Key Insights
The global Human Amniotic Fluid market is poised for substantial expansion, projected to reach a market size of approximately $1,200 million by 2025, with a robust Compound Annual Growth Rate (CAGR) of around 18% anticipated over the forecast period of 2025-2033. This significant growth is primarily propelled by a confluence of increasing awareness regarding the therapeutic potential of amniotic fluid, coupled with advancements in its collection, processing, and application technologies. The rising prevalence of chronic diseases and orthopedic conditions, which are increasingly being managed with regenerative medicine approaches utilizing amniotic fluid derivatives, is a major driver. Furthermore, the expanding research and development activities focused on unlocking novel applications in areas like wound healing, tissue regeneration, and ophthalmology are fueling market demand. The market is characterized by a dynamic landscape where innovations in both pure amniotic fluid products and expertly formulated mixtures are catering to diverse clinical needs across hospitals, specialty clinics, and diagnostics laboratories.
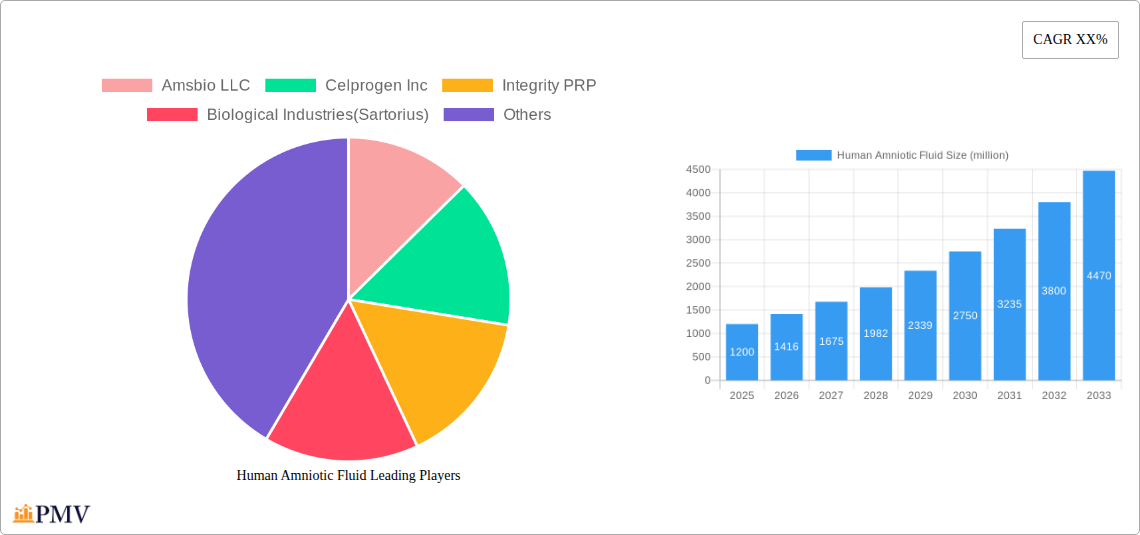
Human Amniotic Fluid Market Size (In Billion)

Despite the promising growth trajectory, the market faces certain restraints that could temper its full potential. Regulatory hurdles and the stringent approval processes for biological products can lead to prolonged development timelines and increased costs, potentially slowing down market penetration. Concerns regarding the ethical sourcing and standardization of amniotic fluid can also present challenges. However, the continuous evolution of bioengineering techniques and the development of standardized protocols are gradually addressing these concerns. The market is witnessing a strong trend towards the development of allogeneic amniotic fluid-derived products, offering a readily available and off-the-shelf solution for a wider patient base. Regionally, North America and Europe are expected to lead the market due to well-established healthcare infrastructure, high R&D spending, and a greater adoption of advanced regenerative therapies. However, the Asia Pacific region, driven by its large population, increasing healthcare expenditure, and growing awareness of regenerative medicine, is anticipated to exhibit the fastest growth rate in the coming years.
Human Amniotic Fluid Company Market Share

Human Amniotic Fluid Market Report: Comprehensive Analysis & Future Outlook (2019-2033)
This in-depth market research report provides a detailed analysis of the global Human Amniotic Fluid market, covering historical trends, current dynamics, and future projections. With a study period from 2019 to 2033, a base year of 2025, and a forecast period extending to 2033, this report offers actionable insights for stakeholders seeking to understand and capitalize on the growth opportunities within this rapidly evolving sector. We delve into market structure, key industry trends, dominant segments, product innovations, growth drivers, challenges, leading players, recent developments, and a strategic market outlook. This report is designed for immediate use without any modification.
Human Amniotic Fluid Market Structure & Competitive Dynamics
The global Human Amniotic Fluid market is characterized by a moderately concentrated structure, with a few key players holding significant market share. The market size is projected to reach several million by 2025, indicating substantial growth potential. Innovation ecosystems are thriving, driven by ongoing research and development in regenerative medicine and cell-based therapies, which heavily rely on amniotic fluid derivatives. Regulatory frameworks, while evolving, play a crucial role in dictating product approvals and market access, particularly concerning the ethical sourcing and processing of human amniotic fluid. Product substitutes, such as synthetic growth factors and other stem cell sources, present a competitive challenge, but the unique biological properties of amniotic fluid continue to drive its demand. End-user trends lean towards minimally invasive treatments and advanced wound healing solutions, fueling the adoption of amniotic fluid-based products. Merger and acquisition (M&A) activities, valued in the millions, are observed as companies seek to expand their product portfolios and geographical reach. The estimated M&A deal value for the forecast period is expected to exceed millions.
- Market Concentration: Moderate, with key players dominating market share.
- Innovation Ecosystems: Strong focus on R&D for regenerative medicine applications.
- Regulatory Frameworks: Evolving, with strict guidelines for ethical sourcing and processing.
- Product Substitutes: Synthetic growth factors, other stem cell sources.
- End-User Trends: Growing demand for regenerative therapies and advanced wound care.
- M&A Activities: Significant, driven by market expansion and portfolio enhancement.
Human Amniotic Fluid Industry Trends & Insights
The Human Amniotic Fluid industry is experiencing robust growth, driven by escalating demand for advanced regenerative medicine and therapeutic applications. The Compound Annual Growth Rate (CAGR) for the forecast period (2025-2033) is estimated to be in the double-digit percentage range, reflecting a significant market penetration by innovative amniotic fluid-derived products. Key market growth drivers include the increasing prevalence of chronic diseases requiring advanced treatment modalities, such as diabetic foot ulcers and burns, where amniotic fluid's regenerative properties offer significant therapeutic benefits. Technological disruptions, particularly advancements in cell culture techniques and bioprocessing, are enabling the isolation and purification of valuable biomolecules from amniotic fluid, leading to the development of novel therapeutics. Consumer preferences are shifting towards personalized medicine and bio-based treatments, further boosting the demand for amniotic fluid products. Competitive dynamics are intensifying as established biotechnology companies and emerging startups vie for market leadership through product innovation and strategic partnerships. The market penetration of amniotic fluid-based therapies is expected to reach approximately xx% by 2033, underscoring its growing importance in the healthcare landscape. Furthermore, the expanding research into the immunomodulatory and anti-inflammatory properties of amniotic fluid is opening up new therapeutic avenues, including its potential use in autoimmune diseases and tissue regeneration. The increasing awareness among healthcare professionals and patients about the benefits of amniotic fluid-derived products, coupled with favorable reimbursement policies in certain regions, are also contributing to the industry's upward trajectory. The ability of amniotic fluid to promote cell proliferation, differentiation, and reduce scarring makes it a highly sought-after biomaterial for a wide range of medical applications, from ophthalmology to orthopedics. The market size in the base year 2025 is estimated to be in the millions. The ongoing development of advanced delivery systems and formulations is also enhancing the efficacy and ease of use of these products, further accelerating market adoption. The market revenue is projected to grow by millions annually.
Dominant Markets & Segments in Human Amniotic Fluid
The Human Amniotic Fluid market exhibits clear dominance in specific regions and segments, driven by a confluence of economic policies, advanced healthcare infrastructure, and a high prevalence of target medical conditions.
Application:
- Hospitals: This segment currently holds the largest market share, estimated at over xx% of the total market value. The primary drivers include the critical care settings within hospitals, where amniotic fluid-derived products are utilized for complex wound healing, post-surgical recovery, and burn treatments. The availability of advanced surgical technologies and a higher volume of patient admissions for acute conditions contribute to this dominance. Economic policies supporting advanced healthcare treatments and established procurement channels within hospital networks further solidify its leading position.
- Specialty Clinics: This segment is witnessing rapid growth, projected to expand at a significant CAGR. Specialty clinics focusing on orthopedics, sports medicine, and dermatology are increasingly incorporating amniotic fluid for regenerative therapies, tendon repair, and skin rejuvenation. Favorable reimbursement policies for niche treatments and a growing patient preference for specialized care are key drivers.
- Diagnostics Laboratories: While a smaller segment, diagnostics laboratories are crucial for the characterization and quality control of amniotic fluid. Their role in research and development, identifying biomarkers, and ensuring product safety contributes to the overall market ecosystem.
- Others: This category encompasses research institutions and academic centers, which are instrumental in advancing the scientific understanding and clinical applications of amniotic fluid. Their innovative research fuels future market growth and product development.
Types:
- Pure Amniotic Fluid: This segment represents a significant portion of the market, valued at millions. Pure amniotic fluid, processed for specific therapeutic applications, offers a concentrated source of growth factors and extracellular matrix components. Its dominance is driven by its direct application in regenerative medicine and tissue engineering.
- Mixtures: This segment, including amniotic membrane and amniotic fluid-derived components in various formulations, is also experiencing substantial growth. The development of advanced mixtures tailored for specific indications, such as wound dressings and injectable therapies, is expanding its market reach. The market size for mixtures is estimated to be millions.
The dominance of these segments is further supported by favorable economic policies that encourage investment in regenerative medicine, robust healthcare infrastructure in developed nations, and an increasing incidence of conditions treatable with amniotic fluid. The market size in North America is expected to be the largest, estimated at over million in 2025, driven by strong research capabilities and advanced healthcare systems.
Human Amniotic Fluid Product Innovations
Recent product innovations in the Human Amniotic Fluid market focus on enhancing the therapeutic efficacy and expanding the application range of amniotic fluid-derived products. Developments include novel purification techniques that yield higher concentrations of key regenerative molecules, such as growth factors and exosomes, leading to more potent regenerative therapies. These innovations aim to improve outcomes in wound healing, cartilage repair, and ophthalmological applications. Furthermore, the development of advanced formulations, including bio-engineered scaffolds and injectable hydrogels, leverages amniotic fluid components for targeted tissue regeneration and drug delivery. These advancements provide competitive advantages by offering improved patient compliance and novel treatment modalities, with a projected market adoption increase of xx% by 2033. The market value of these innovations is estimated to reach millions.
Report Segmentation & Scope
This report meticulously segments the Human Amniotic Fluid market to provide granular insights into its diverse landscape. The segmentation is based on key applications and product types, enabling stakeholders to pinpoint specific growth opportunities and competitive dynamics.
Application:
- Hospital: This segment encompasses the use of amniotic fluid products in acute care settings, surgical procedures, and critical wound management. Growth projections for this segment are stable, with an estimated market size of million in 2025. Competitive dynamics are characterized by established medical device manufacturers and biotechnology firms.
- Specialty Clinic: This segment includes the application of amniotic fluid in outpatient settings for regenerative therapies, orthopedics, and dermatology. This segment is poised for significant growth, with projected market expansion driven by increasing demand for non-surgical regenerative treatments. The competitive landscape is dynamic, with innovative biotech startups gaining traction.
- Diagnostics Laboratory: This segment focuses on the use of amniotic fluid in research, biomarker discovery, and quality control. Its market size is estimated at million, with growth tied to advancements in molecular diagnostics and personalized medicine.
- Others: This broad category includes academic research, preclinical studies, and other niche applications. Its market size is projected to be million, with growth fueled by ongoing scientific exploration.
Types:
- Pure: This segment comprises unprocessed or minimally processed amniotic fluid used in specific therapeutic applications. Its market size is estimated at million in 2025, with stable growth driven by its direct regenerative potential.
- Mixtures: This segment includes amniotic fluid-derived components formulated into various products like gels, membranes, and injectable solutions. This segment is expected to experience robust growth, with an estimated market size of million, driven by the development of targeted and advanced therapeutic formulations.
Key Drivers of Human Amniotic Fluid Growth
The Human Amniotic Fluid market is propelled by several critical growth drivers, shaping its trajectory and unlocking new opportunities.
- Technological Advancements: Continuous innovation in bioprocessing, cell culture, and purification technologies allows for the extraction and utilization of a wider range of bioactive components from amniotic fluid, enhancing its therapeutic potential. This includes the isolation of exosomes and specific growth factors for targeted regenerative therapies.
- Increasing Demand for Regenerative Medicine: The global shift towards regenerative medicine, aiming to repair, replace, or regenerate damaged tissues and organs, directly fuels the demand for amniotic fluid as a natural source of stem cells and growth factors.
- Growing Prevalence of Chronic Diseases: Conditions like diabetic foot ulcers, chronic wounds, and osteoarthritis, which often exhibit poor healing and inflammation, are increasingly being treated with amniotic fluid-based therapies, thereby driving market expansion.
- Favorable Regulatory Landscape (in certain regions): In regions with established regulatory pathways for biological products, companies are more likely to invest in and bring amniotic fluid-derived products to market.
- Expansion of Applications: Ongoing research is uncovering new therapeutic applications for amniotic fluid in areas such as autoimmune diseases, neurodegenerative disorders, and ocular surface reconstruction. The market size is projected to grow by millions annually.
Challenges in the Human Amniotic Fluid Sector
Despite its promising growth, the Human Amniotic Fluid sector faces several significant challenges that could impede its full potential.
- Regulatory Hurdles: Navigating the complex and varied regulatory landscapes across different countries for biological products can be time-consuming and expensive, potentially delaying market entry. The classification of amniotic fluid products as tissue, drug, or medical device can vary, creating ambiguity.
- Ethical and Sourcing Concerns: Ensuring ethical sourcing of amniotic fluid and maintaining donor consent protocols are paramount and can be a complex operational challenge. Public perception and ethical debates surrounding the use of human biological materials can also influence market acceptance.
- Supply Chain Management: Maintaining a consistent and reliable supply of high-quality amniotic fluid can be challenging due to donor variability and the need for stringent quality control measures.
- Competition from Alternative Therapies: The market faces competition from other regenerative medicine sources, such as mesenchymal stem cells from bone marrow or adipose tissue, as well as synthetic biomaterials, which may offer different cost-benefit profiles or perceived advantages.
- Cost of Development and Treatment: The research, development, and clinical trial costs associated with amniotic fluid-derived therapies can be substantial, leading to high treatment costs for patients and potential reimbursement challenges. The market size is expected to be impacted by these challenges.
Leading Players in the Human Amniotic Fluid Market
The competitive landscape of the Human Amniotic Fluid market is dynamic, featuring a mix of established biotechnology firms and innovative startups. Key players driving market growth and innovation include:
- Amsbio LLC
- Celprogen Inc
- Integrity PRP
- Biological Industries (Sartorius)
Key Developments in Human Amniotic Fluid Sector
The Human Amniotic Fluid sector has witnessed significant developments that have shaped its current and future trajectory. These advancements highlight the growing importance and expanding applications of amniotic fluid in regenerative medicine.
- 2023: Launch of novel amniotic membrane-based wound dressings offering enhanced healing properties.
- 2022: Successful completion of clinical trials for an amniotic fluid-derived therapeutic for osteoarthritis, demonstrating significant pain reduction and joint function improvement.
- 2021: Introduction of advanced exosome isolation techniques from amniotic fluid, enabling the development of highly potent regenerative therapies.
- 2020: Expansion of manufacturing capabilities by key players to meet the growing demand for amniotic fluid-derived products.
- 2019: Increased investment in research and development of amniotic fluid for potential applications in neurological disorders.
Strategic Human Amniotic Fluid Market Outlook
The strategic outlook for the Human Amniotic Fluid market is exceptionally positive, driven by the persistent demand for regenerative therapies and ongoing scientific breakthroughs. Growth accelerators include the expanding clinical evidence supporting the efficacy of amniotic fluid in various medical applications, from chronic wound management to tissue repair. The increasing focus on personalized medicine and minimally invasive treatments further positions amniotic fluid-derived products as highly desirable therapeutic options. Strategic opportunities lie in forging partnerships between research institutions and commercial entities to expedite the translation of novel discoveries into clinical applications, as well as in developing cost-effective production methods to improve market accessibility. The projected market value for the forecast period indicates substantial growth, estimated in the millions. Continuous innovation in product formulation and delivery systems will be crucial for capturing market share and addressing unmet medical needs. The market size is expected to grow significantly.
Human Amniotic Fluid Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Specialty Clinic
- 1.3. Diagnostics Laboratory
- 1.4. Others
-
2. Types
- 2.1. Pure
- 2.2. Mixtures
Human Amniotic Fluid Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
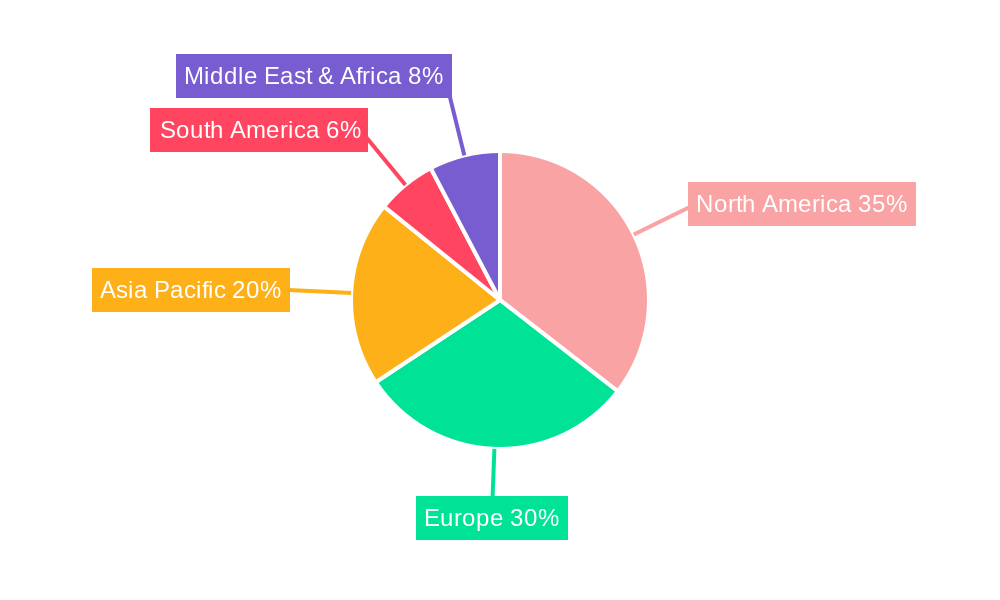
Human Amniotic Fluid Regional Market Share

Geographic Coverage of Human Amniotic Fluid
Human Amniotic Fluid REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Specialty Clinic
- 5.1.3. Diagnostics Laboratory
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Pure
- 5.2.2. Mixtures
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Human Amniotic Fluid Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Specialty Clinic
- 6.1.3. Diagnostics Laboratory
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Pure
- 6.2.2. Mixtures
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Human Amniotic Fluid Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Specialty Clinic
- 7.1.3. Diagnostics Laboratory
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Pure
- 7.2.2. Mixtures
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Human Amniotic Fluid Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Specialty Clinic
- 8.1.3. Diagnostics Laboratory
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Pure
- 8.2.2. Mixtures
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Human Amniotic Fluid Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Specialty Clinic
- 9.1.3. Diagnostics Laboratory
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Pure
- 9.2.2. Mixtures
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Human Amniotic Fluid Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Specialty Clinic
- 10.1.3. Diagnostics Laboratory
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Pure
- 10.2.2. Mixtures
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Human Amniotic Fluid Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospital
- 11.1.2. Specialty Clinic
- 11.1.3. Diagnostics Laboratory
- 11.1.4. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Pure
- 11.2.2. Mixtures
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Amsbio LLC
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Celprogen Inc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Integrity PRP
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Biological Industries(Sartorius)
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.1 Amsbio LLC
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Human Amniotic Fluid Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Human Amniotic Fluid Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Human Amniotic Fluid Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Human Amniotic Fluid Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Human Amniotic Fluid Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Human Amniotic Fluid Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Human Amniotic Fluid Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Human Amniotic Fluid Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Human Amniotic Fluid Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Human Amniotic Fluid Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Human Amniotic Fluid Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Human Amniotic Fluid Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Human Amniotic Fluid Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Human Amniotic Fluid Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Human Amniotic Fluid Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Human Amniotic Fluid Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Human Amniotic Fluid Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Human Amniotic Fluid Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Human Amniotic Fluid Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Human Amniotic Fluid Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Human Amniotic Fluid Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Human Amniotic Fluid Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Human Amniotic Fluid Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Human Amniotic Fluid Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Human Amniotic Fluid Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Human Amniotic Fluid Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Human Amniotic Fluid Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Human Amniotic Fluid Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Human Amniotic Fluid Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Human Amniotic Fluid Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Human Amniotic Fluid Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Human Amniotic Fluid Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Human Amniotic Fluid Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Human Amniotic Fluid Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Human Amniotic Fluid Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Human Amniotic Fluid Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Human Amniotic Fluid Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Human Amniotic Fluid Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Human Amniotic Fluid Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Human Amniotic Fluid Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Human Amniotic Fluid Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Human Amniotic Fluid Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Human Amniotic Fluid Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Human Amniotic Fluid Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Human Amniotic Fluid Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Human Amniotic Fluid Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Human Amniotic Fluid Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Human Amniotic Fluid Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Human Amniotic Fluid Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Human Amniotic Fluid Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Human Amniotic Fluid?
The projected CAGR is approximately 7.3%.
2. Which companies are prominent players in the Human Amniotic Fluid?
Key companies in the market include Amsbio LLC, Celprogen Inc, Integrity PRP, Biological Industries(Sartorius).
3. What are the main segments of the Human Amniotic Fluid?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Human Amniotic Fluid," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Human Amniotic Fluid report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Human Amniotic Fluid?
To stay informed about further developments, trends, and reports in the Human Amniotic Fluid, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
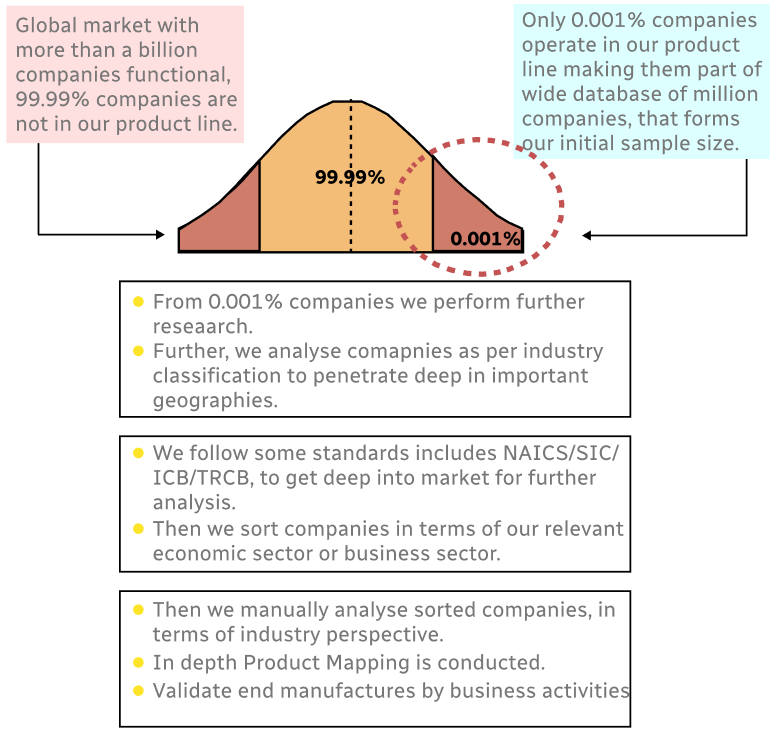
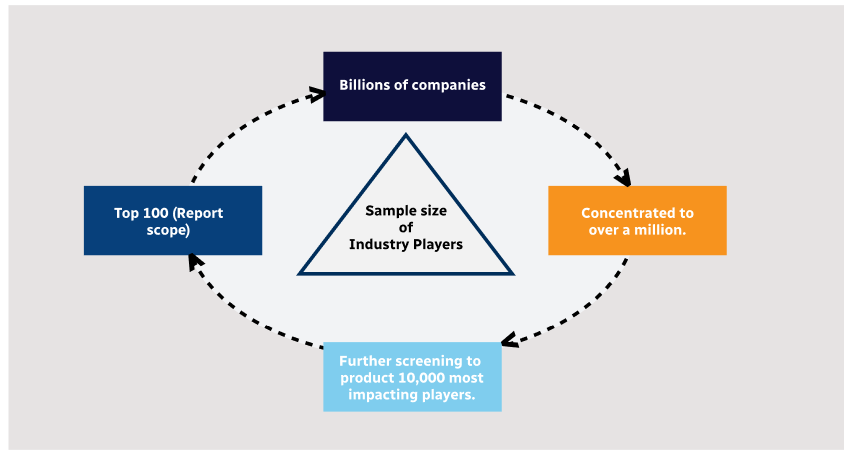
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
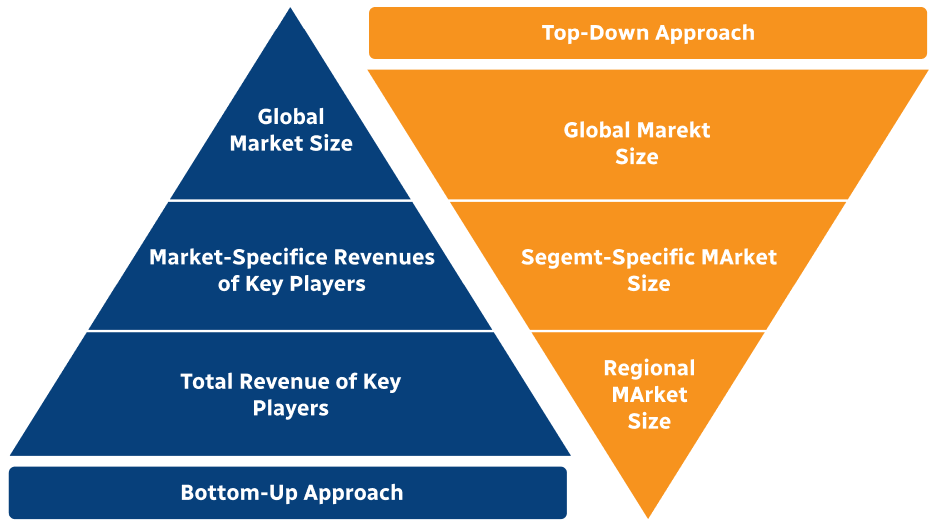
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
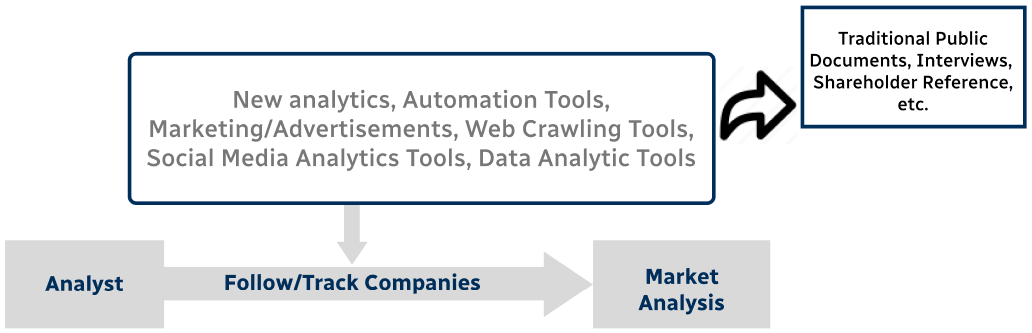
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


