Key Insights
The Medical Flow Twin Valve market is poised for robust growth, projected to reach an estimated USD 11,250.75 million in 2024, expanding at a Compound Annual Growth Rate (CAGR) of 7.15% through 2033. This significant expansion is primarily fueled by the increasing global demand for advanced medical devices, the rising prevalence of chronic respiratory diseases, and the continuous innovation in healthcare infrastructure. The market is witnessing strong adoption across various applications, with Hospitals leading the segment due to the critical need for precise fluid and gas management in critical care settings, surgical procedures, and general patient care. Home healthcare settings are also emerging as a significant growth avenue, driven by the increasing number of patients requiring continuous medical support at home and the growing preference for decentralized healthcare solutions. Clinics, while a smaller segment, are also contributing to market expansion with the adoption of specialized flow control devices for outpatient procedures and diagnostics.
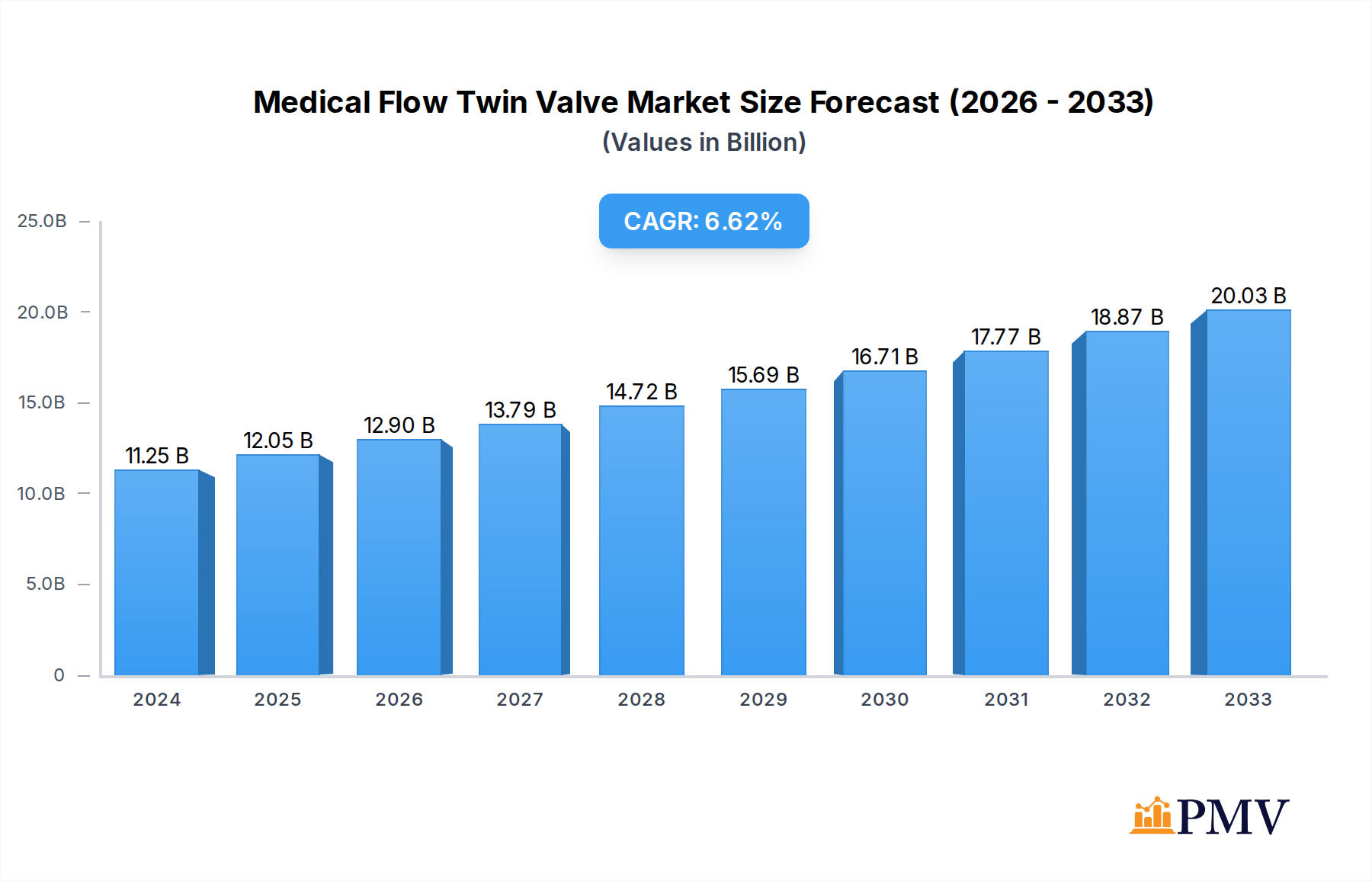
Medical Flow Twin Valve Market Size (In Billion)

The market's trajectory is further shaped by the evolution of product types, with Two-Stage valves gaining traction due to their enhanced precision and control capabilities, particularly in applications demanding fine-tuning of flow rates. Drivers such as the escalating healthcare expenditure worldwide, coupled with government initiatives promoting advanced medical technology adoption, are creating a favorable environment for market players. Emerging trends include the integration of smart technologies, such as IoT connectivity and real-time monitoring, into medical flow twin valves, enhancing their functionality and enabling remote patient management. However, the market also faces certain restraints, including the high cost of advanced technology, stringent regulatory approvals, and the need for skilled personnel for installation and maintenance. Despite these challenges, the inherent demand for reliable and accurate medical flow control solutions, coupled with strategic investments by key companies like Baxter, HEYER Medical, and Megasan Medical Gas Systems, underscores a promising future for the Medical Flow Twin Valve market.
Medical Flow Twin Valve Company Market Share

This in-depth market research report provides a detailed analysis of the global Medical Flow Twin Valve market, covering historical data from 2019 to 2024, a base year of 2025, and a robust forecast period extending to 2033. The report offers actionable insights for stakeholders, including manufacturers, suppliers, distributors, and investors, enabling strategic decision-making in this rapidly evolving healthcare sector. With a focus on key industry developments and market segmentation, this report is an indispensable resource for understanding the current landscape and future trajectory of medical flow twin valves.
The report meticulously examines the market structure, competitive dynamics, industry trends, dominant segments, product innovations, and strategic outlook. It addresses critical factors influencing market growth, challenges faced by industry players, and key developments shaping the sector. The analysis encompasses a wide range of applications including Hospital, Home, and Clinic settings, and categorizes products by types such as Single-Stage and Two-Stage valves. Leading companies like Baxter, HEYER Medical, Megasan Medical Gas Systems, Direct Flow Medical, Tavan Jam, Flow-Meter, DZ Medicale, Cagdas Elektronik Medikal are profiled, alongside significant industry developments throughout the study period.
Medical Flow Twin Valve Market Structure & Competitive Dynamics
The Medical Flow Twin Valve market exhibits a moderate to high degree of market concentration, with a few dominant players accounting for a significant portion of the global market share. Key players are actively investing in research and development to introduce innovative products, fostering a dynamic innovation ecosystem. Regulatory frameworks, particularly those governing medical devices and gas delivery systems, play a crucial role in shaping market entry and product approval. The presence of product substitutes, such as single-valve flow regulators, presents a competitive challenge, although twin valves offer distinct advantages in terms of precision and safety. End-user trends are leaning towards demand for more sophisticated, user-friendly, and digitally integrated medical devices, driving innovation in this space. Mergers and acquisitions (M&A) activities are anticipated to continue as companies seek to expand their product portfolios, gain market access, and achieve economies of scale. Anticipated M&A deal values in the coming forecast period are expected to reach several hundred million, with some significant transactions potentially exceeding one million. The continuous drive for enhanced patient care and improved healthcare outcomes fuels the demand for high-quality medical flow twin valves.
- Market Share Dynamics: Leading companies command substantial market share, with projections indicating a stable but evolving landscape.
- Innovation Ecosystem: Strong emphasis on R&D, with an estimated investment of several million annually by top-tier manufacturers.
- Regulatory Influence: Stringent regulations in major markets necessitate robust compliance strategies for market entry.
- Product Substitutes: Competitors offering single-valve solutions are present, but twin valves offer superior performance for critical applications.
- End-User Preferences: Growing demand for advanced features, connectivity, and enhanced safety protocols.
- M&A Activity: Strategic consolidation is expected to continue, with deal values potentially reaching several hundred million, some individual deals exceeding one million.
Medical Flow Twin Valve Industry Trends & Insights
The Medical Flow Twin Valve industry is experiencing robust growth, propelled by several key market growth drivers and technological disruptions. The Compound Annual Growth Rate (CAGR) for the forecast period is projected to be around XX% to XX%, reflecting a healthy expansion trajectory. A significant factor contributing to this growth is the increasing demand for precise medical gas delivery in critical care settings. Technological advancements, such as the integration of smart features, IoT connectivity for remote monitoring, and miniaturization of components, are reshaping product offerings and enhancing user experience. Consumer preferences are increasingly shifting towards devices that offer greater patient comfort, improved safety, and ease of use, particularly in home healthcare settings. Competitive dynamics are characterized by a blend of established players and emerging innovators, all vying for market share through product differentiation and strategic partnerships. The market penetration of advanced twin valve systems is steadily increasing, driven by rising awareness of their benefits in critical applications like anesthesia, mechanical ventilation, and oxygen therapy. The expanding healthcare infrastructure globally, coupled with a growing elderly population requiring specialized medical care, further fuels the demand for these essential medical devices. Furthermore, the ongoing global health crises have underscored the critical importance of reliable and efficient medical gas supply systems, leading to increased investment and focus on this sector. Innovation in materials science and manufacturing processes is also contributing to the development of more cost-effective and durable twin valve solutions, widening their accessibility across diverse healthcare environments. The shift towards personalized medicine and the increasing complexity of medical procedures also necessitate highly accurate and responsive flow control, which twin valves are well-equipped to provide. The market is witnessing a gradual but significant increase in the adoption of twin valves in emerging economies, driven by improving healthcare standards and increased government spending on medical infrastructure. The global market size is estimated to be in the billions of dollars for the base year 2025, with projected substantial growth over the forecast period.
Dominant Markets & Segments in Medical Flow Twin Valve
The Hospital segment is currently the dominant market for Medical Flow Twin Valves, driven by the critical need for precise medical gas management in operating rooms, intensive care units (ICUs), and emergency departments. The widespread adoption of advanced medical equipment and the increasing complexity of surgical procedures within hospitals necessitate the use of high-precision flow control devices. Economic policies that prioritize healthcare infrastructure development and increased government funding for public hospitals contribute significantly to this dominance. Furthermore, the growing number of hospital beds and the continuous drive to improve patient safety and clinical outcomes in hospital settings further solidify the hospital segment's leading position.
The Single-Stage type of Medical Flow Twin Valve currently holds a larger market share due to its established presence and application in a wide range of medical gas regulation needs. These valves offer a balance of performance and cost-effectiveness, making them a preferred choice for many standard medical gas delivery systems. However, the Two-Stage valve segment is witnessing rapid growth, driven by the increasing demand for extremely precise and stable gas flow regulation in highly sensitive applications such as neonatal care, advanced ventilation, and specialized anesthesia delivery. The technological advancements enabling greater accuracy and responsiveness in two-stage systems are attracting significant interest from healthcare providers seeking the highest levels of patient safety and treatment efficacy.
Regionally, North America and Europe currently represent the largest markets for Medical Flow Twin Valves, owing to their well-established healthcare infrastructure, high healthcare expenditure, and the early adoption of advanced medical technologies. Stringent regulatory standards and a strong emphasis on patient safety in these regions further drive the demand for high-quality, reliable medical flow twin valves.
- Dominant Application: Hospital settings are the primary consumers, demanding precise gas regulation for critical care.
- Key Drivers in Hospitals: Advanced medical procedures, stringent safety protocols, and increasing ICU/OR utilization.
- Dominant Type: Single-Stage valves are widely adopted for general applications.
- Growth Driver for Two-Stage: Demand for ultra-precise flow control in specialized medical applications.
- Leading Regions: North America and Europe due to advanced healthcare infrastructure and high expenditure.
- Regional Economic Factors: Favorable economic policies and substantial government investment in healthcare infrastructure in leading regions.
Medical Flow Twin Valve Product Innovations
Product innovation in the Medical Flow Twin Valve sector is characterized by a relentless pursuit of enhanced accuracy, improved patient safety, and greater user convenience. Key developments include the integration of digital interfaces for real-time monitoring and data logging, enabling better clinical decision-making. Advancements in materials science are leading to the development of more durable, lightweight, and biocompatible valves. Manufacturers are also focusing on miniaturization to create more compact and portable devices, particularly beneficial for home healthcare and portable medical equipment. Competitive advantages are being gained through enhanced flow control precision, reduced dead space, and improved leak prevention. The market is witnessing the emergence of smart valves that can self-diagnose and alert users to potential issues, further bolstering patient safety and operational efficiency.
Report Segmentation & Scope
This report provides a comprehensive segmentation of the Medical Flow Twin Valve market, offering granular insights into various segments. The segmentation is broadly categorized by Application and Type.
Application Segmentation:
- Hospital: This segment is expected to maintain its leading position throughout the forecast period, driven by continuous demand for critical care applications and advanced medical procedures. Market size for this segment is estimated to be in the hundreds of millions for the base year.
- Home: The home healthcare segment is projected for significant growth, fueled by the increasing prevalence of chronic diseases, aging populations, and a preference for remote patient care. This segment's growth is estimated to be in the tens of millions for the base year.
- Clinic: Clinics represent a growing segment, especially those specializing in respiratory care or outpatient procedures, demanding reliable and cost-effective flow control solutions. Market size for this segment is estimated to be in the tens of millions for the base year.
Type Segmentation:
- Single-Stage: While established, this segment is expected to see steady growth, catering to a broad range of applications where extreme precision is not paramount. Projected growth is steady.
- Two-Stage: This segment is poised for substantial expansion, driven by the demand for highly accurate and stable flow control in critical medical applications. This segment's growth rate is projected to be higher than single-stage.
Key Drivers of Medical Flow Twin Valve Growth
The growth of the Medical Flow Twin Valve market is propelled by a confluence of technological, economic, and regulatory factors.
- Technological Advancements: Continuous innovation in precision engineering, material science, and digital integration leads to the development of superior valve functionalities. For instance, advancements in sensor technology enable more accurate real-time flow monitoring, a critical factor in life-support systems.
- Increasing Healthcare Expenditure: Global healthcare spending continues to rise, particularly in emerging economies, leading to increased investment in medical infrastructure and advanced medical devices. This trend directly benefits the demand for high-quality medical flow twin valves, with an estimated several hundred million dollars being invested annually in upgrading medical gas systems.
- Growing Prevalence of Chronic Diseases: The increasing incidence of respiratory illnesses, cardiovascular diseases, and other chronic conditions requiring long-term medical gas therapy is a significant demand driver. The aging global population further exacerbates this trend, necessitating more sophisticated home healthcare solutions.
- Stringent Regulatory Standards: Regulatory bodies worldwide are increasingly emphasizing patient safety and device efficacy, driving the adoption of advanced and reliable medical flow twin valves. Compliance with standards such as ISO 13485 and CE marking is crucial for market access.
Challenges in the Medical Flow Twin Valve Sector
Despite the positive growth trajectory, the Medical Flow Twin Valve sector faces several challenges that can impact market expansion.
- High Cost of Advanced Technologies: The development and integration of cutting-edge technologies in medical flow twin valves often result in higher manufacturing costs, which can translate into premium pricing, potentially limiting adoption in price-sensitive markets.
- Intense Competition: The market is characterized by a moderate to high level of competition, with both established global players and emerging regional manufacturers vying for market share. This intense competition can lead to price erosion and pressure on profit margins.
- Stringent Regulatory Hurdles: While regulatory standards drive quality, the process of obtaining approvals for new medical devices can be lengthy, complex, and expensive, posing a significant barrier to entry for smaller companies.
- Supply Chain Disruptions: Global supply chain vulnerabilities, as witnessed in recent years, can affect the availability of critical raw materials and components, leading to production delays and increased costs. The impact of such disruptions can range from minor delays to significant production halts, affecting hundreds of millions in potential revenue.
Leading Players in the Medical Flow Twin Valve Market
- Baxter
- HEYER Medical
- Megasan Medical Gas Systems
- Direct Flow Medical
- Tavan Jam
- Flow-Meter
- DZ Medicale
- Cagdas Elektronik Medikal
Key Developments in Medical Flow Twin Valve Sector
- 2023 Q4: Baxter launched a new generation of smart medical flow regulators with enhanced connectivity features, aiming to improve patient monitoring in critical care settings.
- 2024 Q1: HEYER Medical announced a strategic partnership with a leading telemedicine provider to integrate their flow twin valve systems with remote patient monitoring platforms, targeting the home healthcare market.
- 2024 Q2: Megasan Medical Gas Systems expanded its manufacturing capacity by opening a new state-of-the-art facility, projecting a significant increase in production volume to meet growing global demand.
- 2024 Q3: Direct Flow Medical secured CE marking for its innovative two-stage medical flow twin valve, enabling wider market access in Europe for its advanced respiratory therapy solutions.
- 2024 Q4: Tavan Jam introduced a new line of compact and lightweight medical flow twin valves designed for portable medical equipment and ambulance services.
Strategic Medical Flow Twin Valve Market Outlook
The strategic outlook for the Medical Flow Twin Valve market is highly positive, driven by sustained demand for precision medical gas delivery and ongoing technological advancements. Growth accelerators include the expanding home healthcare sector, the increasing adoption of smart medical devices, and the continuous need for reliable equipment in critical care. Companies that focus on product innovation, particularly in areas like IoT integration and advanced safety features, will be well-positioned for future success. Strategic partnerships and potential M&A activities will continue to play a vital role in consolidating market share and expanding geographical reach. The market is expected to witness a strong upward trend, with opportunities arising from the development of personalized medicine solutions and the increasing demand for efficient respiratory support systems across diverse healthcare settings. Investment in R&D, coupled with a keen understanding of evolving regulatory landscapes and end-user needs, will be paramount for navigating this dynamic market.
Medical Flow Twin Valve Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Home
- 1.3. Clinic
-
2. Types
- 2.1. Single-Stage
- 2.2. Two-Stage
Medical Flow Twin Valve Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
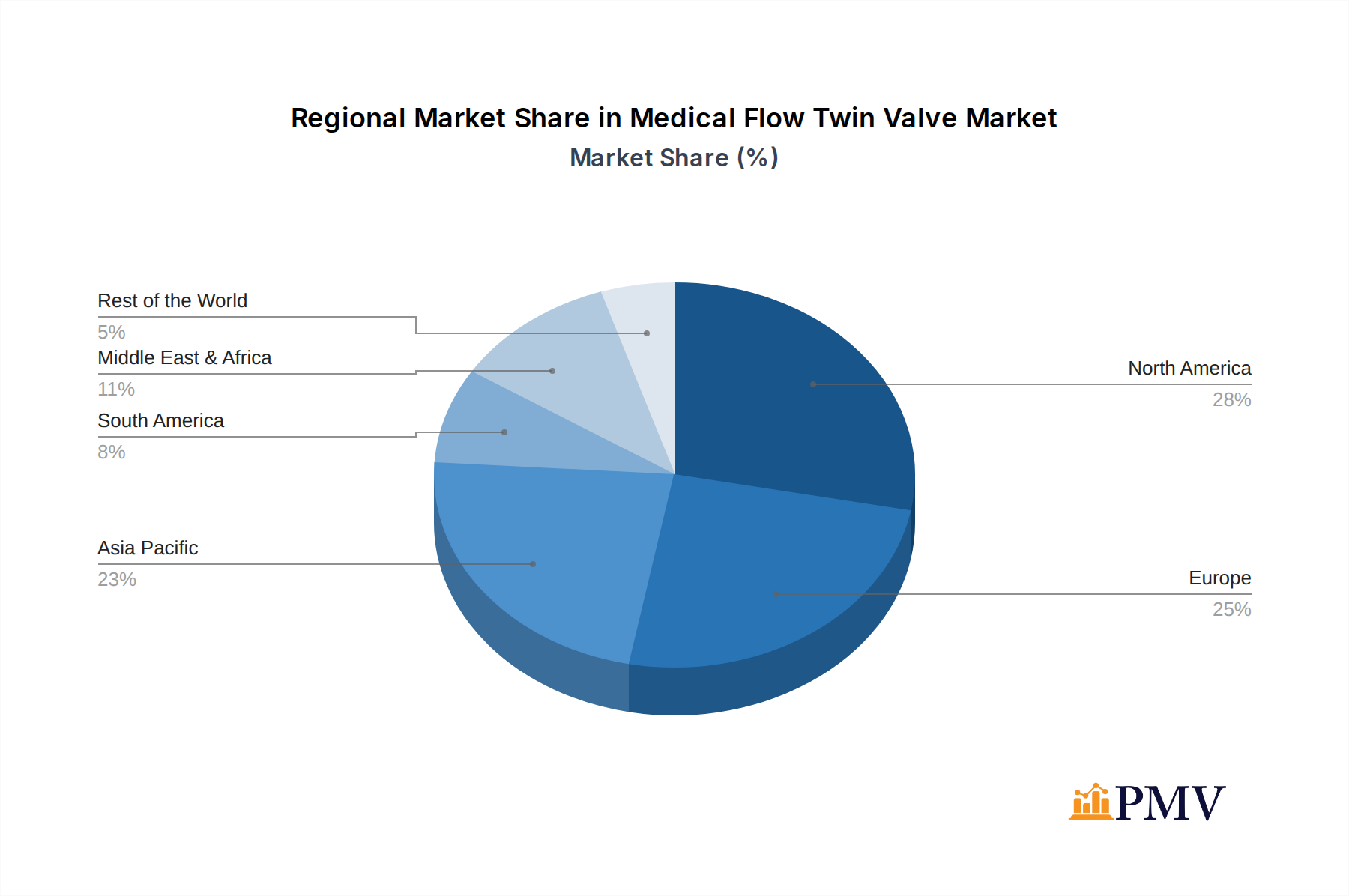
Medical Flow Twin Valve Regional Market Share

Geographic Coverage of Medical Flow Twin Valve
Medical Flow Twin Valve REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.15% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Home
- 5.1.3. Clinic
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Single-Stage
- 5.2.2. Two-Stage
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Medical Flow Twin Valve Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Home
- 6.1.3. Clinic
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Single-Stage
- 6.2.2. Two-Stage
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Medical Flow Twin Valve Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Home
- 7.1.3. Clinic
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Single-Stage
- 7.2.2. Two-Stage
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Medical Flow Twin Valve Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Home
- 8.1.3. Clinic
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Single-Stage
- 8.2.2. Two-Stage
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Medical Flow Twin Valve Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Home
- 9.1.3. Clinic
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Single-Stage
- 9.2.2. Two-Stage
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Medical Flow Twin Valve Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Home
- 10.1.3. Clinic
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Single-Stage
- 10.2.2. Two-Stage
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Medical Flow Twin Valve Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospital
- 11.1.2. Home
- 11.1.3. Clinic
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Single-Stage
- 11.2.2. Two-Stage
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Baxter
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 HEYER Medical
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Megasan Medical Gas Systems
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Direct Flow medical
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Tavan Jam
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Flow-Meter
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 DZ Medicale
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Cagdas Elektronik Medikal
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.1 Baxter
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Medical Flow Twin Valve Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Medical Flow Twin Valve Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Medical Flow Twin Valve Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Medical Flow Twin Valve Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Medical Flow Twin Valve Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Medical Flow Twin Valve Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Medical Flow Twin Valve Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Medical Flow Twin Valve Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Medical Flow Twin Valve Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Medical Flow Twin Valve Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Medical Flow Twin Valve Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Medical Flow Twin Valve Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Medical Flow Twin Valve Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Medical Flow Twin Valve Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Medical Flow Twin Valve Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Medical Flow Twin Valve Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Medical Flow Twin Valve Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Medical Flow Twin Valve Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Medical Flow Twin Valve Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Medical Flow Twin Valve Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Medical Flow Twin Valve Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Medical Flow Twin Valve Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Medical Flow Twin Valve Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Medical Flow Twin Valve Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Medical Flow Twin Valve Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Medical Flow Twin Valve Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Medical Flow Twin Valve Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Medical Flow Twin Valve Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Medical Flow Twin Valve Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Medical Flow Twin Valve Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Medical Flow Twin Valve Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Medical Flow Twin Valve Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Medical Flow Twin Valve Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Medical Flow Twin Valve Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Medical Flow Twin Valve Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Medical Flow Twin Valve Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Medical Flow Twin Valve Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Medical Flow Twin Valve Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Medical Flow Twin Valve Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Medical Flow Twin Valve Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Medical Flow Twin Valve Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Medical Flow Twin Valve Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Medical Flow Twin Valve Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Medical Flow Twin Valve Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Medical Flow Twin Valve Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Medical Flow Twin Valve Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Medical Flow Twin Valve Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Medical Flow Twin Valve Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Medical Flow Twin Valve Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Medical Flow Twin Valve Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Flow Twin Valve?
The projected CAGR is approximately 7.15%.
2. Which companies are prominent players in the Medical Flow Twin Valve?
Key companies in the market include Baxter, HEYER Medical, Megasan Medical Gas Systems, Direct Flow medical, Tavan Jam, Flow-Meter, DZ Medicale, Cagdas Elektronik Medikal.
3. What are the main segments of the Medical Flow Twin Valve?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Medical Flow Twin Valve," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Medical Flow Twin Valve report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Medical Flow Twin Valve?
To stay informed about further developments, trends, and reports in the Medical Flow Twin Valve, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
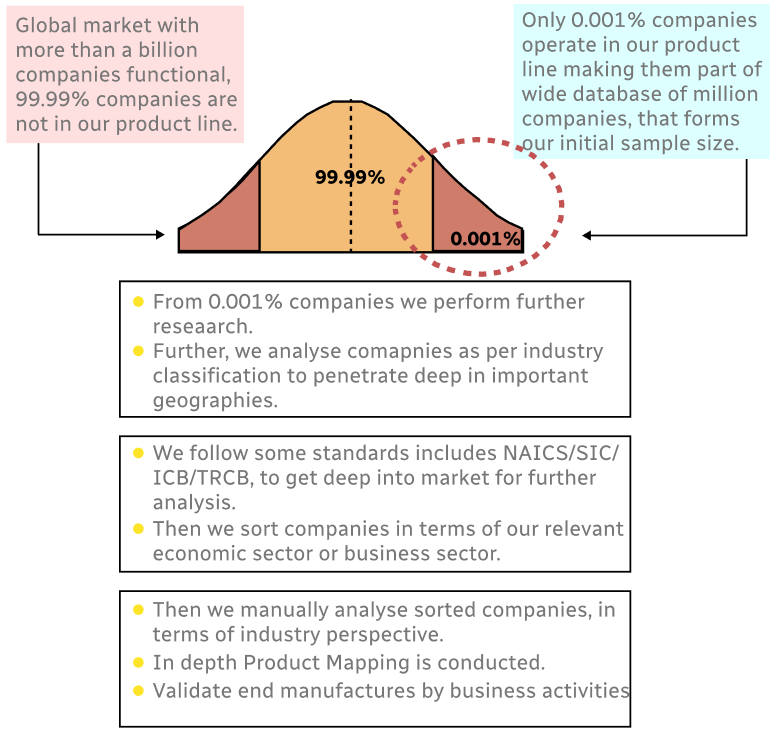
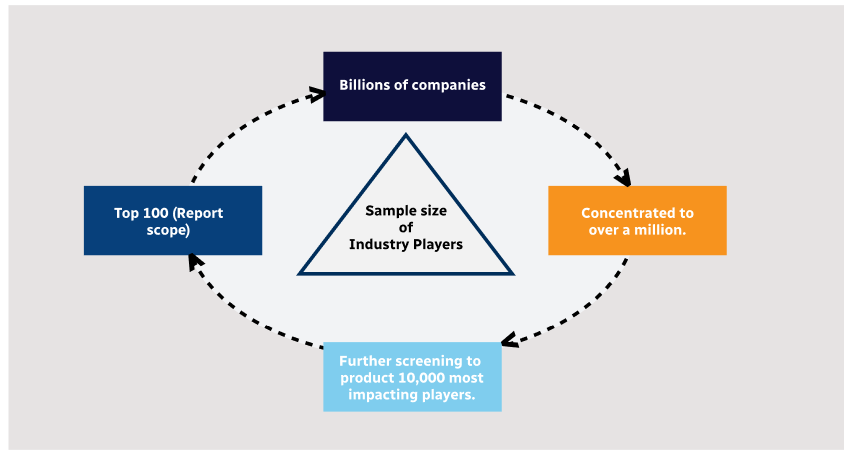
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
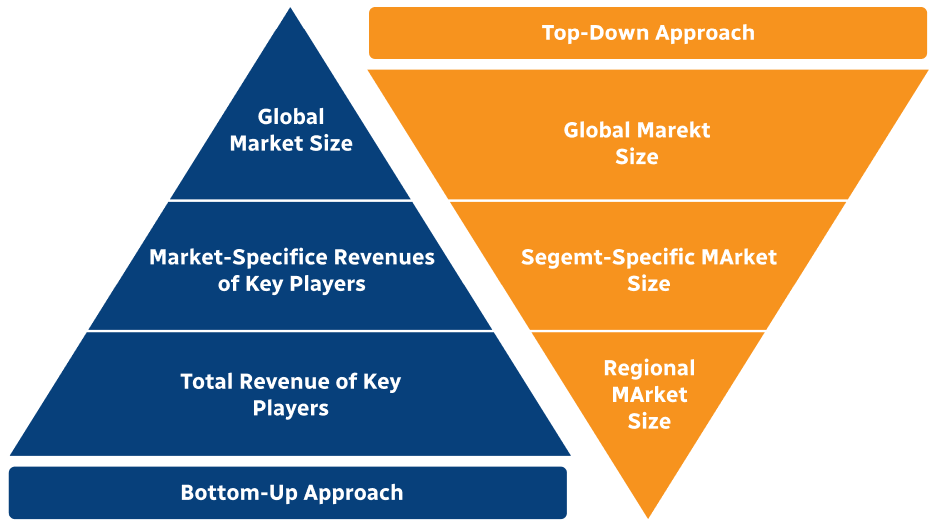
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
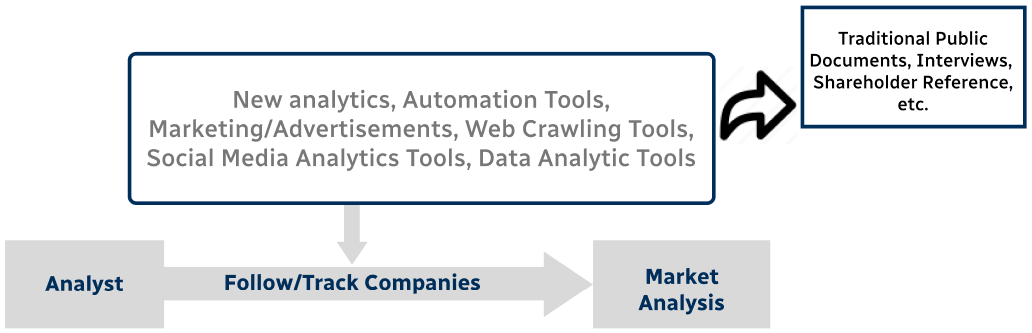
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


