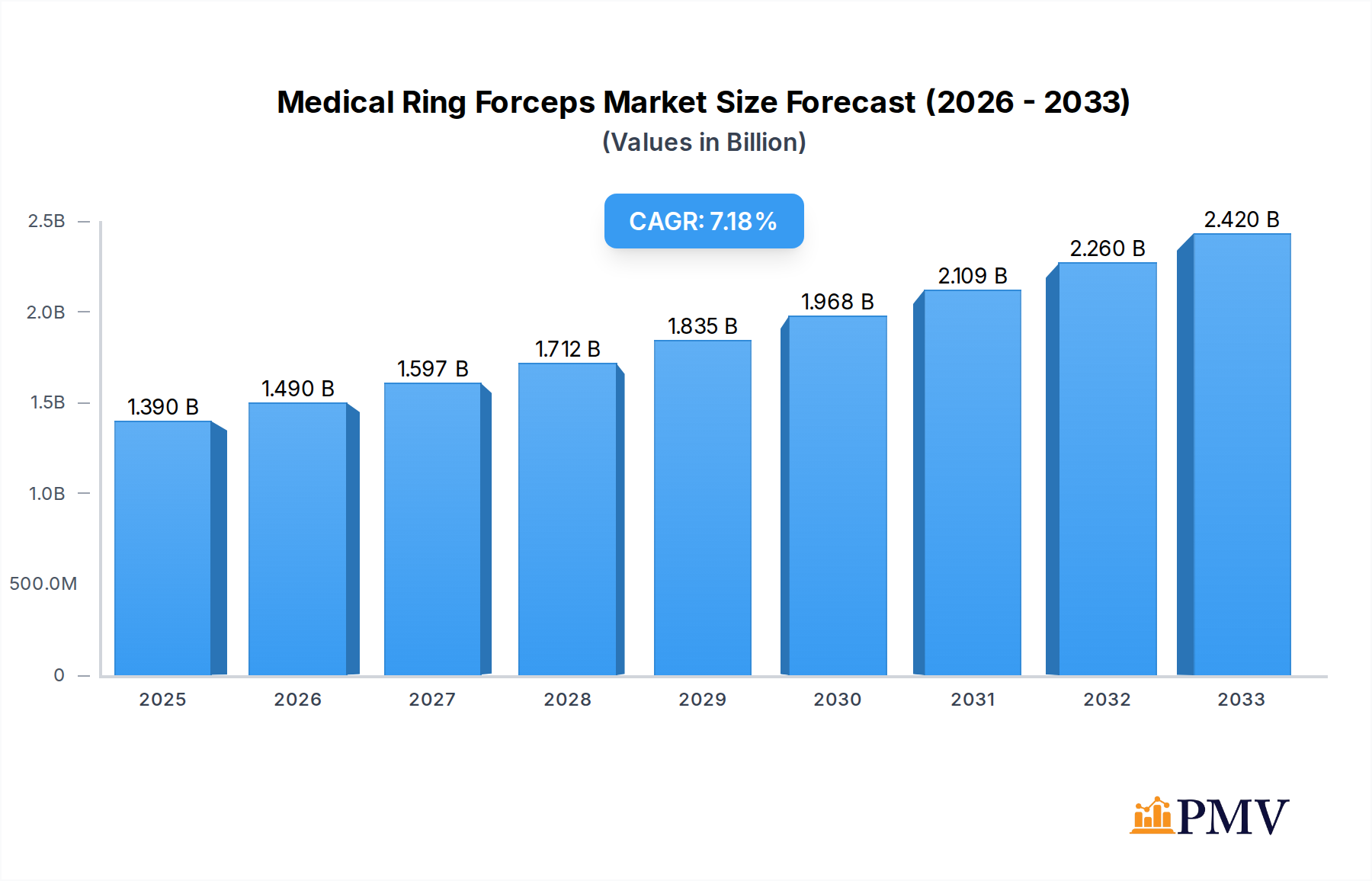
Medical Ring Forceps Market Size (In Billion)

The market's growth trajectory is further supported by the diversification of applications within hospitals and specialty clinics, catering to a wide range of surgical needs. While the market exhibits strong upward momentum, potential restraints such as stringent regulatory approval processes for new medical devices and the high cost associated with technologically advanced forceps could present challenges. However, the inherent necessity of these instruments in countless surgical interventions, from general surgery to specialized fields like gynecology and urology, ensures sustained demand. Leading companies are actively engaged in research and development to introduce innovative products, enhance manufacturing capabilities, and expand their market reach through strategic partnerships and acquisitions. The competitive landscape is characterized by a mix of established players and emerging innovators, all vying for a share of this expanding and critical medical device segment, with a focus on delivering superior quality and cost-effective solutions to healthcare providers worldwide.
Medical Ring Forceps Company Market Share

This comprehensive report delivers a detailed market analysis of Medical Ring Forceps, offering unparalleled insights into market dynamics, competitive landscapes, and future growth trajectories. Covering the period from 2019 to 2033, with a base year of 2025 and an estimated year also of 2025, this study is an indispensable resource for stakeholders seeking to navigate the complexities of the global Medical Ring Forceps market. We meticulously examine medical forceps market share, surgical instrument market trends, healthcare device innovations, and the demand for titanium ring forceps and stainless steel ring forceps. With a focus on hospital application, specialty clinic usage, and the burgeoning others segment, this report provides actionable intelligence for market expansion and strategic planning. The projected market size is expected to reach approximately one billion billion by 2033, with a projected CAGR of xx% during the forecast period (2025–2033).
Medical Ring Forceps Market Structure & Competitive Dynamics
The Medical Ring Forceps market exhibits a moderately concentrated structure, characterized by the presence of both established global players and emerging regional manufacturers. Innovation ecosystems are robust, driven by continuous research and development in materials science and surgical technology. Regulatory frameworks, primarily governed by bodies like the FDA and EMA, play a crucial role in ensuring product safety and efficacy, influencing market entry and product approvals. Product substitutes, while limited in direct application, can include alternative surgical tools or techniques. End-user trends are strongly influenced by the increasing demand for minimally invasive surgical procedures, the aging global population, and the rising prevalence of chronic diseases, all of which necessitate precision surgical instruments. Mergers and acquisition (M&A) activities are a significant aspect of market dynamics, with observed M&A deal values in the range of hundreds of millions billion over the historical period.
- Market Concentration: Key players hold substantial market share, estimated at xx% combined for the top five companies.
- Innovation Ecosystems: Driven by collaborations between instrument manufacturers, research institutions, and healthcare providers.
- Regulatory Frameworks: Strict adherence to quality standards such as ISO 13485 is paramount.
- Product Substitutes: Primarily alternative surgical techniques rather than direct instrument replacements.
- End-User Trends: Growing demand for specialized surgical instruments in procedures like laparoscopy and endoscopy.
- M&A Activities: Strategic acquisitions to expand product portfolios and market reach, with recent deals valued in the range of tens of millions billion.
Medical Ring Forceps Industry Trends & Insights
The global Medical Ring Forceps market is experiencing a consistent upward trajectory, propelled by a confluence of powerful market growth drivers. The increasing global burden of diseases requiring surgical intervention, coupled with advancements in surgical methodologies, directly fuels the demand for high-quality and specialized surgical instruments. Technological disruptions are profoundly shaping the industry, with a notable shift towards the development and adoption of advanced materials like titanium ring forceps, renowned for their biocompatibility, strength, and reduced weight compared to traditional stainless steel ring forceps. Furthermore, the integration of enhanced ergonomic designs and novel surface treatments in medical forceps is improving surgeon comfort and procedural precision, leading to better patient outcomes. The growing emphasis on patient safety and the reduction of healthcare-associated infections is also driving the demand for disposable or advanced sterilization-compatible instruments. Consumer preferences are evolving, with healthcare providers prioritizing instruments that offer superior performance, durability, and cost-effectiveness in the long run. The competitive landscape is characterized by intense innovation, with companies investing heavily in R&D to differentiate their product offerings. Market penetration of advanced ring forceps is projected to reach xx% by 2033, indicating a significant adoption rate across various healthcare settings. The Compound Annual Growth Rate (CAGR) for the Medical Ring Forceps market is anticipated to be a healthy xx% over the forecast period, driven by these multifaceted factors.
Dominant Markets & Segments in Medical Ring Forceps
The Hospital segment stands out as the dominant market for Medical Ring Forceps, accounting for an estimated xx% of the total market revenue in the base year of 2025. This dominance is underpinned by the sheer volume of surgical procedures conducted within hospital settings, ranging from general surgery to complex orthopedic and cardiovascular interventions. The increasing investment in advanced medical infrastructure and technology by hospitals worldwide, particularly in developed economies, further solidifies this segment's leadership.
The Specialty Clinic segment is exhibiting robust growth, projected to expand at a CAGR of xx% during the forecast period. This growth is fueled by the increasing specialization in healthcare, leading to the proliferation of clinics focused on specific surgical disciplines such as ophthalmology, gynecology, and dermatology. These specialized clinics often require precision instruments like ring forceps for intricate procedures.
In terms of Types, Stainless Steel remains the most prevalent material for Medical Ring Forceps, occupying approximately xx% of the market share due to its cost-effectiveness, durability, and established manufacturing processes. However, Titanium is rapidly gaining traction, projected to grow at a CAGR of xx%, driven by its superior biocompatibility, lighter weight, and enhanced corrosion resistance, making it ideal for sensitive surgical applications and reducing patient allergic reactions. The Other Materials segment, encompassing advanced composites and specialized alloys, is a niche but growing area, particularly for novel applications and high-performance requirements.
- Dominant Application Segment (Hospital):
- Key Drivers: High surgical procedure volume, extensive investment in medical technology, presence of multidisciplinary surgical teams, and government healthcare initiatives.
- Detailed Dominance Analysis: Hospitals are the primary adopters of surgical instruments due to the critical nature of procedures performed, requiring a wide array of reliable and precise tools. The ongoing upgrades to surgical suites and the adoption of advanced imaging technologies necessitate complementary high-performance instruments like ring forceps.
- Leading Region (North America):
- Key Drivers: Advanced healthcare infrastructure, high disposable incomes, strong R&D expenditure in medical devices, and a large aging population demanding surgical interventions.
- Detailed Dominance Analysis: North America consistently leads in the adoption of new medical technologies and surgical techniques, making it a prime market for innovative Medical Ring Forceps. The robust reimbursement policies and a well-established network of leading hospitals and surgical centers contribute significantly to market dominance.
- Dominant Type Segment (Stainless Steel):
- Key Drivers: Cost-effectiveness, wide availability, proven track record of durability and reliability in sterilization processes, and established manufacturing expertise.
- Detailed Dominance Analysis: Stainless steel's balance of affordability and performance makes it the default choice for many surgical applications and budgetary constraints in healthcare facilities. Its widespread acceptance by surgeons and compatibility with standard sterilization protocols ensure its continued market dominance.
- Emerging Type Segment (Titanium):
- Key Drivers: Biocompatibility, lightweight properties reducing surgeon fatigue, superior corrosion resistance, and demand for hypoallergenic materials.
- Detailed Dominance Analysis: The growing awareness of material science in healthcare and the increasing focus on patient well-being are driving the adoption of titanium. Its benefits are particularly pronounced in delicate surgeries and for patients with metal sensitivities.
Medical Ring Forceps Product Innovations
Recent product innovations in the Medical Ring Forceps sector are centered on enhancing precision, ergonomics, and material science. Manufacturers are developing titanium ring forceps with advanced coatings for improved lubricity and reduced tissue drag. Innovations include forceps with finer tips for micro-surgical applications and ergonomically designed handles to minimize surgeon fatigue during prolonged procedures. Developments also encompass intelligent sterilization-compatible materials and the integration of antimicrobial properties to further reduce infection risks. These advancements aim to improve procedural efficiency and patient safety, offering a competitive edge in the market.
Report Segmentation & Scope
This report segments the Medical Ring Forceps market across key parameters to provide a granular view. The Application segmentation includes Hospital, Specialty Clinic, and Others. The Type segmentation encompasses Titanium, Stainless Steel, and Other Materials.
- Hospital Segment: Projected to reach approximately hundreds of millions billion by 2033, driven by high procedure volumes.
- Specialty Clinic Segment: Expected to grow at a CAGR of xx%, fueled by increasing medical specialization.
- Others Segment: Includes veterinary clinics, research laboratories, and dental practices, with an estimated market size of tens of millions billion.
- Titanium Segment: Forecasted to grow significantly, with a market size of hundreds of millions billion by 2033, due to its advanced properties.
- Stainless Steel Segment: The largest segment, with a projected market size of billions billion, due to its widespread adoption and cost-effectiveness.
- Other Materials Segment: A niche segment with projected growth of xx%, driven by specialized applications.
Key Drivers of Medical Ring Forceps Growth
The growth of the Medical Ring Forceps market is propelled by several interconnected factors. Technological advancements in surgical instruments, including the development of novel materials and ergonomic designs, are paramount. The economic factor of a rising global healthcare expenditure, coupled with increasing insurance coverage, enables greater access to surgical procedures and advanced medical devices. Regulatory support for innovation and the approval of new, improved surgical tools also contribute significantly. Furthermore, the aging global population is a key demographic driver, as older individuals are more prone to conditions requiring surgical intervention. The rising prevalence of chronic diseases requiring ongoing medical management and potential surgical solutions further bolsters demand.
Challenges in the Medical Ring Forceps Sector
Despite the positive growth outlook, the Medical Ring Forceps sector faces several challenges. Stringent regulatory hurdles for new product approvals can be time-consuming and costly, potentially delaying market entry. Supply chain disruptions, as witnessed in recent global events, can impact the availability of raw materials and finished goods, affecting production and delivery timelines. Intense competitive pressures among manufacturers lead to price sensitivity and the need for continuous innovation to maintain market share. The high cost of advanced materials like titanium can also limit their adoption in budget-constrained healthcare systems. The ongoing need for skilled surgical professionals capable of utilizing these advanced instruments effectively is another consideration.
Leading Players in the Medical Ring Forceps Market
- Blu-Med
- Starrco
- Mecart
- Esco Lifesciences Group
- Beth-El Group
- Terra Universal
- Halton
- BIOBASE
- Stryker
Key Developments in Medical Ring Forceps Sector
- 2023/10: Launch of a new line of antimicrobial-coated stainless steel ring forceps by a leading manufacturer, enhancing infection control capabilities.
- 2023/07: Starrco announces strategic partnership to expand distribution of its titanium ring forceps in emerging Asian markets.
- 2022/12: Mecart receives FDA clearance for its innovative ergonomic ring forceps, designed to reduce surgeon fatigue.
- 2022/05: Esco Lifesciences Group acquires a niche player specializing in micro-ring forceps, strengthening its portfolio for specialized surgical procedures.
- 2021/09: Terra Universal introduces a line of reusable ring forceps with enhanced sterilization protocols, focusing on sustainability.
- 2020/03: Stryker invests heavily in R&D for next-generation surgical instruments, including advanced ring forceps.
Strategic Medical Ring Forceps Market Outlook
The strategic outlook for the Medical Ring Forceps market is exceptionally promising, driven by sustained demand for advanced surgical solutions and a favorable global demographic landscape. Growth accelerators include the continued embrace of minimally invasive surgical techniques, necessitating precision instruments, and the increasing investment in healthcare infrastructure across emerging economies. Opportunities lie in the development of smart forceps with integrated sensor technology, enhanced biocompatible materials, and the expansion into underserved markets. The strategic focus for market players should be on continuous product innovation, building robust distribution networks, and forging strategic alliances to capitalize on the burgeoning demand for high-quality and specialized medical instruments.
Medical Ring Forceps Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Specialty Clinic
- 1.3. Others
-
2. Types
- 2.1. Titanium
- 2.2. Stainless Steel
- 2.3. Other Materials
Medical Ring Forceps Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
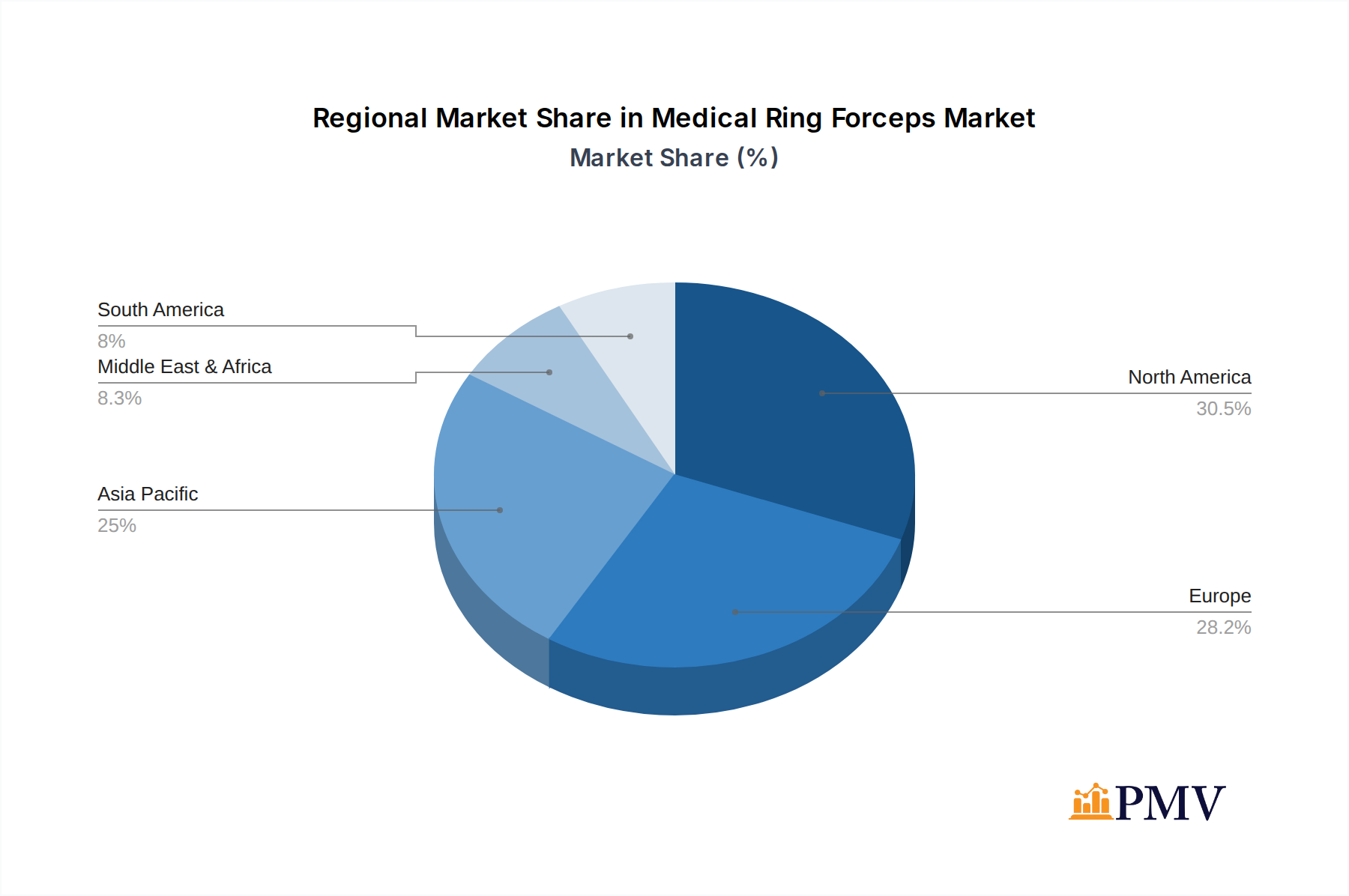
Medical Ring Forceps Regional Market Share

Geographic Coverage of Medical Ring Forceps
Medical Ring Forceps REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Specialty Clinic
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Titanium
- 5.2.2. Stainless Steel
- 5.2.3. Other Materials
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Medical Ring Forceps Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Specialty Clinic
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Titanium
- 6.2.2. Stainless Steel
- 6.2.3. Other Materials
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Medical Ring Forceps Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Specialty Clinic
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Titanium
- 7.2.2. Stainless Steel
- 7.2.3. Other Materials
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Medical Ring Forceps Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Specialty Clinic
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Titanium
- 8.2.2. Stainless Steel
- 8.2.3. Other Materials
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Medical Ring Forceps Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Specialty Clinic
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Titanium
- 9.2.2. Stainless Steel
- 9.2.3. Other Materials
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Medical Ring Forceps Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Specialty Clinic
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Titanium
- 10.2.2. Stainless Steel
- 10.2.3. Other Materials
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Medical Ring Forceps Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospital
- 11.1.2. Specialty Clinic
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Titanium
- 11.2.2. Stainless Steel
- 11.2.3. Other Materials
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Blu-Med
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Starrco
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Mecart
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Esco Lifesciences Group
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Beth-El Group
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Terra Universal
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Halton
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 BIOBASE
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Stryker
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.1 Blu-Med
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Medical Ring Forceps Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: Global Medical Ring Forceps Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Medical Ring Forceps Revenue (billion), by Application 2025 & 2033
- Figure 4: North America Medical Ring Forceps Volume (K), by Application 2025 & 2033
- Figure 5: North America Medical Ring Forceps Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Medical Ring Forceps Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Medical Ring Forceps Revenue (billion), by Types 2025 & 2033
- Figure 8: North America Medical Ring Forceps Volume (K), by Types 2025 & 2033
- Figure 9: North America Medical Ring Forceps Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Medical Ring Forceps Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Medical Ring Forceps Revenue (billion), by Country 2025 & 2033
- Figure 12: North America Medical Ring Forceps Volume (K), by Country 2025 & 2033
- Figure 13: North America Medical Ring Forceps Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Medical Ring Forceps Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Medical Ring Forceps Revenue (billion), by Application 2025 & 2033
- Figure 16: South America Medical Ring Forceps Volume (K), by Application 2025 & 2033
- Figure 17: South America Medical Ring Forceps Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Medical Ring Forceps Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Medical Ring Forceps Revenue (billion), by Types 2025 & 2033
- Figure 20: South America Medical Ring Forceps Volume (K), by Types 2025 & 2033
- Figure 21: South America Medical Ring Forceps Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Medical Ring Forceps Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Medical Ring Forceps Revenue (billion), by Country 2025 & 2033
- Figure 24: South America Medical Ring Forceps Volume (K), by Country 2025 & 2033
- Figure 25: South America Medical Ring Forceps Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Medical Ring Forceps Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Medical Ring Forceps Revenue (billion), by Application 2025 & 2033
- Figure 28: Europe Medical Ring Forceps Volume (K), by Application 2025 & 2033
- Figure 29: Europe Medical Ring Forceps Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Medical Ring Forceps Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Medical Ring Forceps Revenue (billion), by Types 2025 & 2033
- Figure 32: Europe Medical Ring Forceps Volume (K), by Types 2025 & 2033
- Figure 33: Europe Medical Ring Forceps Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Medical Ring Forceps Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Medical Ring Forceps Revenue (billion), by Country 2025 & 2033
- Figure 36: Europe Medical Ring Forceps Volume (K), by Country 2025 & 2033
- Figure 37: Europe Medical Ring Forceps Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Medical Ring Forceps Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Medical Ring Forceps Revenue (billion), by Application 2025 & 2033
- Figure 40: Middle East & Africa Medical Ring Forceps Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Medical Ring Forceps Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Medical Ring Forceps Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Medical Ring Forceps Revenue (billion), by Types 2025 & 2033
- Figure 44: Middle East & Africa Medical Ring Forceps Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Medical Ring Forceps Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Medical Ring Forceps Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Medical Ring Forceps Revenue (billion), by Country 2025 & 2033
- Figure 48: Middle East & Africa Medical Ring Forceps Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Medical Ring Forceps Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Medical Ring Forceps Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Medical Ring Forceps Revenue (billion), by Application 2025 & 2033
- Figure 52: Asia Pacific Medical Ring Forceps Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Medical Ring Forceps Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Medical Ring Forceps Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Medical Ring Forceps Revenue (billion), by Types 2025 & 2033
- Figure 56: Asia Pacific Medical Ring Forceps Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Medical Ring Forceps Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Medical Ring Forceps Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Medical Ring Forceps Revenue (billion), by Country 2025 & 2033
- Figure 60: Asia Pacific Medical Ring Forceps Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Medical Ring Forceps Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Medical Ring Forceps Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Medical Ring Forceps Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Medical Ring Forceps Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Medical Ring Forceps Revenue billion Forecast, by Types 2020 & 2033
- Table 4: Global Medical Ring Forceps Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Medical Ring Forceps Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Global Medical Ring Forceps Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Medical Ring Forceps Revenue billion Forecast, by Application 2020 & 2033
- Table 8: Global Medical Ring Forceps Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Medical Ring Forceps Revenue billion Forecast, by Types 2020 & 2033
- Table 10: Global Medical Ring Forceps Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Medical Ring Forceps Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Global Medical Ring Forceps Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United States Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Canada Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Mexico Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Medical Ring Forceps Revenue billion Forecast, by Application 2020 & 2033
- Table 20: Global Medical Ring Forceps Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Medical Ring Forceps Revenue billion Forecast, by Types 2020 & 2033
- Table 22: Global Medical Ring Forceps Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Medical Ring Forceps Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Global Medical Ring Forceps Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Brazil Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Argentina Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Medical Ring Forceps Revenue billion Forecast, by Application 2020 & 2033
- Table 32: Global Medical Ring Forceps Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Medical Ring Forceps Revenue billion Forecast, by Types 2020 & 2033
- Table 34: Global Medical Ring Forceps Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Medical Ring Forceps Revenue billion Forecast, by Country 2020 & 2033
- Table 36: Global Medical Ring Forceps Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 40: Germany Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: France Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: Italy Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Spain Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 48: Russia Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 50: Benelux Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 52: Nordics Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Medical Ring Forceps Revenue billion Forecast, by Application 2020 & 2033
- Table 56: Global Medical Ring Forceps Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Medical Ring Forceps Revenue billion Forecast, by Types 2020 & 2033
- Table 58: Global Medical Ring Forceps Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Medical Ring Forceps Revenue billion Forecast, by Country 2020 & 2033
- Table 60: Global Medical Ring Forceps Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 62: Turkey Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 64: Israel Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 66: GCC Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 68: North Africa Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 70: South Africa Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Medical Ring Forceps Revenue billion Forecast, by Application 2020 & 2033
- Table 74: Global Medical Ring Forceps Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Medical Ring Forceps Revenue billion Forecast, by Types 2020 & 2033
- Table 76: Global Medical Ring Forceps Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Medical Ring Forceps Revenue billion Forecast, by Country 2020 & 2033
- Table 78: Global Medical Ring Forceps Volume K Forecast, by Country 2020 & 2033
- Table 79: China Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 80: China Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 82: India Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 84: Japan Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 86: South Korea Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 90: Oceania Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Medical Ring Forceps Revenue (billion) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Medical Ring Forceps Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Ring Forceps?
The projected CAGR is approximately 7.3%.
2. Which companies are prominent players in the Medical Ring Forceps?
Key companies in the market include Blu-Med, Starrco, Mecart, Esco Lifesciences Group, Beth-El Group, Terra Universal, Halton, BIOBASE, Stryker.
3. What are the main segments of the Medical Ring Forceps?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.39 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4350.00, USD 6525.00, and USD 8700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Medical Ring Forceps," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Medical Ring Forceps report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Medical Ring Forceps?
To stay informed about further developments, trends, and reports in the Medical Ring Forceps, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
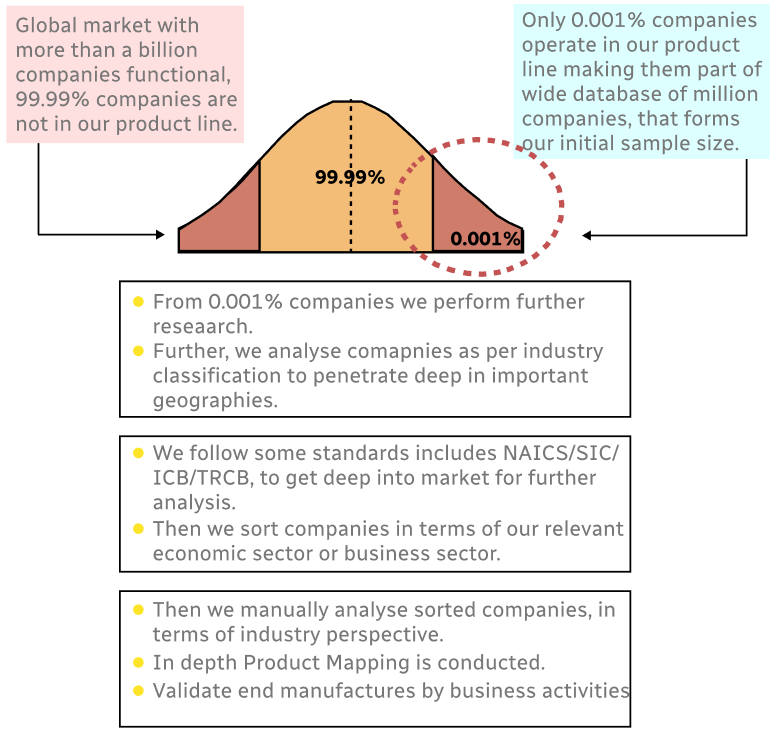
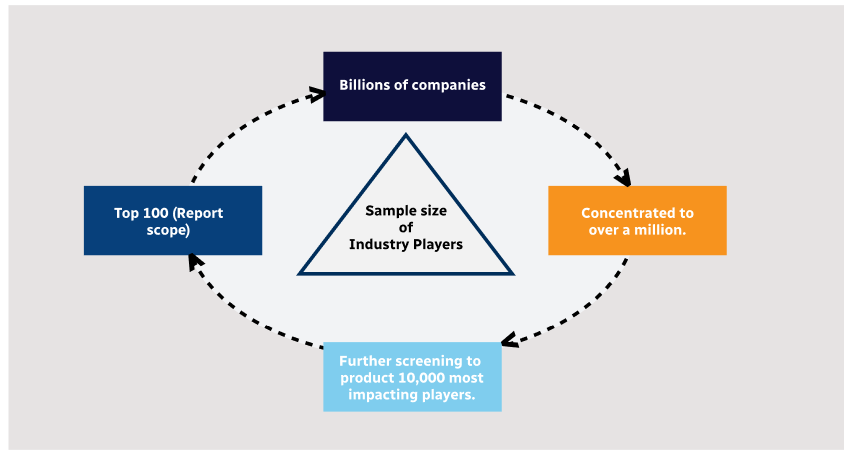
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
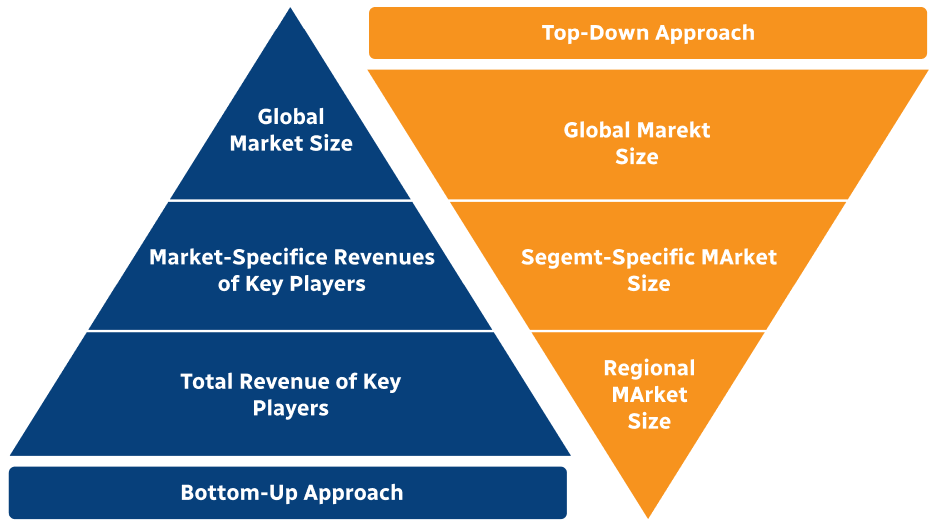
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


