Key Insights
The Chimeric Antigen Receptor (CAR) T-Cell Therapy market is poised for significant expansion, driven by its groundbreaking efficacy in treating hematological malignancies. With a projected market size of $4 billion in 2025, this advanced immunotherapy is rapidly becoming a cornerstone in oncology. The market is expected to witness robust growth, with a Compound Annual Growth Rate (CAGR) of 6.9% from 2025 through 2033. Key drivers fueling this expansion include an increasing incidence of blood cancers like Lymphoma and Multiple Myeloma, coupled with advancements in CAR T-cell engineering that enhance specificity and reduce side effects. The development of novel CAR constructs targeting specific antigens like CD19 and BCMA is a major trend, leading to more personalized and effective treatment regimens. Furthermore, growing investments in research and development by leading pharmaceutical and biotechnology companies, alongside supportive regulatory pathways, are accelerating the adoption of these complex yet life-saving therapies. The therapeutic landscape is continually evolving, with ongoing clinical trials exploring broader applications and combination strategies.
-T-Cell-Therapy.png)
Chimeric Antigen Receptor (CAR) T-Cell Therapy Market Size (In Billion)

Despite the promising outlook, certain restraints could temper the pace of growth. The high cost of CAR T-cell therapy, coupled with complex manufacturing processes and the need for specialized treatment centers, presents a significant barrier to widespread accessibility. Additionally, the risk of adverse events, such as cytokine release syndrome (CRS) and neurotoxicity, necessitates vigilant patient monitoring and management, contributing to operational complexities. However, ongoing research into mitigating these side effects and streamlining manufacturing is expected to alleviate these concerns over time. Emerging markets are also presenting new avenues for growth, as awareness and infrastructure for advanced cancer treatments improve. The competitive landscape is characterized by innovation and strategic collaborations, with established players and emerging biotech firms vying for market share by developing next-generation CAR T-cell products and expanding their therapeutic indications. The overall trajectory points towards a dynamic and rapidly evolving market, with substantial opportunities for innovation and improved patient outcomes.
-T-Cell-Therapy.png)
Chimeric Antigen Receptor (CAR) T-Cell Therapy Company Market Share

This in-depth report provides a detailed examination of the global Chimeric Antigen Receptor (CAR) T-Cell Therapy market, offering critical insights and actionable intelligence for stakeholders. Covering the period from 2019 to 2033, with a base and estimated year of 2025 and a forecast period of 2025-2033, this report analyzes market structure, competitive dynamics, industry trends, dominant segments, product innovations, key growth drivers, challenges, leading players, and future outlook. The report leverages a wealth of data, including market share estimates and M&A transaction values, providing a comprehensive understanding of this rapidly evolving therapeutic landscape.
Chimeric Antigen Receptor (CAR) T-Cell Therapy Market Structure & Competitive Dynamics
The Chimeric Antigen Receptor (CAR) T-Cell Therapy market exhibits a dynamic and evolving structure, characterized by significant innovation and increasing competition. Market concentration is moderate, with a few dominant players holding substantial market share, while numerous emerging companies are actively contributing to the innovation ecosystem. The regulatory frameworks, particularly those governed by agencies like the FDA and EMA, play a crucial role in shaping market entry and product approvals, influencing the pace of development and commercialization. The threat of product substitutes is currently low, given the unique efficacy of CAR T-cell therapies in treating certain hematological malignancies. End-user trends are heavily influenced by clinical trial outcomes, patient access, and reimbursement policies, driving demand for advanced and effective treatments. Mergers and acquisitions (M&A) activity is a significant feature, with deals valued in the billions of dollars reshaping the competitive landscape. For instance, strategic acquisitions of innovative biotech firms by larger pharmaceutical giants underscore the high stakes and potential returns in this segment. Notable M&A transactions are meticulously analyzed, providing a granular view of strategic consolidations and their impact on market share. The report quantures the competitive intensity, highlighting collaborations and strategic partnerships as key to navigating this complex market.
Chimeric Antigen Receptor (CAR) T-Cell Therapy Industry Trends & Insights
The Chimeric Antigen Receptor (CAR) T-Cell Therapy industry is experiencing unprecedented growth and technological advancement, driven by a confluence of factors. The market is poised for substantial expansion, with an estimated Compound Annual Growth Rate (CAGR) exceeding 20 billion during the forecast period. This robust growth is fueled by advancements in genetic engineering, cell culture techniques, and a deeper understanding of cancer immunology. The increasing prevalence of hematological malignancies, particularly lymphoma and multiple myeloma, presents a significant unmet medical need, driving the adoption of CAR T-cell therapies. Technological disruptions, such as the development of allogeneic CAR T-cells and novel targeting strategies, are further revolutionizing the field, promising broader patient access and potentially lower manufacturing costs. Consumer preferences are shifting towards personalized and highly effective treatments, with CAR T-cell therapy emerging as a leading option for refractory and relapsed cancers. Competitive dynamics are intensifying as companies vie for market leadership through pipeline expansion, strategic alliances, and intellectual property protection. Market penetration is projected to rise significantly as regulatory approvals expand and manufacturing capacities scale to meet growing demand. The report delves into the intricacies of these trends, providing a comprehensive overview of the forces shaping the future of CAR T-cell therapy, including insights into manufacturing scale-up, cost reduction initiatives, and the development of next-generation CAR T-cell constructs.
Dominant Markets & Segments in Chimeric Antigen Receptor (CAR) T-Cell Therapy
The Chimeric Antigen Receptor (CAR) T-Cell Therapy market is characterized by significant regional and segmental dominance, driven by a combination of healthcare infrastructure, patient demographics, and regulatory support.
North America currently stands as the leading region, with the United States spearheading market penetration due to its robust healthcare system, extensive research and development capabilities, and favorable reimbursement policies for advanced therapies. Strong economic policies supporting biopharmaceutical innovation and a high incidence of target malignancies contribute to its dominant position.
Europe follows closely, with key markets like Germany, the UK, and France demonstrating substantial growth, driven by increasing investments in advanced therapies and expanding patient access programs. Economic policies focused on healthcare innovation and collaborative research initiatives play a crucial role.
Asia Pacific is emerging as a rapidly growing market, with China and Japan showing significant potential. Government initiatives to boost domestic biopharmaceutical manufacturing, coupled with rising cancer incidence and improving healthcare infrastructure, are key drivers.
Within the Application segments:
Lymphoma represents the largest and most established segment. The approval of several CD19-targeted CAR T-cell therapies for various subtypes of lymphoma, such as diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma, has propelled its dominance. Factors like high remission rates and a well-defined patient population contribute to its market leadership.
Multiple Myeloma is a rapidly expanding segment, with BCMA-targeted CAR T-cell therapies demonstrating remarkable efficacy in relapsed and refractory patients. The significant unmet need and the development of novel BCMA-targeting constructs are fueling its growth.
Regarding Types of CAR T-cell therapies:
CD19-targeted therapies continue to dominate the market, primarily due to their established efficacy in B-cell malignancies like lymphoma. The extensive clinical data and widespread adoption of approved CD19 CAR T-cell products underscore their current market leadership.
BCMA-targeted therapies are experiencing exponential growth, driven by their groundbreaking success in multiple myeloma. The ongoing development and approval of new BCMA-targeted CAR T-cell therapies are positioning this segment for significant future expansion.
Chimeric Antigen Receptor (CAR) T-Cell Therapy Product Innovations
Product innovation in Chimeric Antigen Receptor (CAR) T-Cell Therapy is focused on enhancing efficacy, expanding applicability, and improving safety profiles. Companies are developing next-generation CAR T-cell therapies with improved persistence, reduced on-target off-tumor toxicities, and enhanced ability to overcome tumor resistance mechanisms. Innovations include multi-specific CARs targeting multiple antigens simultaneously, "armored" CARs engineered for enhanced cytokine production, and allogeneic CAR T-cells offering off-the-shelf accessibility. These advancements aim to broaden patient eligibility, improve treatment outcomes, and streamline manufacturing processes, addressing critical unmet needs in oncology.
Report Segmentation & Scope
This report meticulously segments the Chimeric Antigen Receptor (CAR) T-Cell Therapy market based on key criteria to provide granular insights. The Application segment is divided into Lymphoma and Multiple Myeloma, each analyzed for market size, growth projections, and competitive dynamics. The Type segment differentiates between CD19-targeted and BCMA-targeted therapies, detailing their respective market shares, development pipelines, and clinical trial landscapes. The scope encompasses a detailed analysis of the global market, with regional breakdowns and forecasts to 2033.
The Lymphoma segment is projected to continue its strong growth trajectory, driven by ongoing approvals for new indications and advancements in treatment strategies. Market size is estimated in the billions.
The Multiple Myeloma segment is expected to witness a CAGR exceeding 30 billion, fueled by promising clinical data for novel BCMA-targeted therapies.
CD19-targeted therapies, while mature, will continue to hold a significant market share, with ongoing research into new applications and combinations.
BCMA-targeted therapies are poised for substantial expansion, representing a key growth driver for the overall CAR T-cell therapy market. Market size is projected to reach billions.
Key Drivers of Chimeric Antigen Receptor (CAR) T-Cell Therapy Growth
The rapid expansion of the Chimeric Antigen Receptor (CAR) T-Cell Therapy market is propelled by several interconnected factors. Technological advancements in gene editing and cell engineering are enabling the development of more potent and targeted therapies. The increasing global burden of hematological malignancies, particularly lymphoma and multiple myeloma, creates a substantial unmet medical need, driving demand. Favorable regulatory pathways and accelerated approval processes for innovative therapies expedite market entry. Furthermore, growing investments in research and development from both pharmaceutical giants and venture capital firms are fueling pipeline advancements. The demonstration of high remission rates and durable responses in previously intractable cancers is a primary driver of clinical adoption and patient access.
Challenges in the Chimeric Antigen Receptor (CAR) T-Cell Therapy Sector
Despite its immense promise, the Chimeric Antigen Receptor (CAR) T-Cell Therapy sector faces significant challenges. High manufacturing costs and complex logistical requirements contribute to the exorbitant price of these therapies, posing a barrier to widespread patient access and reimbursement. Managing severe side effects, such as cytokine release syndrome (CRS) and neurotoxicity, requires specialized clinical expertise and infrastructure. The limited availability of CAR T-cell manufacturing capacity and the lengthy lead times for personalized cell production are also critical constraints. Moreover, the emergence of resistance mechanisms in patients and the identification of new tumor targets for broader applicability remain ongoing areas of research and development. Navigating stringent regulatory hurdles for new indications and geographical expansions also presents a challenge, demanding significant time and investment.
Leading Players in the Chimeric Antigen Receptor (CAR) T-Cell Therapy Market
The Chimeric Antigen Receptor (CAR) T-Cell Therapy market is populated by a mix of established pharmaceutical giants and innovative biotechnology companies. Key players include:
- Novartis
- Gilead Sciences
- Bristol-Myers Squibb
- J & J
- JW Therapeutics
- FOSUNKite
- CARsgen Therapeutics
- Autolus Therapeutics
- Sorrento Therapeutics
- Mustang Bio
- Bluebird Bio
- Cellectis
- Allogene Therapeutics
- Celyad
Key Developments in Chimeric Antigen Receptor (CAR) T-Cell Therapy Sector
- 2021/2022: Approval of multiple CD19-targeted CAR T-cell therapies for various lymphoma subtypes, significantly expanding patient access.
- 2022/2023: Breakthrough clinical trial results for BCMA-targeted CAR T-cell therapies in relapsed/refractory multiple myeloma, leading to regulatory submissions and approvals.
- 2023/2024: Increased focus on developing allogeneic (off-the-shelf) CAR T-cell therapies to address manufacturing bottlenecks and improve accessibility.
- 2024: Expansion of CAR T-cell therapy approvals to include earlier lines of treatment for certain hematological malignancies.
- Ongoing: Significant investment in research and development of next-generation CAR T-cell constructs targeting novel antigens and overcoming resistance mechanisms.
Strategic Chimeric Antigen Receptor (CAR) T-Cell Therapy Market Outlook
The strategic outlook for the Chimeric Antigen Receptor (CAR) T-Cell Therapy market remains exceptionally bright, characterized by sustained innovation and expanding therapeutic applications. Future growth will be fueled by advancements in next-generation CAR T-cell designs, including allogeneic therapies and multi-antigen targeting, which promise to enhance efficacy and broaden patient eligibility. The penetration into earlier lines of therapy for hematological malignancies and the exploration of CAR T-cell therapy for solid tumors represent significant strategic opportunities. Strategic partnerships, mergers, and acquisitions are expected to continue as companies seek to consolidate their market positions and leverage complementary expertise. The increasing global regulatory approvals and the ongoing efforts to reduce manufacturing costs and improve patient accessibility will further accelerate market expansion, solidifying CAR T-cell therapy's position as a cornerstone of advanced cancer treatment.
Chimeric Antigen Receptor (CAR) T-Cell Therapy Segmentation
-
1. Application
- 1.1. Lymphoma
- 1.2. Multiple Myeloma
-
2. Types
- 2.1. CD19-targeted
- 2.2. BCMA-targeted
Chimeric Antigen Receptor (CAR) T-Cell Therapy Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
-T-Cell-Therapy.png)
Chimeric Antigen Receptor (CAR) T-Cell Therapy Regional Market Share

Geographic Coverage of Chimeric Antigen Receptor (CAR) T-Cell Therapy
Chimeric Antigen Receptor (CAR) T-Cell Therapy REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Lymphoma
- 5.1.2. Multiple Myeloma
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. CD19-targeted
- 5.2.2. BCMA-targeted
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Chimeric Antigen Receptor (CAR) T-Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Lymphoma
- 6.1.2. Multiple Myeloma
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. CD19-targeted
- 6.2.2. BCMA-targeted
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Lymphoma
- 7.1.2. Multiple Myeloma
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. CD19-targeted
- 7.2.2. BCMA-targeted
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Lymphoma
- 8.1.2. Multiple Myeloma
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. CD19-targeted
- 8.2.2. BCMA-targeted
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Lymphoma
- 9.1.2. Multiple Myeloma
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. CD19-targeted
- 9.2.2. BCMA-targeted
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Lymphoma
- 10.1.2. Multiple Myeloma
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. CD19-targeted
- 10.2.2. BCMA-targeted
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Novartis
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Gilead Sciences
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Bristol-Myers Squibb
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 J & J
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 JW Therapeutics
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 FOSUNKite
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 CARsgen Therapeutics
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Autolus Therapeutics
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Sorrento Therapeutics
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Mustang Bio
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Bluebird Bio
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Cellectis
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Allogene Therapeutics
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Celyad
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.1 Novartis
List of Figures
- Figure 1: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Chimeric Antigen Receptor (CAR) T-Cell Therapy Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Chimeric Antigen Receptor (CAR) T-Cell Therapy?
The projected CAGR is approximately 6.9%.
2. Which companies are prominent players in the Chimeric Antigen Receptor (CAR) T-Cell Therapy?
Key companies in the market include Novartis, Gilead Sciences, Bristol-Myers Squibb, J & J, JW Therapeutics, FOSUNKite, CARsgen Therapeutics, Autolus Therapeutics, Sorrento Therapeutics, Mustang Bio, Bluebird Bio, Cellectis, Allogene Therapeutics, Celyad.
3. What are the main segments of the Chimeric Antigen Receptor (CAR) T-Cell Therapy?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 4 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Chimeric Antigen Receptor (CAR) T-Cell Therapy," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Chimeric Antigen Receptor (CAR) T-Cell Therapy report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Chimeric Antigen Receptor (CAR) T-Cell Therapy?
To stay informed about further developments, trends, and reports in the Chimeric Antigen Receptor (CAR) T-Cell Therapy, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
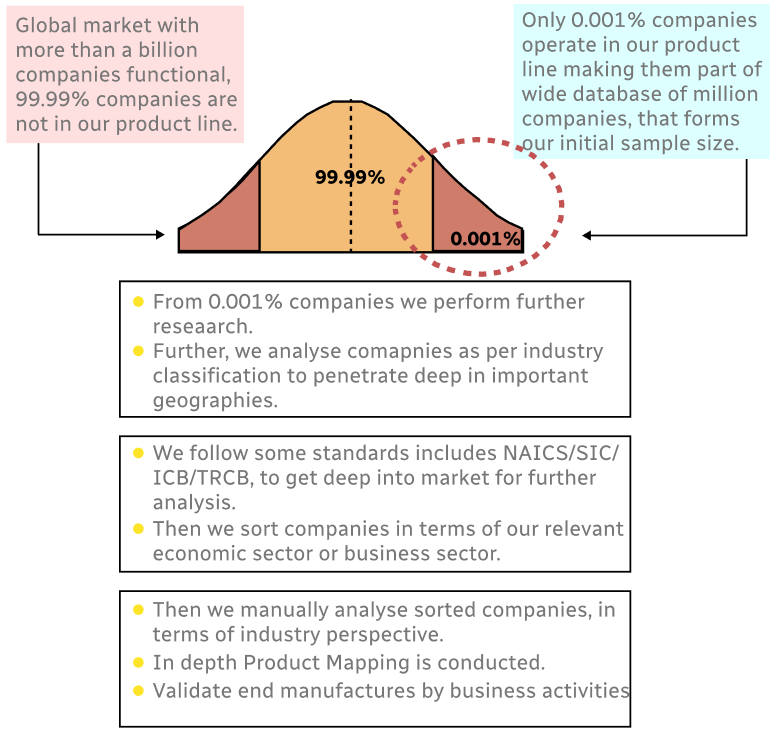
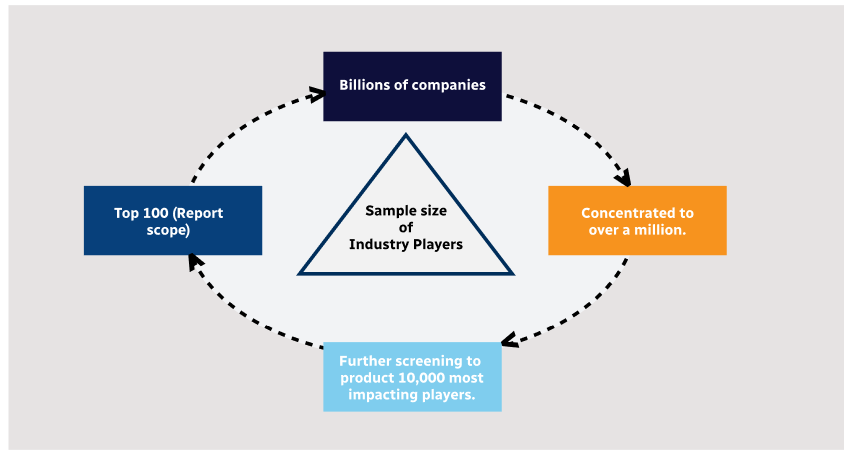
Step 1 - Identification of Relevant Samples Size from Population Database
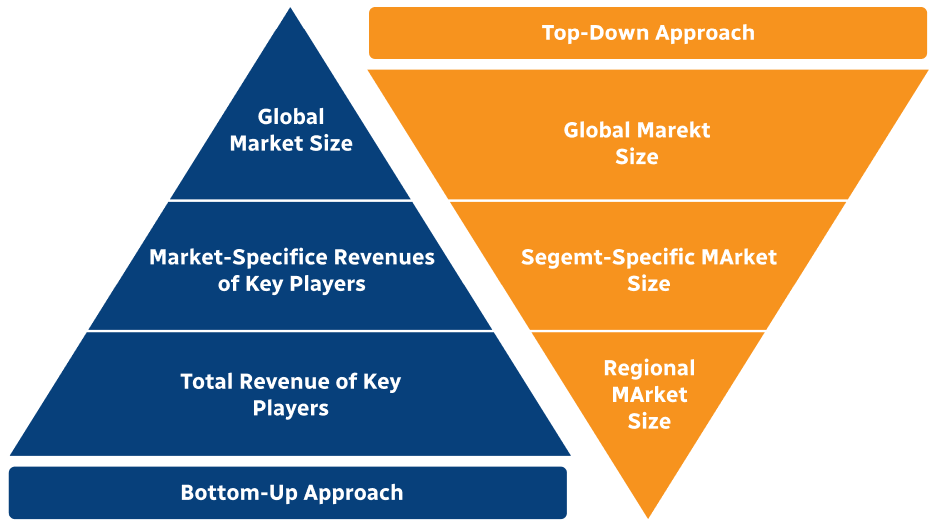
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


