Key Insights
The global Syringe Labels market is poised for substantial growth, with a projected market size of USD 6.84 billion in 2025, driven by a robust CAGR of 15.77%. This expansion is primarily fueled by the increasing demand for enhanced patient safety and accurate medication management within healthcare settings. The pharmaceutical industry's continuous innovation in drug delivery systems, coupled with the growing prevalence of chronic diseases requiring regular treatment, further bolsters the need for reliable and informative syringe labeling solutions. Laboratories and research organizations also contribute significantly to market demand as they require precise and durable labels for sample identification and experimental tracking. The rising adoption of advanced printing technologies and specialized label materials designed for extreme conditions, such as sterilization processes, are also key drivers for this dynamic market.
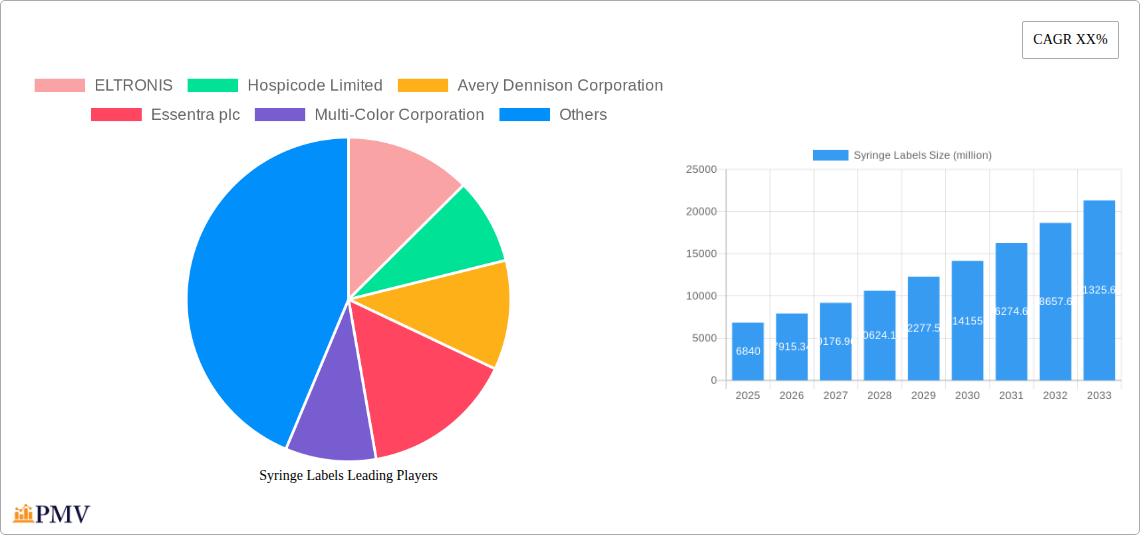
Syringe Labels Market Size (In Billion)

Looking ahead, the market is expected to witness sustained growth through 2033, propelled by technological advancements in label materials and printing, alongside stringent regulatory requirements for pharmaceutical packaging. While the market enjoys strong growth, potential restraints may include fluctuations in raw material costs for label production and intense competition among key players. However, the overarching trend towards digitalization in healthcare, including electronic health records and smart packaging solutions, is creating new opportunities for innovative syringe label functionalities, such as embedded RFID or QR codes for enhanced traceability and patient compliance. This evolution signifies a shift from simple identification to comprehensive data management within the healthcare supply chain.
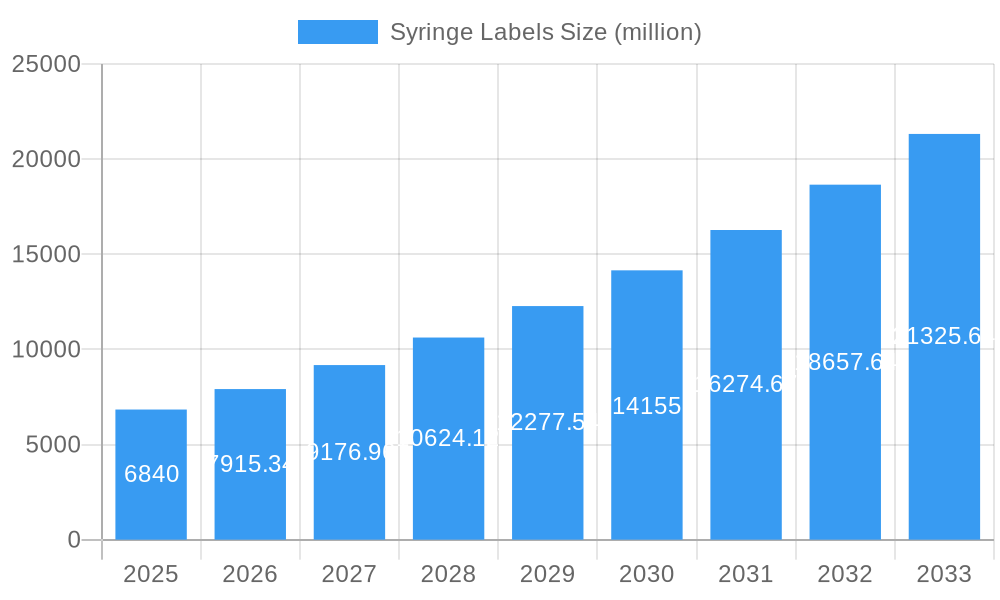
Syringe Labels Company Market Share

This in-depth report provides a comprehensive analysis of the global Syringe Labels market, offering critical insights into its structure, competitive landscape, evolving trends, and future outlook. Covering a study period from 2019 to 2033, with a base year of 2025, the report delves into historical performance, current dynamics, and projected growth. It is an indispensable resource for stakeholders seeking to understand market penetration, innovation, and strategic opportunities within the pharmaceutical, healthcare, and research sectors. The report examines the market across diverse applications, types, and key geographies, detailing influential drivers, challenges, and the strategic maneuvers of leading industry players.
Syringe Labels Market Structure & Competitive Dynamics
The global Syringe Labels market exhibits a moderately concentrated structure, characterized by the presence of both established multinational corporations and agile regional players. Leading entities such as Avery Dennison Corporation, Essentra plc, and Multi-Color Corporation command significant market share due to their extensive product portfolios, robust distribution networks, and ongoing investment in research and development. The innovation ecosystem is vibrant, driven by the increasing demand for advanced labeling solutions that enhance patient safety, traceability, and regulatory compliance. Regulatory frameworks, particularly within the pharmaceutical and healthcare industries, play a pivotal role in shaping market dynamics, necessitating adherence to stringent quality standards and serialization requirements. Product substitutes, while present in simpler labeling solutions, are largely unable to match the specialized functionality and durability offered by advanced syringe labels, especially those designed for harsh sterilization processes or complex drug delivery systems. End-user trends are strongly influenced by the growing emphasis on medication adherence, counterfeit drug prevention, and the increasing adoption of pre-filled syringes. Merger and acquisition (M&A) activities, estimated to be in the billions, are strategic moves by companies to expand their geographical reach, acquire new technologies, and consolidate their market position. Recent M&A deal values are projected to exceed $1.5 billion in the forecast period, underscoring the market's strategic importance.
Syringe Labels Industry Trends & Insights
The Syringe Labels market is experiencing robust growth, driven by a confluence of technological advancements, evolving regulatory mandates, and the escalating demands of the global healthcare industry. A significant market growth driver is the increasing prevalence of chronic diseases and the subsequent rise in pharmaceutical drug production and administration, particularly injectables. Technological disruptions are at the forefront, with the integration of advanced printing technologies, including digital printing, enabling faster turnaround times, greater customization, and improved print quality. The advent of smart labels, incorporating features like RFID and NFC, is revolutionizing traceability and inventory management throughout the supply chain, from manufacturing to patient administration. Consumer preferences are shifting towards enhanced patient safety and ease of use, pushing manufacturers to develop labels with clearer markings, tamper-evident features, and materials that withstand various storage and sterilization conditions. Competitive dynamics are intensifying, with companies focusing on differentiating themselves through specialized product offerings, such as ultra-thin labels for sensitive applications, chemically resistant materials, and labels designed for automated labeling equipment. The market penetration of high-performance synthetic labels is steadily increasing, outperforming traditional paper labels in demanding applications. The compound annual growth rate (CAGR) for the Syringe Labels market is projected to be approximately 7.5% during the forecast period. Market penetration is currently estimated at 60% of all injectable drug administrations, with significant room for growth. The increasing adoption of personalized medicine also fuels demand for highly specific and trackable labeling solutions. Furthermore, the global push for pharmaceutical supply chain integrity and the prevention of counterfeit drugs are significant accelerators, making advanced labeling solutions indispensable. The growing importance of data security and integrity in healthcare records also contributes to the demand for sophisticated labeling that can be seamlessly integrated with digital tracking systems. The development of eco-friendly labeling materials is another emerging trend, aligning with the broader sustainability initiatives within the packaging and healthcare industries.
Dominant Markets & Segments in Syringe Labels
North America currently dominates the Syringe Labels market, driven by a well-established pharmaceutical industry, advanced healthcare infrastructure, and stringent regulatory requirements that mandate high-quality labeling for patient safety and drug traceability. The United States, in particular, represents a substantial portion of this regional dominance, owing to its significant pharmaceutical manufacturing base and high per capita healthcare expenditure.
Application: Pharmaceutical Company: Pharmaceutical companies are the largest consumers of syringe labels, utilizing them for the primary packaging of a vast array of injectable medications. Key drivers for this segment's dominance include the sheer volume of drug production, the critical need for accurate drug identification, dosage information, and batch tracking to prevent medication errors and ensure patient safety. The increasing development of biologics and specialty drugs, often requiring complex storage and handling, further amplifies the demand for specialized syringe labels. The market size for this segment is estimated at $2.8 billion in 2025.
Application: Hospital: Hospitals are crucial end-users, employing syringe labels for the preparation and administration of medications to patients. Drivers include the need for clear identification of medications in high-pressure clinical environments, the prevention of cross-contamination, and the implementation of barcoding systems for inventory management and electronic health records. The rising number of surgical procedures and the growing complexity of patient care further bolster demand. The market size for this segment is projected at $1.5 billion in 2025.
Application: Laboratories: Clinical and research laboratories utilize syringe labels for sample identification, tracking of reagents, and experimental protocols. Drivers include the need for precise labeling to maintain the integrity of research data and ensure accurate results in diagnostic testing. The expansion of genomic research and the development of novel diagnostic assays contribute to sustained demand. The market size for this segment is estimated at $0.7 billion in 2025.
Application: Clinics: Outpatient clinics and specialized treatment centers rely on syringe labels for the administration of a wide range of therapies, including vaccinations, chemotherapy, and pain management. Drivers include the need for clear patient identification, medication details, and the ability of labels to withstand varying environmental conditions. The increasing focus on ambulatory care and preventive medicine fuels this segment. The market size for this segment is projected at $0.4 billion in 2025.
Types: Polyester: Polyester labels are increasingly dominant due to their exceptional durability, resistance to chemicals, moisture, and extreme temperatures, making them ideal for sterilization processes and long-term storage. Their smooth surface also allows for high-resolution printing of intricate details and bar codes, enhancing traceability. The market size for this segment is estimated at $3.2 billion in 2025.
Types: Synthetic: Beyond polyester, other synthetic materials offer excellent tear resistance, printability, and adhesion properties, making them versatile for various syringe labeling applications. Their ability to maintain legibility under adverse conditions makes them a preferred choice in critical healthcare settings. The market size for this segment is projected at $1.8 billion in 2025.
Syringe Labels Product Innovations
Recent product innovations in the Syringe Labels market are centered on enhancing patient safety, improving traceability, and meeting evolving regulatory demands. Advancements include the development of ultra-thin, flexible labels that conform seamlessly to syringe contours without compromising readability. Tamper-evident features, antimicrobial coatings, and chemically resistant materials for use with aggressive pharmaceuticals are also key developments. The integration of specialized adhesives ensures label adhesion through various sterilization methods, including gamma irradiation and autoclaving. Furthermore, the introduction of labels with enhanced printability for variable data printing, such as patient-specific information and batch serialization, provides significant competitive advantages by enabling greater customization and compliance with track-and-trace initiatives.
Report Segmentation & Scope
This report meticulously segments the Syringe Labels market to provide a granular understanding of its dynamics. The segmentation is based on Application, encompassing Pharmaceutical Companies, Hospitals, Laboratories, Clinics, and Other related sectors. Each application segment's growth projections, market sizes, and competitive dynamics are analyzed. Furthermore, the report segments the market by Type, including Paper, Polyester, and Synthetic labels. Detailed market sizes, growth forecasts, and competitive landscapes are provided for each material type, offering insights into their respective market penetration and strategic importance. The scope of the report covers global and regional market analyses, key player strategies, and emerging trends, offering a holistic view of the industry.
Key Drivers of Syringe Labels Growth
The Syringe Labels market is propelled by several key drivers. Technologically, the increasing demand for advanced labeling solutions that offer enhanced traceability, serialization, and data integrity is paramount. Regulatory mandates, such as the Drug Supply Chain Security Act (DSCSA) in the US and similar legislation globally, requiring serialization and track-and-trace capabilities, are significant growth accelerators. Economically, the expanding global pharmaceutical industry, particularly the growth in injectable drug markets and the development of biologics, directly translates to higher demand for specialized syringe labels. Furthermore, the increasing focus on patient safety and the prevention of medication errors in healthcare settings necessitates clear, durable, and informative labeling.
Challenges in the Syringe Labels Sector
Despite robust growth, the Syringe Labels sector faces several challenges. Stringent regulatory hurdles and the constant need for compliance with evolving standards can increase development costs and time-to-market. Supply chain disruptions, particularly for specialized raw materials, can impact production schedules and lead times. Intense competitive pressures among a diverse range of manufacturers, from large corporations to niche providers, can lead to price erosion in certain segments. Furthermore, the development of truly innovative, high-cost solutions must balance market affordability with the advanced functionalities required by end-users. Quantifiable impacts of supply chain issues are estimated to have caused production delays impacting 15% of market output in the historical period.
Leading Players in the Syringe Labels Market
- ELTRONIS
- Hospicode Limited
- Avery Dennison Corporation
- Essentra plc
- Multi-Color Corporation
- SheetLabels
- Sharp
- United Ad Label
- Nordvalls Etikett AB
- Schreiner MediPharm
- WATA LABEL Co.,Ltd.
- Shenzhen Ruilisibo Technology Co.,Ltd.
- Hang Shing Label Printing Product Co. Ltd
Key Developments in Syringe Labels Sector
- 2023 October: Schreiner MediPharm launched a new generation of RFID-enabled syringe labels for enhanced cold chain monitoring of sensitive pharmaceuticals.
- 2023 July: Avery Dennison Corporation announced the acquisition of a specialized label converter, expanding its capabilities in high-performance medical labeling.
- 2022 December: Essentra plc introduced a range of biodegradable syringe labels to address growing environmental sustainability concerns in the healthcare packaging sector.
- 2022 May: Multi-Color Corporation invested in advanced digital printing technology to increase its capacity for customized, serialized syringe labels for the pharmaceutical industry.
- 2021 November: WATA LABEL Co.,Ltd. expanded its product line to include labels with enhanced resistance to sterilization chemicals, catering to the needs of hospitals and pharmaceutical manufacturers.
Strategic Syringe Labels Market Outlook
The strategic outlook for the Syringe Labels market remains highly positive, fueled by continuous innovation and unwavering demand from the healthcare and pharmaceutical sectors. Growth accelerators include the persistent need for patient safety, the imperative for robust drug traceability in combating counterfeiting, and the expanding pipeline of injectable therapeutics. Opportunities lie in the development of smart labels with integrated digital capabilities, personalized labeling solutions for advanced therapies, and eco-friendly material alternatives. Companies that invest in advanced printing technologies, R&D for specialized materials, and strategic partnerships are well-positioned to capitalize on the evolving market landscape and achieve sustained growth in the coming years. The estimated market potential for advanced syringe labels is projected to reach $8.5 billion by 2033.
Syringe Labels Segmentation
-
1. Application
- 1.1. Pharmaceutical Company
- 1.2. Hospital
- 1.3. Laboratories
- 1.4. Clinics
- 1.5. Research Organization
- 1.6. Others
-
2. Types
- 2.1. Paper
- 2.2. Polyester
- 2.3. Synthetic
Syringe Labels Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
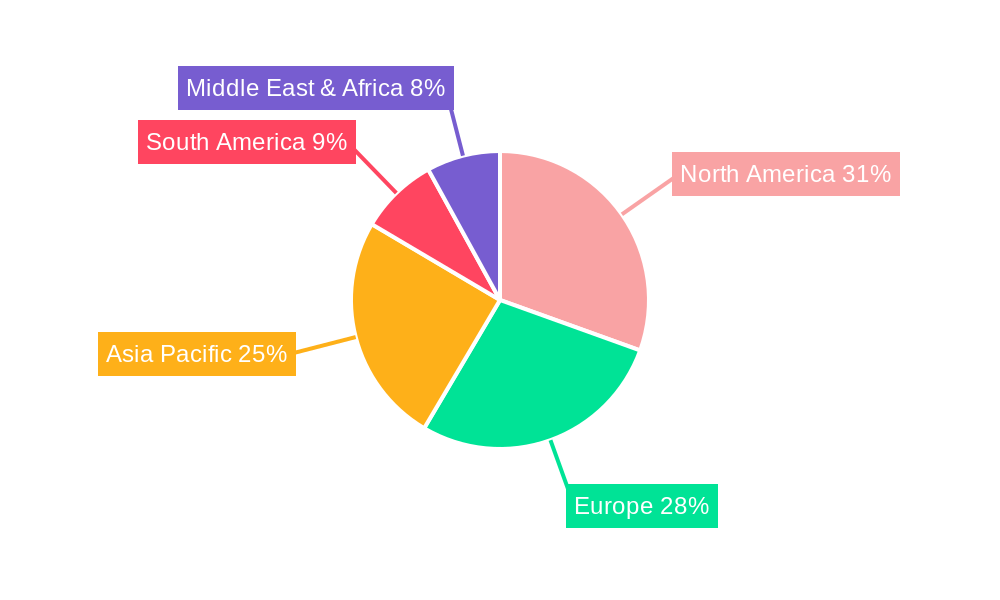
Syringe Labels Regional Market Share

Geographic Coverage of Syringe Labels
Syringe Labels REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Pharmaceutical Company
- 5.1.2. Hospital
- 5.1.3. Laboratories
- 5.1.4. Clinics
- 5.1.5. Research Organization
- 5.1.6. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Paper
- 5.2.2. Polyester
- 5.2.3. Synthetic
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Syringe Labels Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Pharmaceutical Company
- 6.1.2. Hospital
- 6.1.3. Laboratories
- 6.1.4. Clinics
- 6.1.5. Research Organization
- 6.1.6. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Paper
- 6.2.2. Polyester
- 6.2.3. Synthetic
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Syringe Labels Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Pharmaceutical Company
- 7.1.2. Hospital
- 7.1.3. Laboratories
- 7.1.4. Clinics
- 7.1.5. Research Organization
- 7.1.6. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Paper
- 7.2.2. Polyester
- 7.2.3. Synthetic
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Syringe Labels Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Pharmaceutical Company
- 8.1.2. Hospital
- 8.1.3. Laboratories
- 8.1.4. Clinics
- 8.1.5. Research Organization
- 8.1.6. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Paper
- 8.2.2. Polyester
- 8.2.3. Synthetic
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Syringe Labels Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Pharmaceutical Company
- 9.1.2. Hospital
- 9.1.3. Laboratories
- 9.1.4. Clinics
- 9.1.5. Research Organization
- 9.1.6. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Paper
- 9.2.2. Polyester
- 9.2.3. Synthetic
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Syringe Labels Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Pharmaceutical Company
- 10.1.2. Hospital
- 10.1.3. Laboratories
- 10.1.4. Clinics
- 10.1.5. Research Organization
- 10.1.6. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Paper
- 10.2.2. Polyester
- 10.2.3. Synthetic
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Syringe Labels Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Pharmaceutical Company
- 11.1.2. Hospital
- 11.1.3. Laboratories
- 11.1.4. Clinics
- 11.1.5. Research Organization
- 11.1.6. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Paper
- 11.2.2. Polyester
- 11.2.3. Synthetic
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 ELTRONIS
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Hospicode Limited
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Avery Dennison Corporation
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Essentra plc
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Multi-Color Corporation
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 SheetLabels
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Sharp
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 United Ad Label
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Nordvalls Etikett AB
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Schreiner MediPharm
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 WATA LABEL Co.
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Ltd.
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 Shenzhen Ruilisibo Technology Co.
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 Ltd.
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Hang Shing Label Printing Product Co. Ltd
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.1 ELTRONIS
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Syringe Labels Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Syringe Labels Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Syringe Labels Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America Syringe Labels Volume (K), by Application 2025 & 2033
- Figure 5: North America Syringe Labels Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Syringe Labels Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Syringe Labels Revenue (undefined), by Types 2025 & 2033
- Figure 8: North America Syringe Labels Volume (K), by Types 2025 & 2033
- Figure 9: North America Syringe Labels Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Syringe Labels Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Syringe Labels Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Syringe Labels Volume (K), by Country 2025 & 2033
- Figure 13: North America Syringe Labels Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Syringe Labels Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Syringe Labels Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America Syringe Labels Volume (K), by Application 2025 & 2033
- Figure 17: South America Syringe Labels Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Syringe Labels Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Syringe Labels Revenue (undefined), by Types 2025 & 2033
- Figure 20: South America Syringe Labels Volume (K), by Types 2025 & 2033
- Figure 21: South America Syringe Labels Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Syringe Labels Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Syringe Labels Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America Syringe Labels Volume (K), by Country 2025 & 2033
- Figure 25: South America Syringe Labels Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Syringe Labels Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Syringe Labels Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe Syringe Labels Volume (K), by Application 2025 & 2033
- Figure 29: Europe Syringe Labels Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Syringe Labels Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Syringe Labels Revenue (undefined), by Types 2025 & 2033
- Figure 32: Europe Syringe Labels Volume (K), by Types 2025 & 2033
- Figure 33: Europe Syringe Labels Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Syringe Labels Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Syringe Labels Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe Syringe Labels Volume (K), by Country 2025 & 2033
- Figure 37: Europe Syringe Labels Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Syringe Labels Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Syringe Labels Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa Syringe Labels Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Syringe Labels Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Syringe Labels Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Syringe Labels Revenue (undefined), by Types 2025 & 2033
- Figure 44: Middle East & Africa Syringe Labels Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Syringe Labels Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Syringe Labels Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Syringe Labels Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa Syringe Labels Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Syringe Labels Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Syringe Labels Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Syringe Labels Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific Syringe Labels Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Syringe Labels Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Syringe Labels Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Syringe Labels Revenue (undefined), by Types 2025 & 2033
- Figure 56: Asia Pacific Syringe Labels Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Syringe Labels Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Syringe Labels Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Syringe Labels Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific Syringe Labels Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Syringe Labels Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Syringe Labels Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Syringe Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Syringe Labels Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Syringe Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 4: Global Syringe Labels Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Syringe Labels Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Syringe Labels Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Syringe Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Syringe Labels Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Syringe Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 10: Global Syringe Labels Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Syringe Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Syringe Labels Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Syringe Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global Syringe Labels Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Syringe Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 22: Global Syringe Labels Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Syringe Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Syringe Labels Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Syringe Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global Syringe Labels Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Syringe Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 34: Global Syringe Labels Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Syringe Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global Syringe Labels Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Syringe Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global Syringe Labels Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Syringe Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 58: Global Syringe Labels Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Syringe Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Syringe Labels Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Syringe Labels Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global Syringe Labels Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Syringe Labels Revenue undefined Forecast, by Types 2020 & 2033
- Table 76: Global Syringe Labels Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Syringe Labels Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global Syringe Labels Volume K Forecast, by Country 2020 & 2033
- Table 79: China Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Syringe Labels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Syringe Labels Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Syringe Labels?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the Syringe Labels?
Key companies in the market include ELTRONIS, Hospicode Limited, Avery Dennison Corporation, Essentra plc, Multi-Color Corporation, SheetLabels, Sharp, United Ad Label, Nordvalls Etikett AB, Schreiner MediPharm, WATA LABEL Co., Ltd., Shenzhen Ruilisibo Technology Co., Ltd., Hang Shing Label Printing Product Co. Ltd.
3. What are the main segments of the Syringe Labels?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Syringe Labels," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Syringe Labels report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Syringe Labels?
To stay informed about further developments, trends, and reports in the Syringe Labels, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
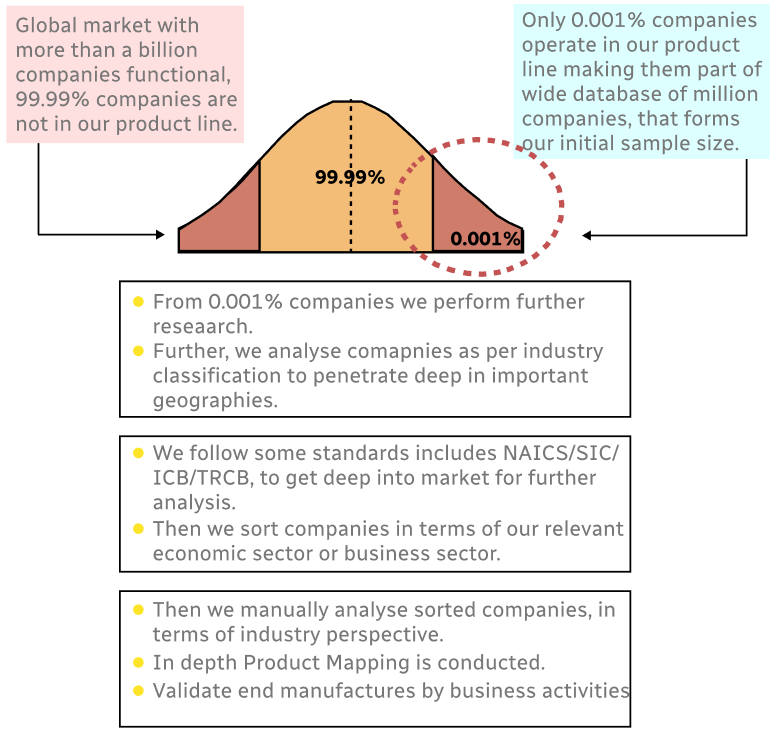
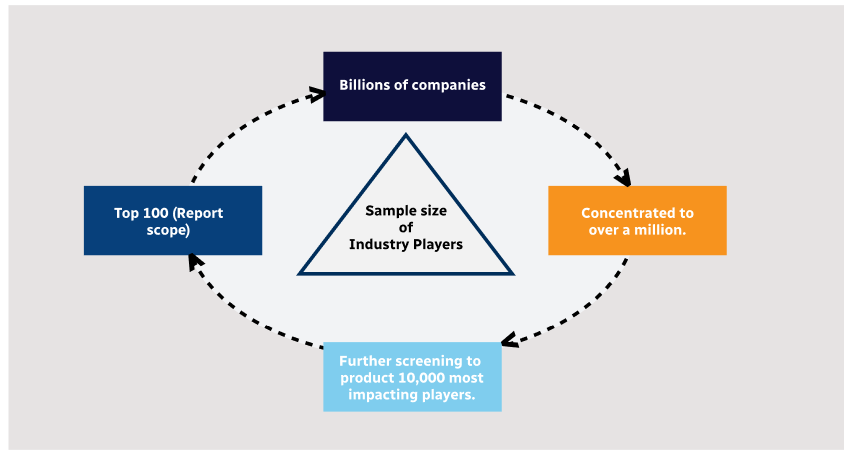
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
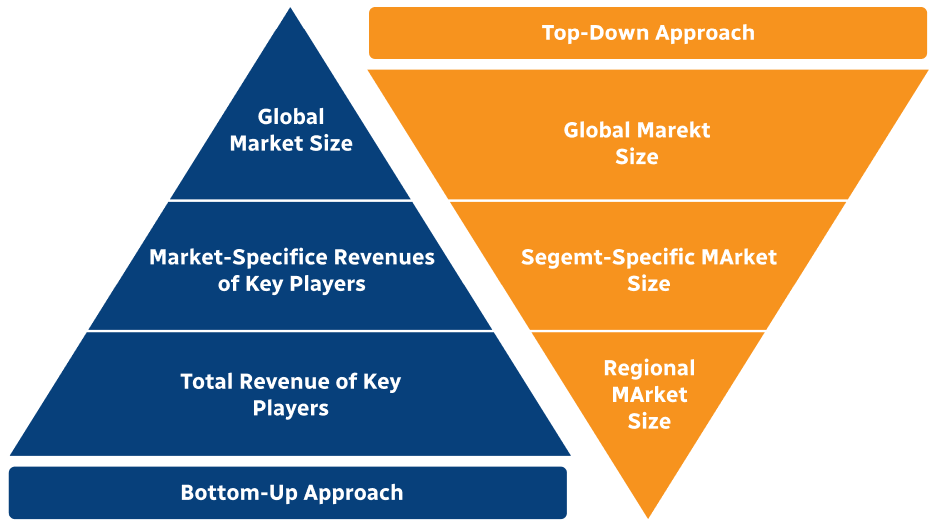
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
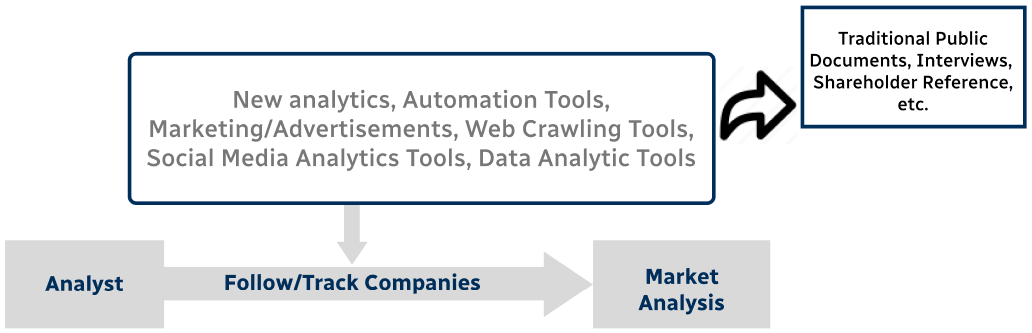
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


