Key Insights
The Microbiome CDMO market is poised for significant expansion, with an estimated market size of $1431 million in 2025 and a projected CAGR of 9.8% from 2019 to 2033. This robust growth is fueled by a confluence of factors, primarily the escalating research and development in microbiome-based therapeutics and diagnostics. Key drivers include the increasing understanding of the microbiome's role in various diseases, from gastrointestinal disorders to immune conditions and even neurological ailments. This burgeoning scientific knowledge is directly translating into a surge in demand for specialized contract development and manufacturing organizations (CDMOs) capable of handling the unique complexities of microbiome products. The pharmaceutical and biopharmaceutical sectors are increasingly outsourcing these specialized services, recognizing the cost-effectiveness and expertise offered by dedicated CDMOs in areas such as strain development, fermentation, formulation, and large-scale manufacturing. Furthermore, advancements in cell therapy and the growing potential of phage therapy and bacteriocin-based treatments are creating new avenues for market growth, pushing the boundaries of what microbiome interventions can achieve.
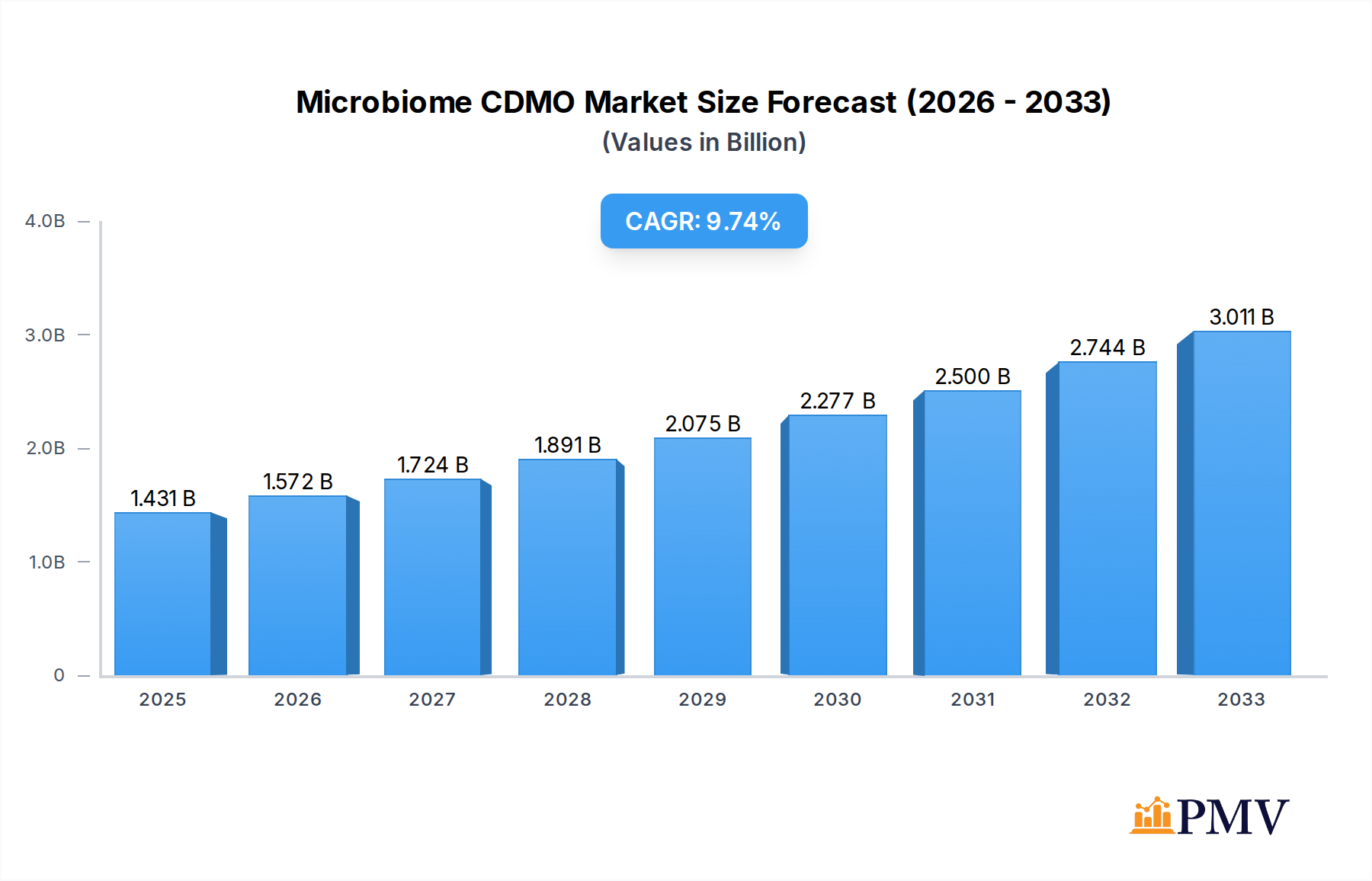
Microbiome CDMO Market Size (In Billion)

The market is characterized by a diverse range of applications, with Vaccine R&D and Biopharmaceutical segments leading the charge, followed closely by the burgeoning Cell Therapy sector. The "Others" category, encompassing novel applications and diagnostics, also presents significant untapped potential. On the supply side, Microbial CDMOs and Antibiotic CDMOs represent the dominant types, reflecting the current focus on bacterial-based therapies. However, the emergence of Phage CDMOs and Bacteriocins CDMOs signifies a strategic shift towards more targeted and innovative microbiome solutions. Geographically, North America and Europe currently hold substantial market shares due to established biopharmaceutical hubs and strong R&D investments. However, the Asia Pacific region is rapidly gaining traction, driven by increasing government support for biotechnology and a growing pool of skilled professionals. Restraints such as stringent regulatory hurdles and the need for specialized manufacturing infrastructure are present but are being systematically addressed by market players investing in advanced technologies and navigating the evolving regulatory landscape.
Microbiome CDMO Company Market Share

This in-depth Microbiome CDMO market report delivers a detailed examination of the global contract development and manufacturing organization (CDMO) landscape for microbiome-based therapeutics. Analyzing the microbial CDMO, antibiotic CDMO, phage CDMO, and bacteriocins CDMO segments, this report provides actionable insights for stakeholders in biopharmaceutical, cell therapy, vaccine R&D, and other emerging microbiome applications. The study spans the historical period from 2019 to 2024, with a base year of 2025, and projects market dynamics through 2033.
Microbiome CDMO Market Structure & Competitive Dynamics
The global Microbiome CDMO market exhibits a moderately concentrated structure, driven by a growing number of specialized players and established biopharmaceutical manufacturers expanding their capabilities. Innovation ecosystems are rapidly evolving, fueled by significant investment in microbiome research and development, leading to a dynamic competitive landscape. Regulatory frameworks, while still maturing for microbiome-based products, are becoming more defined, influencing market entry and operational standards. Product substitutes are limited due to the unique nature of microbiome therapeutics, but alternative therapeutic modalities continue to emerge. End-user trends show increasing demand for personalized medicine and targeted therapies, directly benefiting the microbiome CDMO sector. Merger and acquisition (M&A) activities are a significant feature, with recent deals valued in the hundreds of millions, consolidating market share and expanding service offerings. For instance, several microbial CDMO firms have been acquired to bolster the capabilities of larger organizations.
- Market Concentration: Moderate, with a blend of niche specialists and larger CDMOs.
- Innovation Ecosystems: Highly active, driven by academic research and venture capital funding.
- Regulatory Frameworks: Evolving, with a focus on safety, efficacy, and manufacturing standards.
- Product Substitutes: Limited for microbiome therapeutics, but competition from other novel drug modalities exists.
- End-User Trends: Growing preference for targeted and personalized therapies.
- M&A Activities: Significant, with deal values often exceeding several hundred million dollars, signifying consolidation and strategic expansion.
Microbiome CDMO Industry Trends & Insights
The microbiome CDMO industry is poised for substantial growth, propelled by a confluence of escalating market growth drivers, disruptive technological advancements, and shifting consumer preferences. The inherent potential of the human microbiome to address unmet medical needs in areas ranging from gastrointestinal disorders to oncology and infectious diseases is a primary catalyst. Advancements in genomic sequencing, bioinformatics, and microbial cultivation techniques are enabling a deeper understanding of microbial consortia and their therapeutic applications, facilitating the development of more sophisticated microbiome CDMO services. Consumer awareness regarding the impact of the microbiome on overall health and well-being is also on the rise, translating into increased demand for microbiome-based products and, consequently, for specialized CDMO support. The competitive dynamics are characterized by both collaboration and intense rivalry, as companies strive to establish leadership in this nascent yet rapidly expanding market. The microbial CDMO segment, in particular, is experiencing robust expansion as more companies seek expertise in handling and manufacturing live microbial products. The projected Compound Annual Growth Rate (CAGR) for the microbiome CDMO market is estimated to be in the high teens, reflecting significant market penetration opportunities.
Dominant Markets & Segments in Microbiome CDMO
The Microbiome CDMO market is witnessing significant dominance in specific regions and application segments. North America, particularly the United States, leads the market due to robust research infrastructure, substantial venture capital investments in biotechnology, and a favorable regulatory environment for novel therapeutics. The Biopharmaceutical application segment is currently the most dominant, driven by the extensive pipeline of microbiome-based drugs targeting various chronic and complex diseases. Within the types of CDMO services, the Microbial CDMO segment holds the largest market share, reflecting the foundational nature of live microbial therapies.
- Leading Region: North America, driven by advanced research, funding, and regulatory support.
- Dominant Application Segment: Biopharmaceutical, encompassing a wide range of therapeutic indications.
- Dominant Type Segment: Microbial CDMO, owing to the widespread focus on live bacterial consortia.
Key drivers for this dominance include:
- Economic Policies: Government incentives and tax credits supporting biotech R&D.
- Infrastructure: Well-established network of research institutions, clinical trial sites, and manufacturing facilities.
- Investment Landscape: Strong presence of venture capital firms actively funding microbiome startups and CDMOs.
- Regulatory Agility: While evolving, regulatory pathways in leading regions are more defined for novel therapies.
The dominance of the Biopharmaceutical segment is further bolstered by advancements in understanding disease mechanisms and developing targeted interventions. Similarly, the Microbial CDMO segment’s leadership is attributed to the maturity of techniques for culturing, preserving, and formulating live microbial products for therapeutic use. As the market matures, other segments like Cell Therapy and specialized areas such as Phage CDMO and Bacteriocins CDMO are expected to gain significant traction.
Microbiome CDMO Product Innovations
Product innovations within the Microbiome CDMO market are primarily centered on the development of live biotherapeutic products (LBPs), engineered microbes, and microbial-derived compounds. CDMOs are increasingly offering specialized services for the formulation, fill-finish, and scale-up of these novel therapeutic modalities. Key applications include the treatment of inflammatory bowel disease, metabolic disorders, and infectious diseases. Competitive advantages are being built on expertise in strain selection, fermentation optimization, and ensuring product stability and shelf-life. Technological trends favor the development of precision microbiome interventions and multi-strain consortia, which require sophisticated manufacturing capabilities.
Report Segmentation & Scope
This report meticulously segments the Microbiome CDMO market across key dimensions to provide granular insights.
- Application Segmentation:
- Vaccine R&D: Focuses on CDMO support for developing microbiome-based vaccines. Projected market size in the range of several hundred million dollars with a strong CAGR.
- Biopharmaceutical: The largest segment, encompassing therapeutic applications across various disease areas. Expected to reach several billion dollars in market value.
- Cell Therapy: CDMO services for integrating microbiome components with cell-based therapies. Growing segment with significant future potential.
- Others: Includes diagnostics, nutritional supplements, and research tools.
- Type Segmentation:
- Microbial CDMO: The foundational segment, offering services for live microbial products. Expected to maintain its leading position with a market value in the billions.
- Antibiotic CDMO: Services related to the development and manufacturing of microbiome-derived antibiotics. A niche but growing area.
- Phage CDMO: Specializing in the production of bacteriophages for therapeutic applications. High growth potential as phage therapy gains traction.
- Bacteriocins CDMO: Focusing on the manufacturing of bacteriocin-based antimicrobials.
Key Drivers of Microbiome CDMO Growth
The Microbiome CDMO market is propelled by several interconnected factors. Technological advancements in genomics and bioinformatics enable deeper understanding and targeted development of microbiome interventions. The increasing prevalence of chronic diseases and the limitations of existing treatments are creating a demand for novel therapeutic approaches like microbiome-based drugs. Furthermore, the growing body of scientific evidence demonstrating the therapeutic potential of the microbiome is fueling R&D investment. Favorable regulatory pathways for microbiome therapeutics, although still evolving, are encouraging commercialization.
- Technological Advancements: Next-generation sequencing, advanced fermentation techniques, and AI-driven data analysis.
- Disease Burden: Rising incidence of chronic diseases and the need for new treatment modalities.
- Scientific Validation: Robust clinical trial data supporting the efficacy of microbiome interventions.
- Investment & Funding: Significant venture capital and government funding for microbiome research and development.
Challenges in the Microbiome CDMO Sector
Despite its promising outlook, the Microbiome CDMO sector faces notable challenges. Regulatory hurdles remain a significant barrier, with a lack of standardized guidelines for the manufacturing and approval of live biotherapeutic products. Supply chain complexities, particularly in maintaining the viability and consistency of live microbial products, present operational difficulties. Intense competition from both established CDMOs and emerging specialized players can also put pressure on pricing and market share. Ensuring product quality and safety at scale is paramount and requires specialized expertise.
- Regulatory Uncertainty: Evolving guidelines for live biotherapeutic products.
- Supply Chain Complexity: Maintaining viability and consistency of live microbial products.
- Manufacturing Scale-up: Challenges in scaling up fermentation and formulation processes.
- Quality Control: Ensuring product purity, potency, and stability.
Leading Players in the Microbiome CDMO Market
- Arranta Bio
- Wacker Chemie AG
- Bactera
- SGS Quay Pharma
- Biose
- EnteroBiotix
- Genome And Company
- Lallemand Health Solutions
- List Bio
- Rise BioServices
- Lonza
- MaaT Pharma
- NIZO
- Quay Pharmaceuticals
- Recipharm
Key Developments in Microbiome CDMO Sector
- 2023: Arranta Bio expands its manufacturing capacity by 10 million, focusing on LBPs.
- 2023: Lonza announces a strategic partnership with MaaT Pharma to advance cell therapy manufacturing, including microbiome components.
- 2024: Wacker Chemie AG invests 50 million in advanced fermentation technologies for microbial products.
- 2024: Biose acquires a specialized microbial strain library, enhancing its R&D capabilities.
- 2024: Recipharm announces a new facility dedicated to aseptic fill-finish for microbiome therapeutics, with a capacity for several million doses.
- 2024: EnteroBiotix secures 20 million in funding to scale its manufacturing operations for gut microbiome therapies.
- 2024: Genome And Company secures a significant deal worth 100 million for the development of novel microbiome-based cancer therapies.
- 2024: Lallemand Health Solutions launches a new platform for customized microbial consortia development.
Strategic Microbiome CDMO Market Outlook
The strategic outlook for the Microbiome CDMO market is exceptionally strong, driven by ongoing scientific breakthroughs and increasing therapeutic validation. Market accelerators include the continuous innovation in LBP development, expansion into new therapeutic areas beyond gastrointestinal health, and the increasing adoption of integrated CDMO services. Opportunities lie in catering to the growing pipeline of phage CDMO and bacteriocins CDMO applications, as well as supporting the integration of microbiome components in advanced cell therapy platforms. Strategic investments in advanced manufacturing technologies and regulatory expertise will be crucial for CDMOs to capitalize on this dynamic and rapidly expanding market. The market is projected to witness sustained growth, reaching tens of billions in value by 2033.
Microbiome CDMO Segmentation
-
1. Application
- 1.1. Vaccine R&D
- 1.2. Biopharmaceutical
- 1.3. Cell Therapy
- 1.4. Others
-
2. Types
- 2.1. Microbial CDMO
- 2.2. Antibiotic CDMO
- 2.3. Phage CDMO
- 2.4. Bacteriocins CDMO
Microbiome CDMO Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
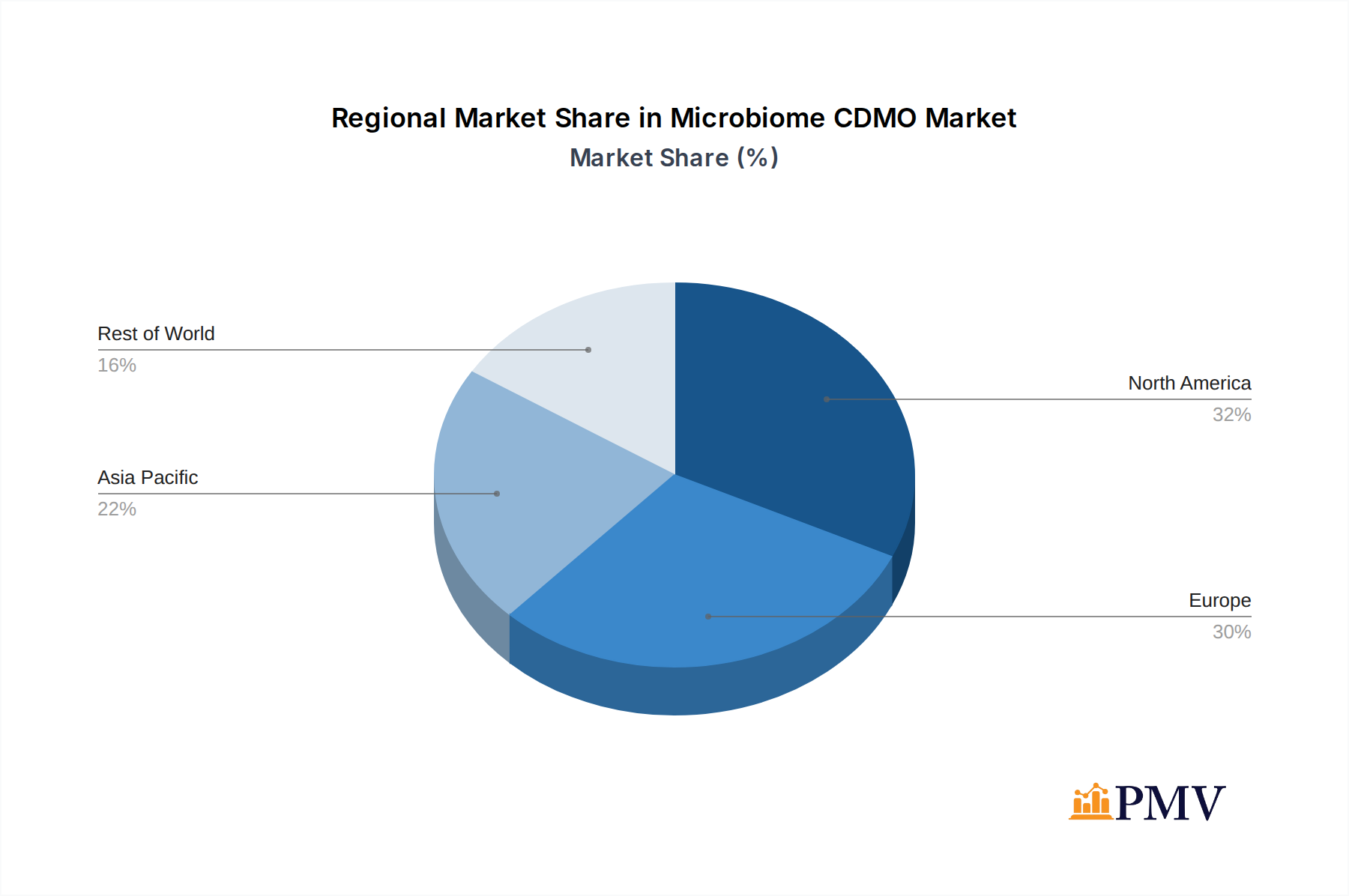
Microbiome CDMO Regional Market Share

Geographic Coverage of Microbiome CDMO
Microbiome CDMO REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Vaccine R&D
- 5.1.2. Biopharmaceutical
- 5.1.3. Cell Therapy
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Microbial CDMO
- 5.2.2. Antibiotic CDMO
- 5.2.3. Phage CDMO
- 5.2.4. Bacteriocins CDMO
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Microbiome CDMO Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Vaccine R&D
- 6.1.2. Biopharmaceutical
- 6.1.3. Cell Therapy
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Microbial CDMO
- 6.2.2. Antibiotic CDMO
- 6.2.3. Phage CDMO
- 6.2.4. Bacteriocins CDMO
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Microbiome CDMO Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Vaccine R&D
- 7.1.2. Biopharmaceutical
- 7.1.3. Cell Therapy
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Microbial CDMO
- 7.2.2. Antibiotic CDMO
- 7.2.3. Phage CDMO
- 7.2.4. Bacteriocins CDMO
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Microbiome CDMO Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Vaccine R&D
- 8.1.2. Biopharmaceutical
- 8.1.3. Cell Therapy
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Microbial CDMO
- 8.2.2. Antibiotic CDMO
- 8.2.3. Phage CDMO
- 8.2.4. Bacteriocins CDMO
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Microbiome CDMO Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Vaccine R&D
- 9.1.2. Biopharmaceutical
- 9.1.3. Cell Therapy
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Microbial CDMO
- 9.2.2. Antibiotic CDMO
- 9.2.3. Phage CDMO
- 9.2.4. Bacteriocins CDMO
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Microbiome CDMO Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Vaccine R&D
- 10.1.2. Biopharmaceutical
- 10.1.3. Cell Therapy
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Microbial CDMO
- 10.2.2. Antibiotic CDMO
- 10.2.3. Phage CDMO
- 10.2.4. Bacteriocins CDMO
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Microbiome CDMO Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Vaccine R&D
- 11.1.2. Biopharmaceutical
- 11.1.3. Cell Therapy
- 11.1.4. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Microbial CDMO
- 11.2.2. Antibiotic CDMO
- 11.2.3. Phage CDMO
- 11.2.4. Bacteriocins CDMO
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Arranta Bio
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Wacker Chemie AG
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Bactera
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 SGS Quay Pharma
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Biose
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 EnteroBiotix
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Genome And Company
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Lallemand Health Solutions
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 List Bio
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Rise BioServices
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Lonza
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 MaaT Pharma
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 NIZO
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 Quay Pharmaceuticals
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Recipharm
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.1 Arranta Bio
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Microbiome CDMO Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Microbiome CDMO Revenue (million), by Application 2025 & 2033
- Figure 3: North America Microbiome CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Microbiome CDMO Revenue (million), by Types 2025 & 2033
- Figure 5: North America Microbiome CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Microbiome CDMO Revenue (million), by Country 2025 & 2033
- Figure 7: North America Microbiome CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Microbiome CDMO Revenue (million), by Application 2025 & 2033
- Figure 9: South America Microbiome CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Microbiome CDMO Revenue (million), by Types 2025 & 2033
- Figure 11: South America Microbiome CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Microbiome CDMO Revenue (million), by Country 2025 & 2033
- Figure 13: South America Microbiome CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Microbiome CDMO Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Microbiome CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Microbiome CDMO Revenue (million), by Types 2025 & 2033
- Figure 17: Europe Microbiome CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Microbiome CDMO Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Microbiome CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Microbiome CDMO Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Microbiome CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Microbiome CDMO Revenue (million), by Types 2025 & 2033
- Figure 23: Middle East & Africa Microbiome CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Microbiome CDMO Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Microbiome CDMO Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Microbiome CDMO Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Microbiome CDMO Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Microbiome CDMO Revenue (million), by Types 2025 & 2033
- Figure 29: Asia Pacific Microbiome CDMO Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Microbiome CDMO Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Microbiome CDMO Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Microbiome CDMO Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Microbiome CDMO Revenue million Forecast, by Types 2020 & 2033
- Table 3: Global Microbiome CDMO Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Microbiome CDMO Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Microbiome CDMO Revenue million Forecast, by Types 2020 & 2033
- Table 6: Global Microbiome CDMO Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Microbiome CDMO Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Microbiome CDMO Revenue million Forecast, by Types 2020 & 2033
- Table 12: Global Microbiome CDMO Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Microbiome CDMO Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Microbiome CDMO Revenue million Forecast, by Types 2020 & 2033
- Table 18: Global Microbiome CDMO Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Microbiome CDMO Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Microbiome CDMO Revenue million Forecast, by Types 2020 & 2033
- Table 30: Global Microbiome CDMO Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Microbiome CDMO Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Microbiome CDMO Revenue million Forecast, by Types 2020 & 2033
- Table 39: Global Microbiome CDMO Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Microbiome CDMO Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Microbiome CDMO?
The projected CAGR is approximately 9.8%.
2. Which companies are prominent players in the Microbiome CDMO?
Key companies in the market include Arranta Bio, Wacker Chemie AG, Bactera, SGS Quay Pharma, Biose, EnteroBiotix, Genome And Company, Lallemand Health Solutions, List Bio, Rise BioServices, Lonza, MaaT Pharma, NIZO, Quay Pharmaceuticals, Recipharm.
3. What are the main segments of the Microbiome CDMO?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 1431 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4350.00, USD 6525.00, and USD 8700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Microbiome CDMO," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Microbiome CDMO report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Microbiome CDMO?
To stay informed about further developments, trends, and reports in the Microbiome CDMO, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
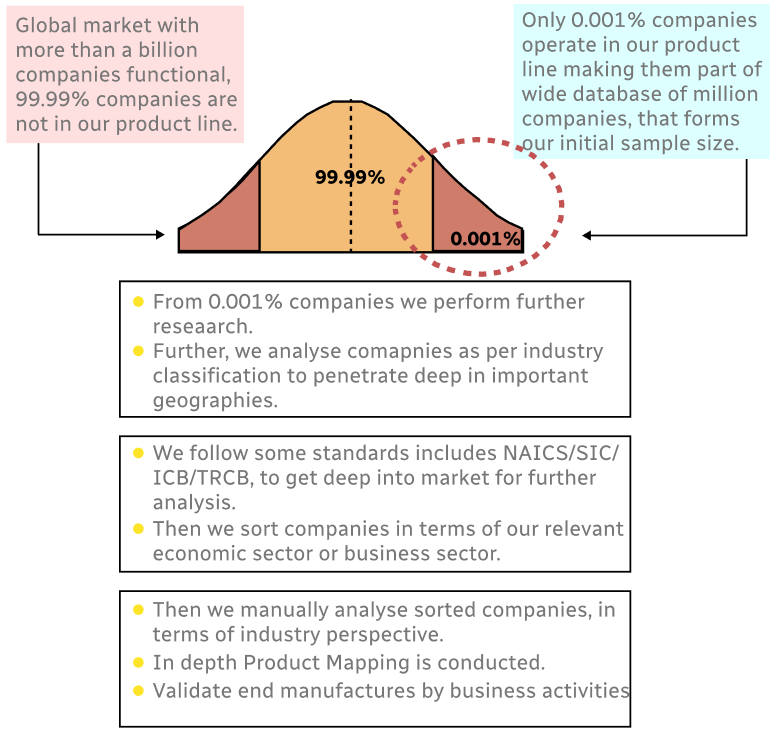
Step 1 - Identification of Relevant Samples Size from Population Database
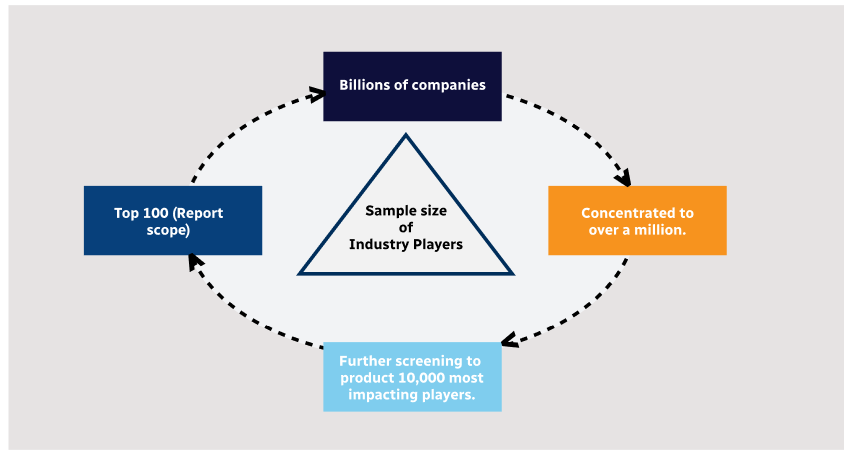
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
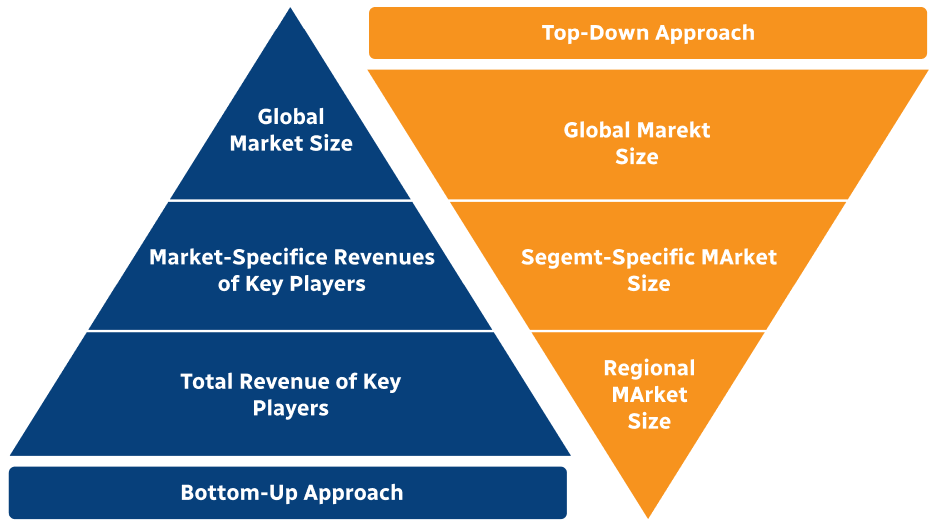
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


