Key Insights
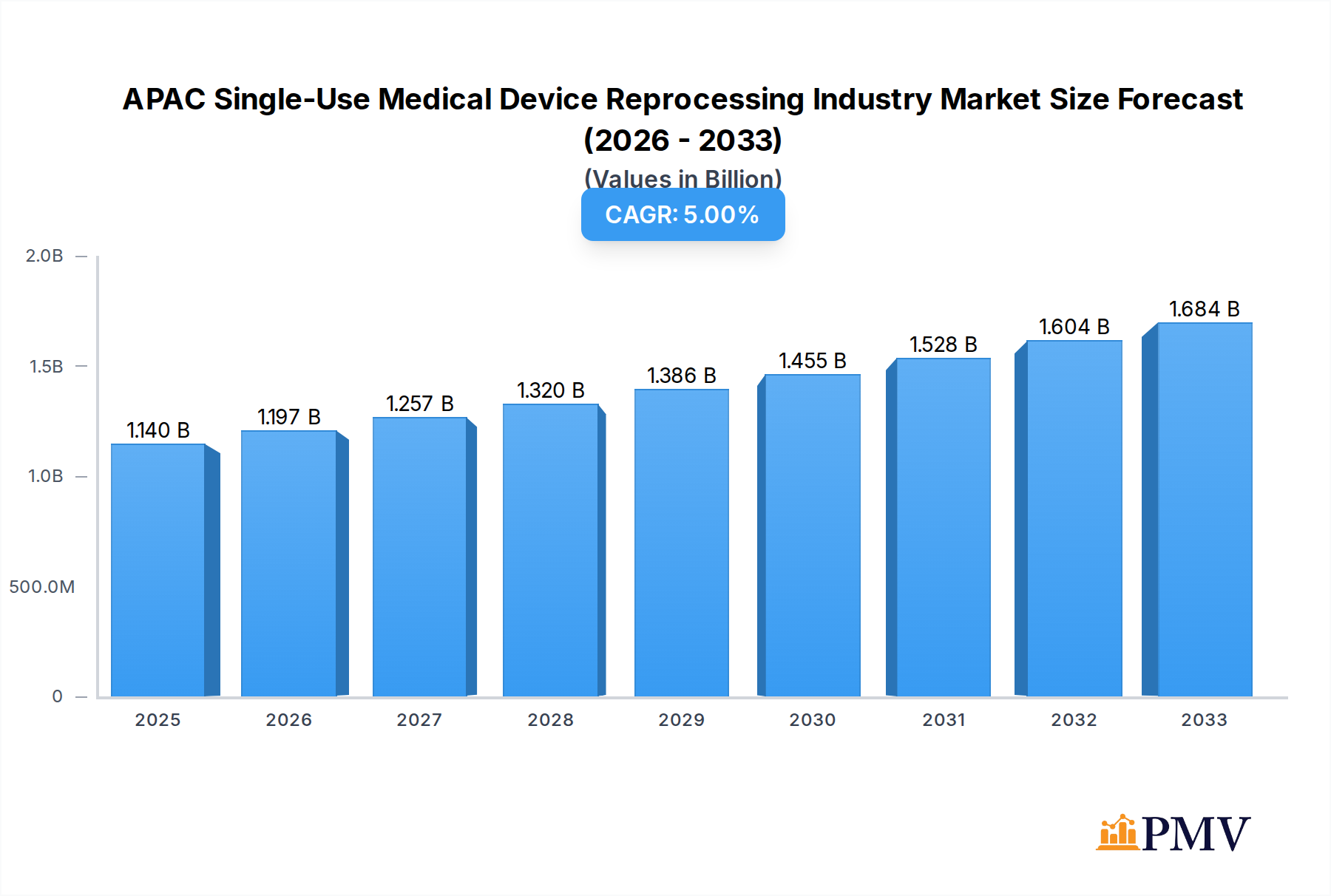
The APAC Single-Use Medical Device Reprocessing Industry is poised for substantial growth, with an estimated market size of USD 1.14 billion in 2025 and a projected Compound Annual Growth Rate (CAGR) of 5% through 2033. This expansion is primarily driven by increasing healthcare expenditure across the Asia-Pacific region, a growing awareness of cost-saving measures in healthcare systems, and the rising adoption of advanced reprocessing technologies. The critical need to manage escalating medical waste and the imperative for sustainable healthcare practices further bolster the market's upward trajectory. As healthcare infrastructure develops and patient populations grow, the demand for efficient and safe reprocessing of single-use medical devices will intensify, creating significant opportunities for market players.
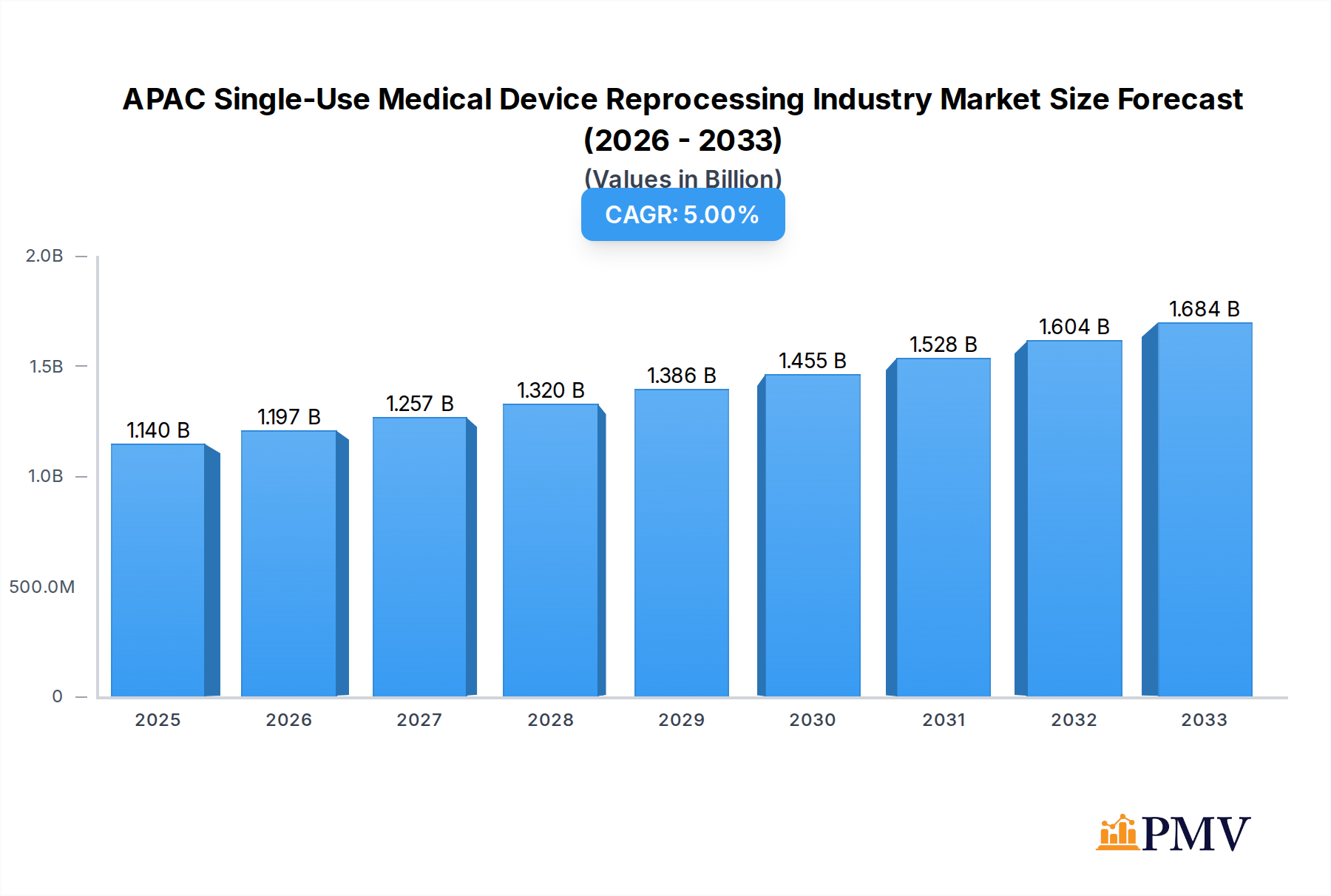
APAC Single-Use Medical Device Reprocessing Industry Market Size (In Billion)

The market is segmented into Class I and Class II devices, with a notable focus on reprocessing items like laparoscopic graspers, forceps, scissors, pulse oximeter sensors, sequential compression sleeves, and catheters. Geographically, China and India are expected to be the dominant markets, fueled by their vast populations and expanding healthcare sectors. Japan and South Korea, with their technologically advanced healthcare systems, also present significant growth potential, alongside Australia and other emerging economies in the Rest of Asia-Pacific. While the market benefits from strong demand, potential restraints include stringent regulatory frameworks, the need for robust quality control protocols to ensure patient safety, and consumer perception regarding reprocessed devices. However, as standards are refined and the benefits of reprocessing become more evident, these restraints are likely to be mitigated, paving the way for sustained and robust market expansion.
APAC Single-Use Medical Device Reprocessing Industry Company Market Share

This in-depth report offers a definitive analysis of the APAC Single-Use Medical Device Reprocessing Industry, exploring market dynamics, key trends, dominant segments, product innovations, and strategic outlook from 2019 to 2033. With a base year of 2025 and a forecast period extending to 2033, this report provides critical insights for stakeholders in the medical device reprocessing, healthcare sustainability, and infection prevention sectors. Discover market sizes estimated in billions, CAGR projections, and the impact of emerging technologies and regulations on this rapidly evolving market. We meticulously cover Class I Devices (Laparoscopic Graspers, Forceps, Scissors, Other Class I Devices) and Class II Devices (Pulse Oximeter Sensors, Sequential Compression Sleeves, Catheters and Guidewires, Other Class II Devices) across key geographies including China, Japan, India, Australia, South Korea, and the Rest of Asia-Pacific.
APAC Single-Use Medical Device Reprocessing Industry Market Structure & Competitive Dynamics
The APAC Single-Use Medical Device Reprocessing Industry is characterized by a dynamic competitive landscape, with market concentration varying across different device types and geographic regions. While some established players hold significant market share, the growing demand for cost-effective and sustainable healthcare solutions is fostering innovation and attracting new entrants. The innovation ecosystem is driven by advancements in sterilization technologies, material science, and digital tracking systems, all aimed at ensuring patient safety and regulatory compliance. Regulatory frameworks in the APAC region are progressively evolving, with an increasing emphasis on standardization and quality control for reprocessed medical devices. Product substitutes, primarily new single-use devices, continue to present competition, but the economic benefits and environmental advantages of reprocessing are gaining traction. End-user trends, particularly in hospitals and healthcare facilities, are leaning towards adopting reprocessing programs to manage costs and reduce medical waste, contributing to market growth. Mergers and Acquisitions (M&A) activities are a significant aspect of market consolidation, with notable deal values contributing to the expansion of leading companies. For instance, the acquisition of ReNu Medical Inc. by Arjo Inc. signifies a strategic move to strengthen their reprocessing capabilities. Market share data indicates a steady rise in the reprocessing of both Class I and Class II medical devices, driven by their demonstrable cost savings, estimated to reach over $XX billion by 2033.
- Market Concentration: Moderate to high, with key players dominating specific segments.
- Innovation Ecosystem: Driven by technological advancements in sterilization, traceability, and material science.
- Regulatory Frameworks: Evolving to ensure safety, efficacy, and standardization of reprocessed devices.
- Product Substitutes: New single-use devices remain a key competitor.
- End-User Trends: Growing adoption for cost reduction and waste management.
- M&A Activities: Strategic acquisitions and partnerships shaping the competitive landscape.
- Estimated Market Share Growth for Reprocessed Devices: Anticipated to exceed XX% by 2033.
APAC Single-Use Medical Device Reprocessing Industry Industry Trends & Insights
The APAC Single-Use Medical Device Reprocessing Industry is experiencing robust growth, propelled by a confluence of factors including increasing healthcare expenditure, a growing patient population, and a rising awareness of the economic and environmental benefits associated with medical device reprocessing. The Compound Annual Growth Rate (CAGR) for this sector is projected to be approximately XX% over the forecast period (2025-2033), indicating significant expansion. Technological disruptions are at the forefront, with advancements in automated cleaning, advanced sterilization techniques like low-temperature plasma sterilization, and sophisticated tracking and traceability systems enhancing the efficiency and safety of the reprocessing workflow. These innovations are crucial for ensuring that reprocessed devices meet stringent quality standards and maintain their intended functionality. Consumer preferences are shifting, with healthcare providers actively seeking sustainable and cost-effective alternatives to traditional single-use device disposal. This shift is further amplified by government initiatives aimed at promoting circular economy principles and reducing healthcare waste. The competitive dynamics are intensifying, with established medical device manufacturers and specialized reprocessing companies vying for market share. Companies are investing heavily in R&D to develop new reprocessing protocols for a wider range of devices and to enhance the reliability of their services. Market penetration of reprocessing services is steadily increasing, particularly in developed markets within APAC, as hospitals recognize the substantial cost savings, often exceeding XX% on certain device categories, and the positive environmental impact. The market size is projected to reach over $XX billion by the end of the forecast period, driven by the continued adoption of reprocessing across various medical specialties. The rising demand for minimally invasive surgical procedures also contributes to the increased volume of single-use devices available for reprocessing.
Dominant Markets & Segments in APAC Single-Use Medical Device Reprocessing Industry
The APAC Single-Use Medical Device Reprocessing Industry showcases distinct dominance across its various segments, with China and Japan leading the market due to their advanced healthcare infrastructure, high healthcare spending, and established regulatory frameworks for medical device reprocessing. The Rest of Asia-Pacific, encompassing countries like Southeast Asian nations, is emerging as a significant growth frontier, driven by rapid economic development and increasing investments in healthcare.
Within the Device Type segmentation, Class II Devices are poised for greater market penetration in reprocessing compared to Class I Devices. This is primarily due to the higher cost of Class II devices, such as Pulse Oximeter Sensors, Sequential Compression Sleeves, and Catheters and Guidewires, making their reprocessing a more economically attractive option for healthcare facilities. The demand for reprocessing these devices is amplified by their widespread use in critical care and post-operative recovery.
Geographic Dominance:
- China: Driven by a large patient population, increasing healthcare investments, and a growing number of hospitals adopting reprocessing programs. The market size is estimated to reach over $XX billion by 2033.
- Japan: Characterized by a well-established healthcare system, stringent quality control, and early adoption of reprocessing technologies, contributing to its significant market share.
- India: Experiencing rapid growth due to a burgeoning medical tourism sector, increasing demand for affordable healthcare, and supportive government policies aimed at waste reduction.
- Australia & South Korea: Mature markets with strong regulatory oversight, focusing on advanced reprocessing techniques and high-value device reprocessing.
- Rest of Asia-Pacific: Presents substantial untapped potential, with a rising middle class and expanding healthcare access fueling demand for efficient and cost-effective medical solutions.
Device Type Dominance:
- Class II Devices:
- Pulse Oximeter Sensors: High volume usage and significant cost savings through reprocessing make them a key segment.
- Sequential Compression Sleeves: Essential for preventing deep vein thrombosis, their frequent use drives reprocessing demand.
- Catheters and Guidewires: Critical in interventional cardiology and radiology, reprocessing offers substantial economic benefits and reduces waste.
- Other Class II Devices: A broad category with increasing reprocessing potential as more devices become eligible.
- Class I Devices:
- Laparoscopic Graspers, Forceps, and Scissors: While often reprocessed, the lower individual cost might influence adoption rates compared to Class II devices. However, the high volume of these surgical instruments still makes them a considerable segment.
- Other Class I Devices: Opportunities exist for reprocessing specific instruments with robust material compositions.
- Class II Devices:
The increasing adoption of reprocessing for Catheters and Guidewires is a significant trend, driven by their complex design and the substantial cost differential between new and reprocessed units, often exceeding XX% savings. The market penetration of reprocessed Class II devices is projected to grow by XX% by 2033, solidifying their dominance.
APAC Single-Use Medical Device Reprocessing Industry Product Innovations
Product innovations in the APAC Single-Use Medical Device Reprocessing Industry are primarily focused on enhancing the safety, efficacy, and traceability of reprocessed devices. Advanced cleaning and sterilization technologies, such as peracetic acid reprocessing and low-temperature plasma sterilization, are gaining traction, enabling the reprocessing of a wider range of complex instruments, including those with lumens and intricate mechanisms. Developments in tracking and tracing systems, often utilizing RFID or barcode technology, are crucial for maintaining a detailed lifecycle record of each reprocessed device, ensuring accountability and compliance with regulatory requirements. Furthermore, innovations in material science are addressing concerns about device integrity post-reprocessing, ensuring that reprocessed devices maintain their original performance characteristics and patient safety profiles. The market fit for these innovations is strong, driven by healthcare providers' increasing demand for reliable, cost-effective, and sustainable reprocessing solutions.
Report Segmentation & Scope
This report segments the APAC Single-Use Medical Device Reprocessing Industry comprehensively, providing granular analysis of market dynamics, growth projections, and competitive landscapes. The segmentation covers:
Device Type:
- Class I Devices: This segment includes Laparoscopic Graspers, Forceps, Scissors, and Other Class I Devices. Growth projections indicate a steady increase in reprocessing volume, driven by cost-effectiveness and waste reduction initiatives.
- Class II Devices: This segment encompasses Pulse Oximeter Sensors, Sequential Compression Sleeves, Catheters and Guidewires, and Other Class II Devices. This segment is expected to witness the highest growth due to the higher cost of these devices, making reprocessing a more significant economic advantage. Market sizes for Class II device reprocessing are projected to reach over $XX billion by 2033.
Geography:
- China: Expected to be the largest market, with significant growth driven by increasing healthcare infrastructure and adoption of reprocessing.
- Japan: A mature market with high standards and a strong focus on advanced reprocessing techniques.
- India: Demonstrating rapid expansion due to growing healthcare demand and cost-consciousness.
- Australia: A developed market with robust regulatory frameworks, focusing on quality and safety.
- South Korea: Exhibits a similar growth trajectory to Australia, with technological advancements in reprocessing.
- Rest of Asia-Pacific: This segment represents substantial untapped potential, with emerging economies poised for significant healthcare sector growth, thereby driving demand for reprocessing services.
Key Drivers of APAC Single-Use Medical Device Reprocessing Industry Growth
The APAC Single-Use Medical Device Reprocessing Industry is propelled by several key drivers. Economically, the significant cost savings achieved through reprocessing, often estimated at XX% for eligible devices, directly addresses the increasing pressure on healthcare budgets. Technologically, advancements in sterilization methods (e.g., peracetic acid, low-temperature plasma) and automated cleaning systems are expanding the range of reprocessable devices and enhancing reprocessing efficiency and safety. Regulatory bodies are increasingly focusing on sustainability and waste reduction, creating a favorable environment for reprocessing programs. Furthermore, a growing awareness among healthcare professionals and institutions about the environmental benefits of diverting medical waste from landfills is a crucial driver. The expanding healthcare infrastructure across the APAC region, leading to increased utilization of medical devices, indirectly fuels the demand for reprocessing services.
Challenges in the APAC Single-Use Medical Device Reprocessing Industry Sector
Despite the promising growth, the APAC Single-Use Medical Device Reprocessing Industry faces several challenges. Regulatory hurdles remain a significant barrier, as varying standards and approval processes across different APAC countries can complicate market entry and expansion. Supply chain complexities for collecting used devices and distributing reprocessed ones across vast geographies can also impact efficiency and cost-effectiveness. Public perception and potential concerns regarding the safety and efficacy of reprocessed devices, though often unfounded with proper protocols, can still influence adoption rates. Fierce competition from manufacturers of new single-use devices, who actively promote their products, also poses a continuous challenge. Furthermore, the initial investment required for establishing robust reprocessing facilities and training skilled personnel can be substantial, particularly for smaller healthcare providers.
Leading Players in the APAC Single-Use Medical Device Reprocessing Industry Market
- Sterilmed Inc (Johnson & Johnson)
- Soma Technology Inc
- ReNu Medical Inc (Arjo Inc)
- Stryker Corporation
- NEScientific Inc
- Medline Industries Inc
Key Developments in APAC Single-Use Medical Device Reprocessing Industry Sector
- August 2022: Ecolab Inc. expanded its Healthcare division to serve hospitals and healthcare facilities in Southeast Asia. A leader in infection prevention solutions and services, Ecolab Healthcare, offers a comprehensive array of hygiene and infection prevention solutions for hospitals, surgical centers, and healthcare facilities. They also provide instrument & endoscopy reprocessing, surgical drapes, and environmental hand hygiene, enhancing the available reprocessing support infrastructure in the region.
- April 2022: The 'state-of-the-art' Central Sterile Services Department (CSSD) of AIIMS Bhubaneswar received 2nd prize under the Government Health Care Organization category in the Consortium of Accredited Health Care Organizations Awareness Compliances and Excellence (CAHO-ACE) program for the year 2021. This recognition highlights the growing emphasis on best practices and excellence in sterile processing within the region, indirectly supporting the reprocessing industry by promoting high standards.
Strategic APAC Single-Use Medical Device Reprocessing Industry Market Outlook
The strategic outlook for the APAC Single-Use Medical Device Reprocessing Industry is highly positive, driven by sustained demand for cost-effective healthcare solutions and an increasing commitment to environmental sustainability. Growth accelerators include the ongoing expansion of healthcare infrastructure across emerging economies in the APAC region, leading to higher device utilization. Furthermore, advancements in reprocessing technologies, particularly those that broaden the range of eligible devices and enhance process efficiency, will be critical. Strategic opportunities lie in developing robust partnerships with healthcare providers, investing in advanced traceability systems to ensure complete regulatory compliance, and focusing on educating stakeholders about the safety and economic benefits of reprocessing. The market is expected to see continued consolidation through M&A activities as larger players seek to expand their service offerings and geographic reach. The shift towards a circular economy model in healthcare will further solidify the long-term growth trajectory of this industry.
APAC Single-Use Medical Device Reprocessing Industry Segmentation
-
1. Device Type
-
1.1. Class I Devices
- 1.1.1. Laparoscopic Graspers
- 1.1.2. Forceps
- 1.1.3. Scissors
- 1.1.4. Other Class I Devices
-
1.2. Class II Device
- 1.2.1. Pulse Oximeter Sensors
- 1.2.2. Sequential Compression Sleeves
- 1.2.3. Catheters and Guidewires
- 1.2.4. Other Class II Devices
-
1.1. Class I Devices
-
2. Geography
- 2.1. China
- 2.2. Japan
- 2.3. India
- 2.4. Australia
- 2.5. South Korea
- 2.6. Rest of Asia-Pacific
APAC Single-Use Medical Device Reprocessing Industry Segmentation By Geography
- 1. China
- 2. Japan
- 3. India
- 4. Australia
- 5. South Korea
- 6. Rest of Asia Pacific
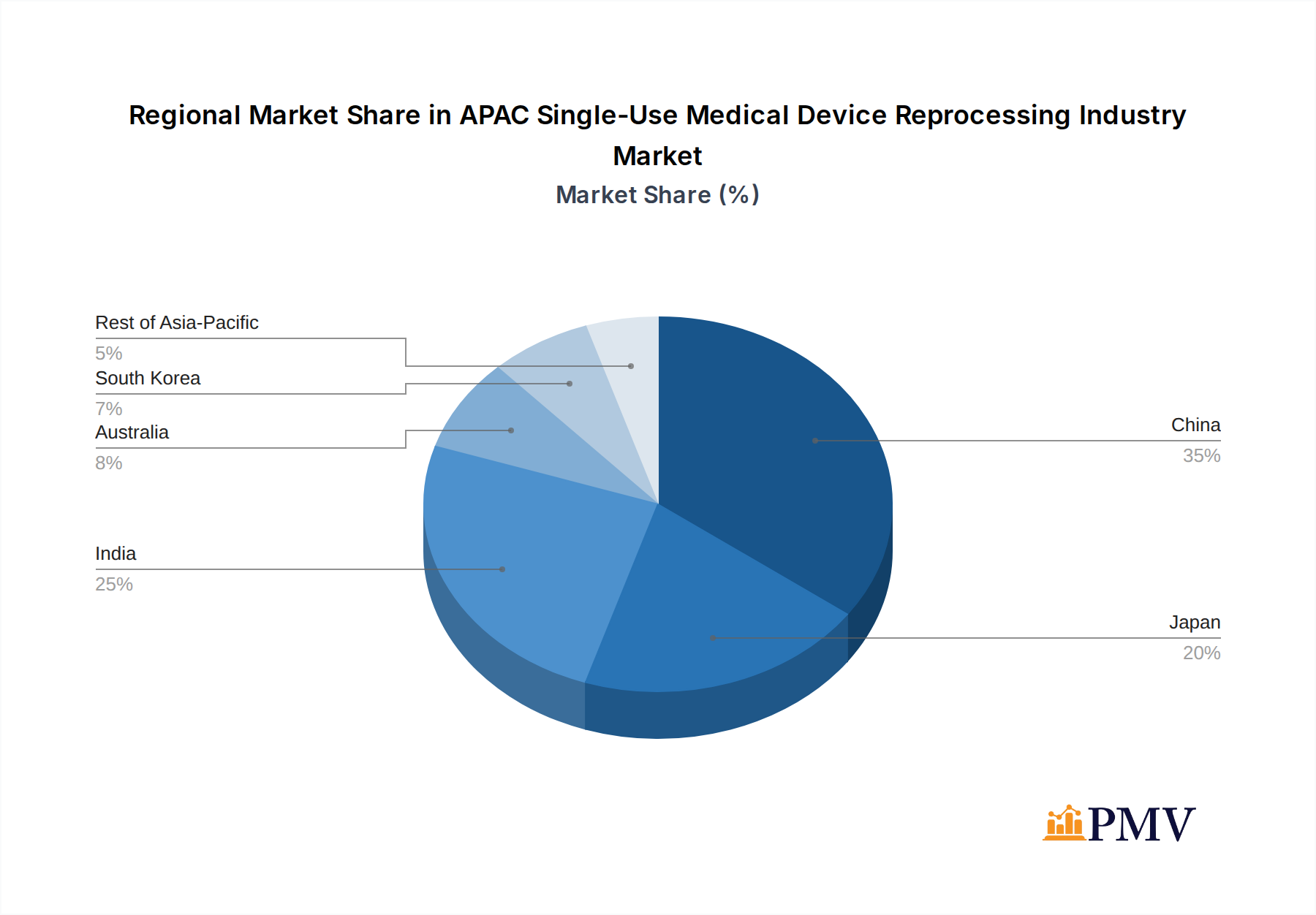
APAC Single-Use Medical Device Reprocessing Industry Regional Market Share

Geographic Coverage of APAC Single-Use Medical Device Reprocessing Industry
APAC Single-Use Medical Device Reprocessing Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 5.1.1. Class I Devices
- 5.1.1.1. Laparoscopic Graspers
- 5.1.1.2. Forceps
- 5.1.1.3. Scissors
- 5.1.1.4. Other Class I Devices
- 5.1.2. Class II Device
- 5.1.2.1. Pulse Oximeter Sensors
- 5.1.2.2. Sequential Compression Sleeves
- 5.1.2.3. Catheters and Guidewires
- 5.1.2.4. Other Class II Devices
- 5.1.1. Class I Devices
- 5.2. Market Analysis, Insights and Forecast - by Geography
- 5.2.1. China
- 5.2.2. Japan
- 5.2.3. India
- 5.2.4. Australia
- 5.2.5. South Korea
- 5.2.6. Rest of Asia-Pacific
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. China
- 5.3.2. Japan
- 5.3.3. India
- 5.3.4. Australia
- 5.3.5. South Korea
- 5.3.6. Rest of Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 6. Global APAC Single-Use Medical Device Reprocessing Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 6.1.1. Class I Devices
- 6.1.1.1. Laparoscopic Graspers
- 6.1.1.2. Forceps
- 6.1.1.3. Scissors
- 6.1.1.4. Other Class I Devices
- 6.1.2. Class II Device
- 6.1.2.1. Pulse Oximeter Sensors
- 6.1.2.2. Sequential Compression Sleeves
- 6.1.2.3. Catheters and Guidewires
- 6.1.2.4. Other Class II Devices
- 6.1.1. Class I Devices
- 6.2. Market Analysis, Insights and Forecast - by Geography
- 6.2.1. China
- 6.2.2. Japan
- 6.2.3. India
- 6.2.4. Australia
- 6.2.5. South Korea
- 6.2.6. Rest of Asia-Pacific
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 7. China APAC Single-Use Medical Device Reprocessing Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Device Type
- 7.1.1. Class I Devices
- 7.1.1.1. Laparoscopic Graspers
- 7.1.1.2. Forceps
- 7.1.1.3. Scissors
- 7.1.1.4. Other Class I Devices
- 7.1.2. Class II Device
- 7.1.2.1. Pulse Oximeter Sensors
- 7.1.2.2. Sequential Compression Sleeves
- 7.1.2.3. Catheters and Guidewires
- 7.1.2.4. Other Class II Devices
- 7.1.1. Class I Devices
- 7.2. Market Analysis, Insights and Forecast - by Geography
- 7.2.1. China
- 7.2.2. Japan
- 7.2.3. India
- 7.2.4. Australia
- 7.2.5. South Korea
- 7.2.6. Rest of Asia-Pacific
- 7.1. Market Analysis, Insights and Forecast - by Device Type
- 8. Japan APAC Single-Use Medical Device Reprocessing Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Device Type
- 8.1.1. Class I Devices
- 8.1.1.1. Laparoscopic Graspers
- 8.1.1.2. Forceps
- 8.1.1.3. Scissors
- 8.1.1.4. Other Class I Devices
- 8.1.2. Class II Device
- 8.1.2.1. Pulse Oximeter Sensors
- 8.1.2.2. Sequential Compression Sleeves
- 8.1.2.3. Catheters and Guidewires
- 8.1.2.4. Other Class II Devices
- 8.1.1. Class I Devices
- 8.2. Market Analysis, Insights and Forecast - by Geography
- 8.2.1. China
- 8.2.2. Japan
- 8.2.3. India
- 8.2.4. Australia
- 8.2.5. South Korea
- 8.2.6. Rest of Asia-Pacific
- 8.1. Market Analysis, Insights and Forecast - by Device Type
- 9. India APAC Single-Use Medical Device Reprocessing Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Device Type
- 9.1.1. Class I Devices
- 9.1.1.1. Laparoscopic Graspers
- 9.1.1.2. Forceps
- 9.1.1.3. Scissors
- 9.1.1.4. Other Class I Devices
- 9.1.2. Class II Device
- 9.1.2.1. Pulse Oximeter Sensors
- 9.1.2.2. Sequential Compression Sleeves
- 9.1.2.3. Catheters and Guidewires
- 9.1.2.4. Other Class II Devices
- 9.1.1. Class I Devices
- 9.2. Market Analysis, Insights and Forecast - by Geography
- 9.2.1. China
- 9.2.2. Japan
- 9.2.3. India
- 9.2.4. Australia
- 9.2.5. South Korea
- 9.2.6. Rest of Asia-Pacific
- 9.1. Market Analysis, Insights and Forecast - by Device Type
- 10. Australia APAC Single-Use Medical Device Reprocessing Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Device Type
- 10.1.1. Class I Devices
- 10.1.1.1. Laparoscopic Graspers
- 10.1.1.2. Forceps
- 10.1.1.3. Scissors
- 10.1.1.4. Other Class I Devices
- 10.1.2. Class II Device
- 10.1.2.1. Pulse Oximeter Sensors
- 10.1.2.2. Sequential Compression Sleeves
- 10.1.2.3. Catheters and Guidewires
- 10.1.2.4. Other Class II Devices
- 10.1.1. Class I Devices
- 10.2. Market Analysis, Insights and Forecast - by Geography
- 10.2.1. China
- 10.2.2. Japan
- 10.2.3. India
- 10.2.4. Australia
- 10.2.5. South Korea
- 10.2.6. Rest of Asia-Pacific
- 10.1. Market Analysis, Insights and Forecast - by Device Type
- 11. South Korea APAC Single-Use Medical Device Reprocessing Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Device Type
- 11.1.1. Class I Devices
- 11.1.1.1. Laparoscopic Graspers
- 11.1.1.2. Forceps
- 11.1.1.3. Scissors
- 11.1.1.4. Other Class I Devices
- 11.1.2. Class II Device
- 11.1.2.1. Pulse Oximeter Sensors
- 11.1.2.2. Sequential Compression Sleeves
- 11.1.2.3. Catheters and Guidewires
- 11.1.2.4. Other Class II Devices
- 11.1.1. Class I Devices
- 11.2. Market Analysis, Insights and Forecast - by Geography
- 11.2.1. China
- 11.2.2. Japan
- 11.2.3. India
- 11.2.4. Australia
- 11.2.5. South Korea
- 11.2.6. Rest of Asia-Pacific
- 11.1. Market Analysis, Insights and Forecast - by Device Type
- 12. Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Device Type
- 12.1.1. Class I Devices
- 12.1.1.1. Laparoscopic Graspers
- 12.1.1.2. Forceps
- 12.1.1.3. Scissors
- 12.1.1.4. Other Class I Devices
- 12.1.2. Class II Device
- 12.1.2.1. Pulse Oximeter Sensors
- 12.1.2.2. Sequential Compression Sleeves
- 12.1.2.3. Catheters and Guidewires
- 12.1.2.4. Other Class II Devices
- 12.1.1. Class I Devices
- 12.2. Market Analysis, Insights and Forecast - by Geography
- 12.2.1. China
- 12.2.2. Japan
- 12.2.3. India
- 12.2.4. Australia
- 12.2.5. South Korea
- 12.2.6. Rest of Asia-Pacific
- 12.1. Market Analysis, Insights and Forecast - by Device Type
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Sterilmed Inc (Johnson & Johnson)
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Soma Technology Inc
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 ReNu Medical Inc (Arjo Inc )
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Stryker Corporation
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 NEScientific Inc
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 Medline Industries Inc
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.1 Sterilmed Inc (Johnson & Johnson)
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Global APAC Single-Use Medical Device Reprocessing Industry Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global APAC Single-Use Medical Device Reprocessing Industry Volume Breakdown (K Unit, %) by Region 2025 & 2033
- Figure 3: China APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Device Type 2025 & 2033
- Figure 4: China APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Device Type 2025 & 2033
- Figure 5: China APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Device Type 2025 & 2033
- Figure 6: China APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Device Type 2025 & 2033
- Figure 7: China APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Geography 2025 & 2033
- Figure 8: China APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Geography 2025 & 2033
- Figure 9: China APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Geography 2025 & 2033
- Figure 10: China APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Geography 2025 & 2033
- Figure 11: China APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 12: China APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 13: China APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: China APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Country 2025 & 2033
- Figure 15: Japan APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Device Type 2025 & 2033
- Figure 16: Japan APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Device Type 2025 & 2033
- Figure 17: Japan APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Device Type 2025 & 2033
- Figure 18: Japan APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Device Type 2025 & 2033
- Figure 19: Japan APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Geography 2025 & 2033
- Figure 20: Japan APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Geography 2025 & 2033
- Figure 21: Japan APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Geography 2025 & 2033
- Figure 22: Japan APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Geography 2025 & 2033
- Figure 23: Japan APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 24: Japan APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 25: Japan APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Japan APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Country 2025 & 2033
- Figure 27: India APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Device Type 2025 & 2033
- Figure 28: India APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Device Type 2025 & 2033
- Figure 29: India APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Device Type 2025 & 2033
- Figure 30: India APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Device Type 2025 & 2033
- Figure 31: India APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Geography 2025 & 2033
- Figure 32: India APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Geography 2025 & 2033
- Figure 33: India APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Geography 2025 & 2033
- Figure 34: India APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Geography 2025 & 2033
- Figure 35: India APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 36: India APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 37: India APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 38: India APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Country 2025 & 2033
- Figure 39: Australia APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Device Type 2025 & 2033
- Figure 40: Australia APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Device Type 2025 & 2033
- Figure 41: Australia APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Device Type 2025 & 2033
- Figure 42: Australia APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Device Type 2025 & 2033
- Figure 43: Australia APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Geography 2025 & 2033
- Figure 44: Australia APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Geography 2025 & 2033
- Figure 45: Australia APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Geography 2025 & 2033
- Figure 46: Australia APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Geography 2025 & 2033
- Figure 47: Australia APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 48: Australia APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 49: Australia APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 50: Australia APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Country 2025 & 2033
- Figure 51: South Korea APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Device Type 2025 & 2033
- Figure 52: South Korea APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Device Type 2025 & 2033
- Figure 53: South Korea APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Device Type 2025 & 2033
- Figure 54: South Korea APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Device Type 2025 & 2033
- Figure 55: South Korea APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Geography 2025 & 2033
- Figure 56: South Korea APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Geography 2025 & 2033
- Figure 57: South Korea APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Geography 2025 & 2033
- Figure 58: South Korea APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Geography 2025 & 2033
- Figure 59: South Korea APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 60: South Korea APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 61: South Korea APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 62: South Korea APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Country 2025 & 2033
- Figure 63: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Device Type 2025 & 2033
- Figure 64: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Device Type 2025 & 2033
- Figure 65: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Device Type 2025 & 2033
- Figure 66: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Device Type 2025 & 2033
- Figure 67: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Geography 2025 & 2033
- Figure 68: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Geography 2025 & 2033
- Figure 69: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Geography 2025 & 2033
- Figure 70: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Geography 2025 & 2033
- Figure 71: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 72: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Volume (K Unit), by Country 2025 & 2033
- Figure 73: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 74: Rest of Asia Pacific APAC Single-Use Medical Device Reprocessing Industry Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Device Type 2020 & 2033
- Table 2: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 3: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 4: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 5: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Region 2020 & 2033
- Table 7: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Device Type 2020 & 2033
- Table 8: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 9: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 10: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 11: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 13: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Device Type 2020 & 2033
- Table 14: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 15: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 16: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 17: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 18: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 19: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Device Type 2020 & 2033
- Table 20: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 21: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 22: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 23: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Device Type 2020 & 2033
- Table 26: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 27: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 28: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 29: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 30: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 31: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Device Type 2020 & 2033
- Table 32: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 33: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 34: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 35: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Country 2020 & 2033
- Table 37: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Device Type 2020 & 2033
- Table 38: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 39: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Geography 2020 & 2033
- Table 40: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Geography 2020 & 2033
- Table 41: Global APAC Single-Use Medical Device Reprocessing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 42: Global APAC Single-Use Medical Device Reprocessing Industry Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the APAC Single-Use Medical Device Reprocessing Industry?
The projected CAGR is approximately 5%.
2. Which companies are prominent players in the APAC Single-Use Medical Device Reprocessing Industry?
Key companies in the market include Sterilmed Inc (Johnson & Johnson), Soma Technology Inc, ReNu Medical Inc (Arjo Inc ), Stryker Corporation, NEScientific Inc, Medline Industries Inc.
3. What are the main segments of the APAC Single-Use Medical Device Reprocessing Industry?
The market segments include Device Type, Geography.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
Rising Significance of Medical Waste Minimization; Reprocessing Single-use Devices Results in Environment Sustainability; Cost-Saving through Reprocessing Single-use Devices.
6. What are the notable trends driving market growth?
Sequential Compression Sleeves Segment is Expected to Witness Growth Over the Forecast Period.
7. Are there any restraints impacting market growth?
Poor Regulation of SUD Reprocessing in Emerging Markets; Preconceived Notion regarding the Quality of Reprocessed Single-use Medical Devices.
8. Can you provide examples of recent developments in the market?
August 2022- Ecolab Inc. expanded its Healthcare division to serve hospitals and healthcare facilities in Southeast Asia. A leader in infection prevention solutions and services, Ecolab Healthcare, offers a comprehensive array of hygiene and infection prevention solutions for hospitals, surgical centers, and healthcare facilities. They also provide instrument & endoscopy reprocessing, surgical drapes, and environmental hand hygiene.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "APAC Single-Use Medical Device Reprocessing Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the APAC Single-Use Medical Device Reprocessing Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the APAC Single-Use Medical Device Reprocessing Industry?
To stay informed about further developments, trends, and reports in the APAC Single-Use Medical Device Reprocessing Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
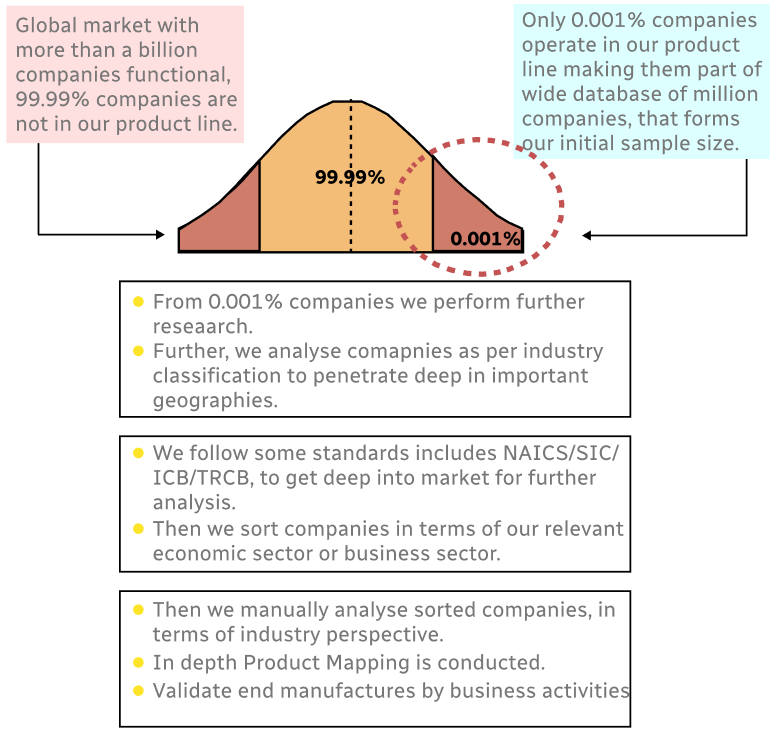
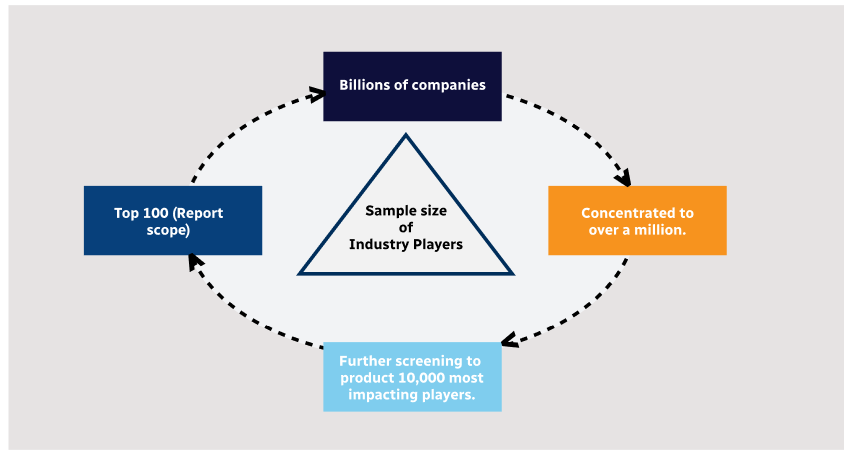
Step 1 - Identification of Relevant Samples Size from Population Database
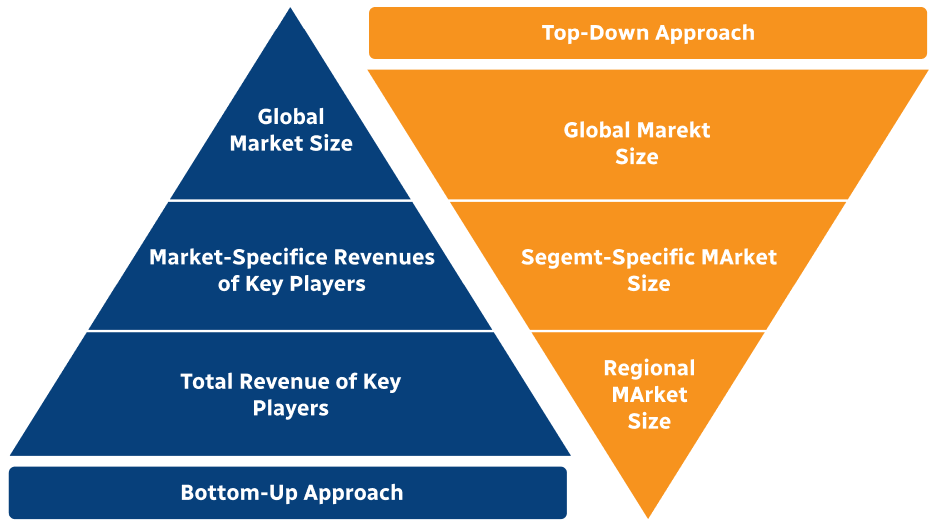
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
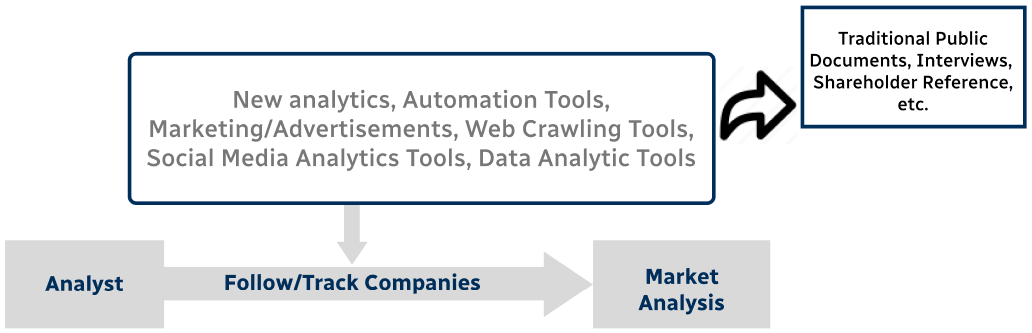
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


