Key Insights
The European Pharmaceutical Contract Manufacturing (CMO) market is poised for significant expansion, projected to reach USD 187.67 billion in 2024, with a robust Compound Annual Growth Rate (CAGR) of 8.6% through 2033. This impressive growth is underpinned by several key drivers. The increasing complexity of drug development and manufacturing, coupled with the rising demand for specialized dosage forms like injectables and advanced solid dose formulations, are compelling pharmaceutical companies to outsource production to specialized CMOs. Furthermore, the ongoing trend of pharmaceutical companies focusing on core competencies such as R&D and marketing, while delegating manufacturing to experts, fuels this market. The need for stringent regulatory compliance across Europe also benefits established CMOs with proven track records and advanced quality systems. Innovations in manufacturing technologies, including continuous manufacturing and advanced automation, are further enhancing efficiency and capacity, making outsourcing a more attractive proposition.
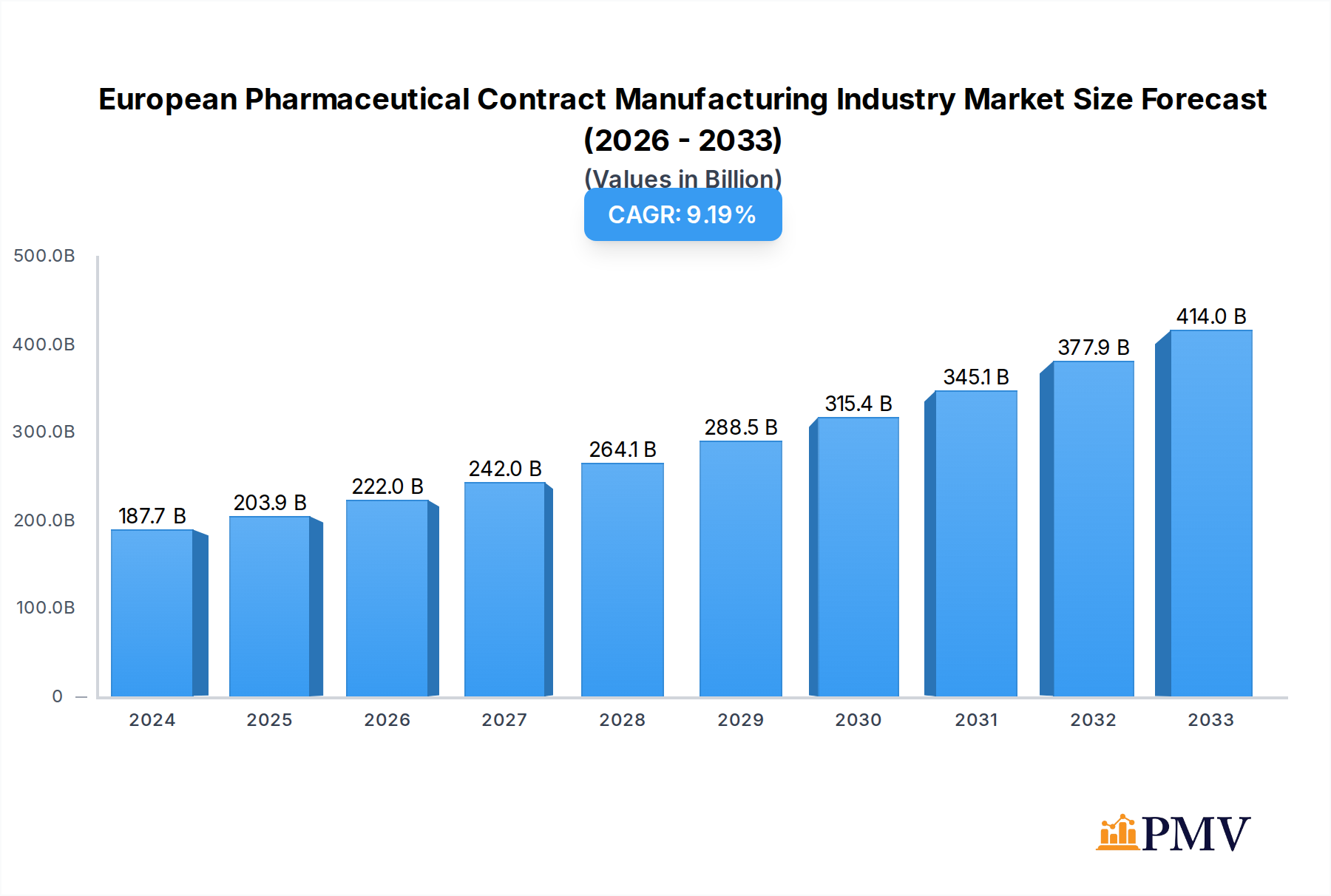
European Pharmaceutical Contract Manufacturing Industry Market Size (In Billion)

Restrains such as the increasing stringency of regulatory requirements, potential supply chain disruptions, and the high cost of specialized facilities and equipment can pose challenges. However, the market is actively addressing these by investing in advanced technologies and strengthening supply chain resilience. The market is segmented across various service types, including Active Pharmaceutical Ingredient (API) manufacturing, finished dosage form formulations (solid dose, liquid dose, and injectables), and secondary packaging. Leading companies like Lonza Group, Boehringer Ingelheim Group, and Recipharm AB are at the forefront, investing in capacity expansion and technological advancements to cater to the evolving needs of the pharmaceutical industry. Europe, with its strong pharmaceutical base and stringent quality standards, is a dominant region, with countries like the United Kingdom, Germany, France, and Italy showing significant activity in contract manufacturing. The demand for efficient and cost-effective manufacturing solutions will continue to drive this market's upward trajectory.
European Pharmaceutical Contract Manufacturing Industry Company Market Share

This comprehensive report delves into the dynamic European pharmaceutical contract manufacturing industry, providing in-depth analysis and a robust forecast from 2019 to 2033, with a base year of 2025. We examine the intricate market structure, competitive landscape, evolving trends, and critical growth drivers shaping this multi-billion dollar sector. For industry stakeholders seeking actionable intelligence on pharmaceutical CDMO services, API manufacturing, and finished dosage form production, this report offers unparalleled insights into market concentration, regulatory influences, and strategic opportunities.
European Pharmaceutical Contract Manufacturing Industry Market Structure & Competitive Dynamics
The European pharmaceutical contract manufacturing market is characterized by a moderate to high degree of concentration, with a handful of key players holding significant market share, contributing to a global market estimated to reach billions. Innovation ecosystems thrive, fueled by substantial R&D investments and strategic collaborations between pharmaceutical innovators and Contract Development and Manufacturing Organizations (CDMOs). Regulatory frameworks, particularly stringent in Europe, dictate operational standards and quality control, influencing market entry and competitive advantages. While product substitutes are limited in the core CDMO service offering, outsourcing strategies and technological advancements present indirect competitive pressures. End-user trends are increasingly focused on specialized formulations, complex biologics, and novel drug delivery systems, driving demand for advanced CDMO capabilities. Mergers & Acquisitions (M&A) activity is a significant aspect of market dynamics, with deal values in the billions, as companies seek to expand service portfolios, geographic reach, and technological expertise. For instance, consolidation through M&A is a key strategy for gaining market share and addressing the increasing complexity of drug development and manufacturing.
European Pharmaceutical Contract Manufacturing Industry Industry Trends & Insights
The European pharmaceutical contract manufacturing industry is experiencing robust growth, projected to expand at a significant Compound Annual Growth Rate (CAGR) throughout the forecast period, reaching a market size of billions. Key growth drivers include the escalating R&D pipelines of pharmaceutical companies, the increasing demand for specialized manufacturing capabilities, and the growing trend of outsourcing non-core activities by both large pharmaceutical enterprises and emerging biotech firms. Technological disruptions are continuously reshaping the landscape, with advancements in areas like continuous manufacturing, advanced analytics for process optimization, and the rise of personalized medicine requiring flexible and specialized CDMO services. Consumer preferences are indirectly influencing the market through a greater emphasis on drug efficacy, patient convenience, and innovative drug delivery systems, all of which necessitate sophisticated manufacturing solutions. Competitive dynamics are intense, with CDMOs differentiating themselves through specialized expertise, adherence to stringent quality standards (e.g., GMP compliance), and the ability to handle complex molecules and formulations. The market penetration of advanced manufacturing technologies is steadily increasing, as CDMOs invest in state-of-the-art facilities and skilled workforces to meet evolving industry demands.
Dominant Markets & Segments in European Pharmaceutical Contract Manufacturing Industry
The European pharmaceutical contract manufacturing industry demonstrates distinct dominance across various segments, driven by strong economic policies, advanced infrastructure, and a well-established pharmaceutical ecosystem.
- Leading Region: Western Europe, particularly Germany, Switzerland, the UK, and France, collectively represents the dominant market. This dominance is attributable to the presence of major pharmaceutical R&D hubs, a highly skilled workforce, and a favorable regulatory environment that supports innovation and high-quality manufacturing.
- Active Pharmaceutical Ingredient (API) Manufacturing: This segment is a critical driver of market growth, with significant investment in advanced synthesis technologies and capacity expansion. European CDMOs excel in producing complex and high-potency APIs, catering to the stringent requirements of global pharmaceutical companies.
- Finished Dosage Formulations:
- Solid Dose Formulation: This segment remains a cornerstone, driven by the prevalence of oral medications. CDMOs offer a wide array of services, from tablet compression to capsule filling, with a focus on enhanced bioavailability and patient compliance.
- Liquid Dose Formulation: Demand for sterile and non-sterile liquid formulations, including oral solutions, suspensions, and syrups, is substantial. The segment is characterized by specialized capabilities in sterile filling and aseptic processing.
- Injectable Dose Formulation: This highly specialized segment is experiencing rapid growth due to the increasing development of biologics and complex parenteral drugs. European CDMOs are investing heavily in aseptic manufacturing, lyophilization, and pre-filled syringe capabilities to meet this demand.
- Secondary Packaging: While often considered a supporting service, secondary packaging is crucial for regulatory compliance, product integrity, and market appeal. European CDMOs provide comprehensive solutions, including blister packaging, blistering, cartoning, and serialization, ensuring products meet diverse market requirements.
The economic policies in these leading regions foster innovation and investment in advanced manufacturing technologies, further solidifying their dominant position.
European Pharmaceutical Contract Manufacturing Industry Product Innovations
Product innovations within the European pharmaceutical contract manufacturing industry are primarily centered on enhancing manufacturing efficiency, improving drug delivery, and expanding the capabilities to handle novel therapeutic modalities. CDMOs are investing in technologies for continuous manufacturing, which promises higher yields, reduced waste, and improved quality control. Innovations in sterile fill-finish technologies, particularly for biologics and advanced therapies, are critical. Furthermore, specialized expertise in areas like high-potency API synthesis and the formulation of complex molecules continues to be a key competitive advantage, allowing CDMOs to cater to the evolving needs of drug developers and meet market demands for precision medicine.
Report Segmentation & Scope
This report meticulously segments the European pharmaceutical contract manufacturing industry to provide granular insights into specific market areas. The scope encompasses the following key service types:
- Active Pharmaceutical Ingredient (API) Manufacturing: This segment focuses on the synthesis and production of the core chemical compounds used in pharmaceutical drugs. Growth projections and market sizes for API manufacturing are detailed, considering the increasing demand for complex and specialized APIs.
- Finished Dosage Formulations: This broad category is further divided into:
- Solid Dose Formulation: Analysis of market size, growth, and competitive dynamics for tablets, capsules, and other solid dosage forms.
- Liquid Dose Formulation: Examination of oral solutions, suspensions, and injectables, including sterile and non-sterile manufacturing.
- Injectable Dose Formulation: In-depth coverage of sterile injectables, lyophilized products, and pre-filled syringes, a rapidly expanding segment.
- Secondary Packaging: This segment addresses the crucial aspect of packaging drug products, including blistering, cartoning, labeling, and serialization services, vital for regulatory compliance and market distribution.
Key Drivers of European Pharmaceutical Contract Manufacturing Industry Growth
Several key factors are propelling the growth of the European pharmaceutical contract manufacturing industry.
- Increasing R&D Pipelines: A robust pipeline of new drug candidates from pharmaceutical and biotech companies necessitates outsourcing manufacturing to specialized CDMOs.
- Technological Advancements: The adoption of new technologies like continuous manufacturing and single-use bioreactors enhances efficiency and reduces costs.
- Focus on Core Competencies: Pharmaceutical companies are increasingly outsourcing manufacturing to focus on their core competencies in R&D and marketing.
- Regulatory Compliance and Quality Standards: European CDMOs' adherence to stringent GMP and regulatory standards is a significant draw for clients.
- Cost Optimization: Outsourcing can offer cost advantages compared to in-house manufacturing, especially for smaller companies.
Challenges in the European Pharmaceutical Contract Manufacturing Industry Sector
Despite strong growth, the European pharmaceutical contract manufacturing industry faces several challenges.
- Stringent Regulatory Hurdles: Navigating diverse and evolving regulatory landscapes across European countries can be complex and time-consuming.
- Supply Chain Disruptions: Global supply chain volatility and geopolitical events can impact the availability of raw materials and lead times.
- Intense Competition: The market is highly competitive, with pressure on pricing and the need for continuous innovation and service differentiation.
- Talent Shortages: A shortage of skilled labor in specialized manufacturing areas can hinder capacity expansion and operational efficiency.
- Intellectual Property Protection: Ensuring robust protection of client intellectual property is paramount and requires advanced security measures.
Leading Players in the European Pharmaceutical Contract Manufacturing Industry Market
- Lonza Group
- Boehringer Ingelheim Group
- Recipharm AB
- Aenova Group
- Fareva Holdings SA
- Famar SA
- Cenexi - Laboratoires Thissen SA
- Almac Group
Key Developments in European Pharmaceutical Contract Manufacturing Industry Sector
- March 2022: MorphoSys, Germany, sacked US R&D to consolidate work in Germany, taking USD 254 million in charges. MorphoSys axed its early pipeline and U.S. R&D work that came with the USD 1.7 billion purchase of Constellation Pharmaceuticals, meaning a more than USD 250 million impairment charge as the German pharma shifted the focus home. This illustrates strategic consolidation and efficiency drives impacting CDMO demand for specific services.
- Feb 2022: Merck, Germany, restructured its business line to strengthen the CDMO business. Merck consolidated its existing Contract Development and Manufacturing Organization (CDMO) and contract testing services into one organization, Life Science Services (LSS), focused on traditional and novel modalities, along with the respective sales and marketing, research, and development (R&D), manufacturing, and supply chain operations. This indicates a strategic push by major players to enhance their CDMO offerings and cater to evolving market needs.
Strategic European Pharmaceutical Contract Manufacturing Industry Market Outlook
The strategic outlook for the European pharmaceutical contract manufacturing industry is exceptionally positive, driven by sustained demand for outsourced manufacturing services and the ongoing innovation in pharmaceutical R&D. Growth accelerators include the expanding market for biologics and cell and gene therapies, requiring highly specialized CDMO capabilities. Investments in advanced manufacturing technologies, such as digitalization and automation, will further enhance efficiency and competitiveness. Strategic opportunities lie in developing specialized service offerings for niche therapeutic areas and expanding into emerging markets. The increasing trend of pharmaceutical companies focusing on their core competencies, coupled with the need for cost-effective and high-quality manufacturing solutions, positions European CDMOs for significant expansion and value creation in the coming years.
European Pharmaceutical Contract Manufacturing Industry Segmentation
-
1. Service Type
- 1.1. Active P
-
1.2. Finished
- 1.2.1. Solid Dose Formulation
- 1.2.2. Liquid Dose Formulation
- 1.2.3. Injectable Dose Formulation
- 1.3. Secondary Packaging
European Pharmaceutical Contract Manufacturing Industry Segmentation By Geography
-
1. Europe
- 1.1. United Kingdom
- 1.2. Germany
- 1.3. France
- 1.4. Italy
- 1.5. Spain
- 1.6. Netherlands
- 1.7. Belgium
- 1.8. Sweden
- 1.9. Norway
- 1.10. Poland
- 1.11. Denmark
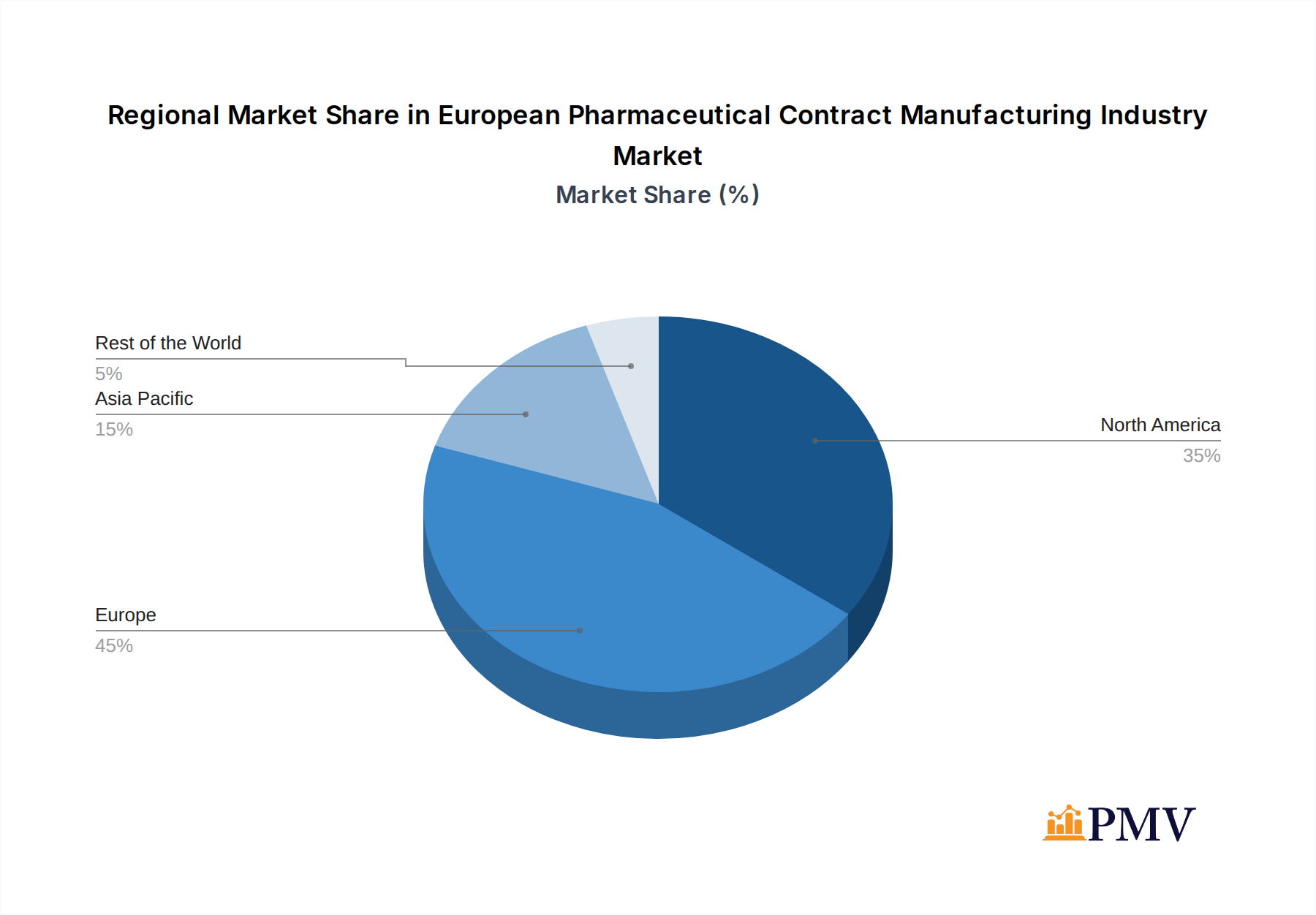
European Pharmaceutical Contract Manufacturing Industry Regional Market Share

Geographic Coverage of European Pharmaceutical Contract Manufacturing Industry
European Pharmaceutical Contract Manufacturing Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.6% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Service Type
- 5.1.1. Active P
- 5.1.2. Finished
- 5.1.2.1. Solid Dose Formulation
- 5.1.2.2. Liquid Dose Formulation
- 5.1.2.3. Injectable Dose Formulation
- 5.1.3. Secondary Packaging
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. Europe
- 5.1. Market Analysis, Insights and Forecast - by Service Type
- 6. European Pharmaceutical Contract Manufacturing Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Service Type
- 6.1.1. Active P
- 6.1.2. Finished
- 6.1.2.1. Solid Dose Formulation
- 6.1.2.2. Liquid Dose Formulation
- 6.1.2.3. Injectable Dose Formulation
- 6.1.3. Secondary Packaging
- 6.1. Market Analysis, Insights and Forecast - by Service Type
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Fareva Holdings SA
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Famar SA
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Cenexi - Laboratoires Thissen SA
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Almac Group*List Not Exhaustive
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Lonza Group
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Aenova Group
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Boehringer Ingelheim Group
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Recipharm AB
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.1 Fareva Holdings SA
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: European Pharmaceutical Contract Manufacturing Industry Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: European Pharmaceutical Contract Manufacturing Industry Share (%) by Company 2025
List of Tables
- Table 1: European Pharmaceutical Contract Manufacturing Industry Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 2: European Pharmaceutical Contract Manufacturing Industry Revenue undefined Forecast, by Region 2020 & 2033
- Table 3: European Pharmaceutical Contract Manufacturing Industry Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 4: European Pharmaceutical Contract Manufacturing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 5: United Kingdom European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 6: Germany European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 7: France European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Italy European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Spain European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Netherlands European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 11: Belgium European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 12: Sweden European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 13: Norway European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Poland European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Denmark European Pharmaceutical Contract Manufacturing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the European Pharmaceutical Contract Manufacturing Industry?
The projected CAGR is approximately 8.6%.
2. Which companies are prominent players in the European Pharmaceutical Contract Manufacturing Industry?
Key companies in the market include Fareva Holdings SA, Famar SA, Cenexi - Laboratoires Thissen SA, Almac Group*List Not Exhaustive, Lonza Group, Aenova Group, Boehringer Ingelheim Group, Recipharm AB.
3. What are the main segments of the European Pharmaceutical Contract Manufacturing Industry?
The market segments include Service Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
Increasing Outsourcing Volume by Pharmaceutical Companies; Increasing Investment in R&D.
6. What are the notable trends driving market growth?
Rising Investment in R&D will Drive The Market Growth.
7. Are there any restraints impacting market growth?
Increasing Lead Time and Logistics Costs; Stringent Regulatory Requirements; Capacity Utilization Issues Affecting the Profitability of CMOs.
8. Can you provide examples of recent developments in the market?
March 2022: MorphoSys sacked US R&D to consolidate work in Germany, taking USD 254 million in charges. MorphoSys axed its early pipeline and U.S. R&D work that came with the USD 1.7 billion purchase of Constellation Pharmaceuticals, meaning a more than USD 250 million impairment charge as the German pharma shifted the focus home.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "European Pharmaceutical Contract Manufacturing Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the European Pharmaceutical Contract Manufacturing Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the European Pharmaceutical Contract Manufacturing Industry?
To stay informed about further developments, trends, and reports in the European Pharmaceutical Contract Manufacturing Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
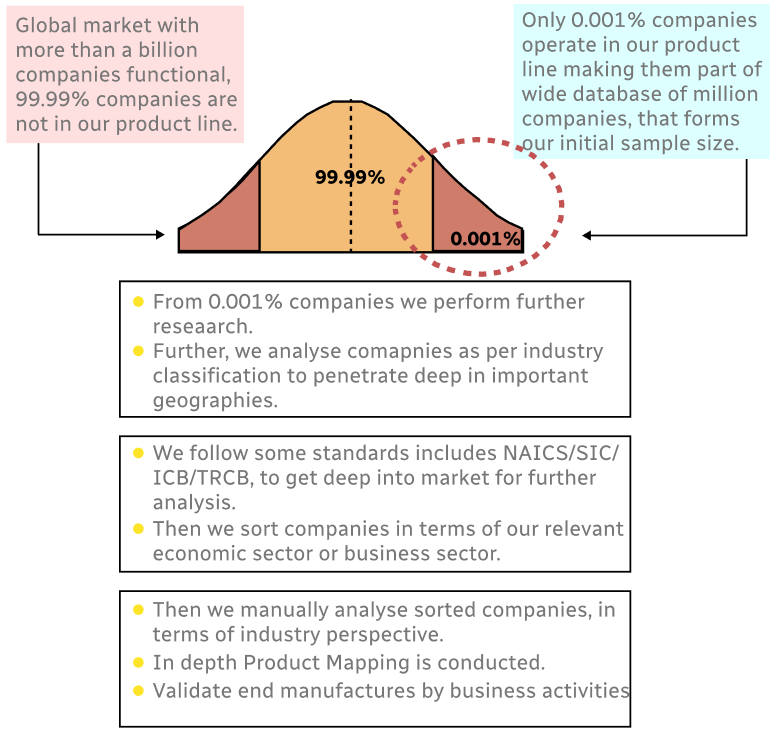
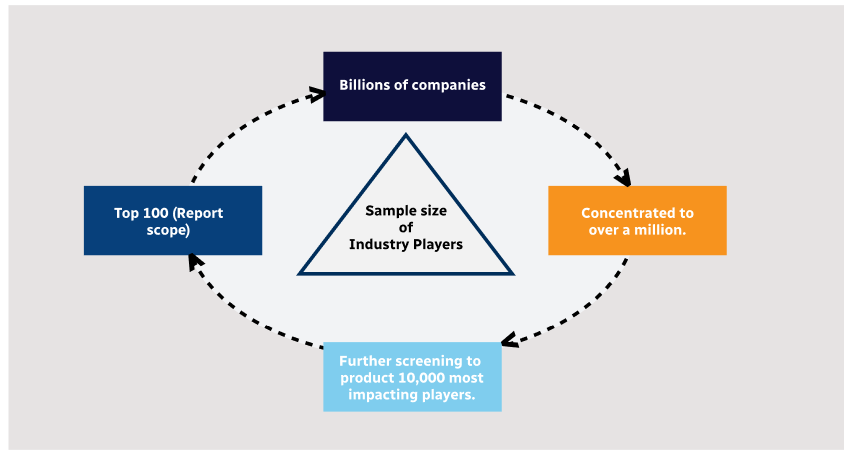
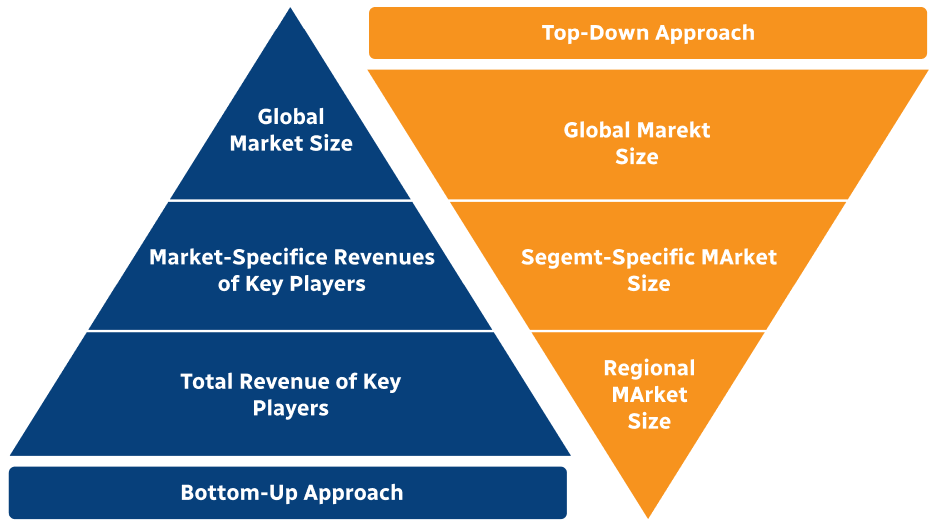
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


