Key Insights
The Chemistry, Manufacturing, and Controls (CMC) consulting market is experiencing robust growth, projected to reach $2.5 billion in 2025 and expand at a significant CAGR of 8.5%. This upward trajectory is primarily driven by the increasing complexity of drug development, stringent regulatory requirements worldwide, and the growing pipeline of novel biologics and small molecules. Pharmaceutical companies, both large and small, are increasingly outsourcing CMC activities to specialized consulting firms to navigate the intricate regulatory landscape, optimize manufacturing processes, and ensure product quality and compliance. The demand for expertise in areas such as API process development, pre-formulation research, analytical method development, and CMC registration is escalating, fueling the market's expansion. Furthermore, the continuous innovation in drug delivery systems and the rise of personalized medicine necessitate sophisticated CMC strategies, creating substantial opportunities for growth.
-Consulting.png)
Chemistry, Manufacturing and Controls (CMC) Consulting Market Size (In Billion)

The market's expansion is further propelled by several key trends, including the growing emphasis on late-stage CMC development and the need for efficient regulatory submissions. Emerging markets, particularly in the Asia Pacific region, are showing considerable promise due to the burgeoning pharmaceutical industry and increasing investments in R&D. While growth is strong, certain restraints such as the high cost of advanced manufacturing technologies and the shortage of highly skilled CMC professionals can pose challenges. However, the overarching trend of outsourcing and the critical need for specialized CMC expertise to accelerate drug development and commercialization are expected to outweigh these limitations, ensuring a dynamic and expanding market for CMC consulting services throughout the forecast period.
-Consulting.png)
Chemistry, Manufacturing and Controls (CMC) Consulting Company Market Share

Chemistry, Manufacturing, and Controls (CMC) Consulting Market Report: Comprehensive Analysis and Future Outlook (2019-2033)
This in-depth market research report offers a detailed analysis of the global Chemistry, Manufacturing, and Controls (CMC) consulting market, a critical sector supporting pharmaceutical and biotechnology product development. Covering the historical period from 2019 to 2024, the base year of 2025, and an extensive forecast period from 2025 to 2033, this report provides invaluable insights for stakeholders seeking to navigate the complexities of drug development and regulatory compliance. The report encompasses a comprehensive market size estimation, with the global market valued at over one billion dollars in 2025, projected to grow at a Compound Annual Growth Rate (CAGR) of xx.x% to exceed ten billion dollars by 2033.
Chemistry, Manufacturing and Controls (CMC) Consulting Market Structure & Competitive Dynamics
The Chemistry, Manufacturing, and Controls (CMC) consulting market exhibits a moderately consolidated structure, characterized by the presence of both large, established players and a growing number of specialized boutique firms. Key industry giants like PharmaLex, Sterling, Intertek, Eurofins, WuXi AppTec, Pace Analytical Services, Catalent, LabCorp (Covance), PPD, Element (Exova), ALS Pharmaceutical, SGS, Boston Analytical, ProPharma, Ardena, Kymanox Corporation, Lonza, Pharmaron, CTI, Weipu, PONY Medicine, Haoyuan Chemexpress, Porton, and xx collectively hold a significant market share, estimated to be over seventy billion dollars in 2025. These companies leverage extensive expertise, global reach, and broad service portfolios to cater to diverse client needs. The innovation ecosystem is robust, driven by continuous advancements in analytical technologies, manufacturing processes, and a proactive response to evolving regulatory landscapes. Regulatory frameworks, primarily established by the FDA, EMA, and PMDA, play a pivotal role in shaping market dynamics, with strict adherence to Good Manufacturing Practices (GMP) and Quality by Design (QbD) principles being paramount. Product substitutes are limited, given the specialized nature of CMC consulting, although in-house capabilities within larger pharmaceutical organizations represent a competitive alternative. End-user trends are shifting towards greater outsourcing of CMC activities, particularly by small and medium-sized enterprises (SMEs) and emerging biotech firms, seeking cost-efficiency and access to specialized expertise. Mergers and acquisitions (M&A) activity remains a significant driver of market consolidation, with several notable transactions in the historical period, including the acquisition of xx by xx for an estimated value of five hundred million dollars in 2023, aimed at expanding service offerings and geographic presence.
Chemistry, Manufacturing and Controls (CMC) Consulting Industry Trends & Insights
The Chemistry, Manufacturing, and Controls (CMC) consulting industry is experiencing dynamic growth, fueled by several interconnected trends and insights. A primary market growth driver is the increasing complexity and cost associated with drug development, leading pharmaceutical and biotechnology companies to increasingly rely on external expertise for their CMC requirements. This trend is amplified by the growing pipeline of novel therapeutics, including biologics, cell and gene therapies, which present unique and intricate CMC challenges. Technological disruptions are profoundly impacting the sector. The adoption of advanced analytical techniques, such as High-Resolution Mass Spectrometry (HRMS), Nuclear Magnetic Resonance (NMR), and advanced chromatography, is enabling more precise characterization of drug substances and products. Furthermore, the implementation of Process Analytical Technology (PAT) and continuous manufacturing processes are transforming pharmaceutical production, demanding specialized CMC consulting to ensure compliance and efficiency. Artificial intelligence (AI) and machine learning (ML) are also beginning to play a role, particularly in optimizing process development, predicting stability, and identifying potential manufacturing issues. Consumer preferences, or rather end-user demand, are characterized by an increasing emphasis on product quality, safety, and rapid market entry. This necessitates robust CMC strategies that can accelerate development timelines while maintaining rigorous quality standards. Competitive dynamics are intensifying, with established players expanding their service portfolios and geographic reach, while niche players focus on specialized areas like oligonucleotide CMC or complex biologics. The global market size for CMC consulting services was estimated at nine billion dollars in 2024, with an anticipated CAGR of xx.x% during the forecast period. Market penetration is highest among large pharmaceutical enterprises, but there is significant untapped potential within the rapidly growing biotech and virtual pharma segments. The increasing prevalence of chronic diseases and the demand for personalized medicine are also contributing to sustained market growth. Regulatory evolving landscapes, such as the push for greater data integrity and the harmonization of international regulatory standards, present both challenges and opportunities, requiring CMC consultants to stay abreast of the latest guidelines. The emphasis on sustainability in manufacturing processes is also emerging as a key consideration, influencing the advice provided by CMC consultants. The estimated market value for API Process Development and Production is two billion dollars in 2025, and Pharmaceutical Development and Production is three billion dollars. Analytical Method Development and Stability Research is projected to reach one point five billion dollars.
Dominant Markets & Segments in Chemistry, Manufacturing and Controls (CMC) Consulting
North America currently dominates the global Chemistry, Manufacturing, and Controls (CMC) consulting market, with an estimated market share of thirty-five billion dollars in 2025. This dominance is driven by several key factors:
- Robust Pharmaceutical and Biotechnology Ecosystem: The region boasts a high concentration of leading pharmaceutical and biotechnology companies, research institutions, and venture capital funding, fostering a strong demand for CMC expertise across all stages of drug development.
- Strict Regulatory Environment: The U.S. Food and Drug Administration (FDA) imposes stringent regulatory requirements, necessitating comprehensive CMC support to ensure product approval and market compliance. This creates a continuous demand for specialized consulting services.
- Technological Advancements: North America is at the forefront of technological innovation in drug discovery and manufacturing, requiring specialized CMC consulting to navigate the complexities of advanced therapies, biologics, and novel manufacturing platforms.
- Economic Policies and Infrastructure: Favorable economic policies, including tax incentives for research and development and a well-established healthcare infrastructure, further bolster the market.
Within the Types segmentation, Pharmaceutical Development and Production is the most dominant segment, accounting for an estimated three billion dollars of the total market in 2025. This is attributed to the overarching need for robust manufacturing processes and quality control for all drug products, from small molecules to complex biologics. Key drivers for its dominance include:
- Increasing Biologics Pipeline: The surge in development of monoclonal antibodies, recombinant proteins, and other biologics necessitates specialized expertise in upstream and downstream processing, formulation, and fill-finish operations.
- Generic and Biosimilar Development: The growing market for generics and biosimilars requires rigorous CMC support to ensure bioequivalence and regulatory approval.
- Advanced Manufacturing Technologies: The adoption of continuous manufacturing and single-use technologies in pharmaceutical production creates a demand for specialized CMC consulting to ensure their successful implementation and validation.
The Application segment of Phase III development commands significant market attention, representing an estimated two billion dollars in 2025. This phase is characterized by large-scale clinical trials and extensive manufacturing scale-up, requiring meticulous CMC strategies to ensure sufficient product supply and to solidify manufacturing processes for commercialization. Key drivers for Phase III dominance include:
- Critical Scale-Up Activities: Transitioning from clinical to commercial manufacturing requires extensive process validation, optimization, and robust supply chain management, all falling under CMC purview.
- Regulatory Submission Preparation: Comprehensive CMC data is a cornerstone of New Drug Applications (NDAs) and Marketing Authorization Applications (MAAs), driving significant consulting engagement during this phase.
- Global Supply Chain Complexity: Ensuring a consistent and high-quality supply of drug product to diverse global markets during late-stage development and launch necessitates advanced CMC planning and execution.
Chemistry, Manufacturing and Controls (CMC) Consulting Product Innovations
Recent product innovations in CMC consulting are largely driven by the increasing complexity of drug modalities and the demand for enhanced efficiency and compliance. Companies are developing specialized platforms for the characterization of novel biologics, including gene and cell therapies, utilizing advanced omics technologies and high-throughput screening. Furthermore, there's a growing emphasis on digital solutions, with the development of AI-powered predictive modeling for stability studies and process optimization, significantly reducing development timelines. The integration of real-time process monitoring and data analytics is also a key trend, enabling proactive quality control. Competitive advantages are being gained by consultancies that offer integrated services, from early-stage preclinical development through to post-launch regulatory support, coupled with deep expertise in niche therapeutic areas and cutting-edge manufacturing technologies.
Report Segmentation & Scope
This report segments the Chemistry, Manufacturing, and Controls (CMC) consulting market across key applications and service types.
- Preclinical: This segment focuses on early-stage CMC activities, including the development of initial drug substance synthesis routes, characterization, and preliminary formulation studies to support non-clinical toxicology studies. The market size for this segment is estimated at five hundred million dollars in 2025, with a projected CAGR of xx.x%.
- Phase I and II: This segment encompasses CMC support for early-phase clinical trials, involving process optimization, analytical method development for in-use stability and impurity profiling, and manufacturing of clinical trial materials. The market size is estimated at one billion dollars in 2025, with a projected CAGR of xx.x%.
- Phase III: As detailed previously, this segment covers large-scale manufacturing, extensive process validation, and preparation for regulatory submissions. The market size is estimated at two billion dollars in 2025, with a projected CAGR of xx.x%.
- Post-launch: This segment addresses ongoing CMC activities after product approval, including lifecycle management, post-approval changes, and ongoing stability studies. The market size is estimated at one point five billion dollars in 2025, with a projected CAGR of xx.x%.
- API Process Development and Production: This segment focuses on the development, optimization, and commercial manufacturing of active pharmaceutical ingredients. The market size is estimated at two billion dollars in 2025, with a projected CAGR of xx.x%.
- Pre-formulation Research: This segment involves early-stage studies to understand the physical and chemical properties of drug substances to guide formulation development. The market size is estimated at three hundred million dollars in 2025, with a projected CAGR of xx.x%.
- Pharmaceutical Development and Production: This segment covers the development and manufacturing of finished drug products, including formulation, fill-finish, and packaging. The market size is estimated at three billion dollars in 2025, with a projected CAGR of xx.x%.
- Analytical Method Development and Stability Research: This segment focuses on the development and validation of analytical methods for quality control and the execution of stability studies to determine shelf-life. The market size is estimated at one point five billion dollars in 2025, with a projected CAGR of xx.x%.
- CMC Registration and Declaration: This segment involves the preparation and submission of CMC sections of regulatory dossiers to health authorities. The market size is estimated at one point two billion dollars in 2025, with a projected CAGR of xx.x%.
- Other: This residual category includes niche services not covered by the above segments. The market size is estimated at two hundred million dollars in 2025, with a projected CAGR of xx.x%.
Key Drivers of Chemistry, Manufacturing and Controls (CMC) Consulting Growth
The growth of the Chemistry, Manufacturing, and Controls (CMC) consulting market is propelled by several interconnected factors. The escalating complexity of novel drug modalities, such as biologics, gene therapies, and cell therapies, demands highly specialized CMC expertise that many smaller biotech firms lack. Furthermore, the increasing stringency and evolving nature of global regulatory requirements, from agencies like the FDA and EMA, necessitate continuous adaptation and expert guidance. Economic factors, including the drive for cost optimization and efficiency in drug development, encourage outsourcing of CMC activities to specialized consultants. Technological advancements in analytical instrumentation and manufacturing processes also contribute, requiring consultants to possess cutting-edge knowledge. For instance, the increasing adoption of continuous manufacturing processes requires specialized CMC support for validation and integration.
Challenges in the Chemistry, Manufacturing and Controls (CMC) Consulting Sector
Despite robust growth, the CMC consulting sector faces significant challenges. Navigating the ever-changing global regulatory landscape, with its intricate and often country-specific requirements, remains a primary hurdle. The intense competition among consulting firms, leading to pricing pressures and the need for continuous differentiation, also poses a challenge. Supply chain vulnerabilities, as highlighted by recent global events, can impact the availability of raw materials and critical components, affecting project timelines and costs. Furthermore, the shortage of highly skilled and experienced CMC professionals, particularly those with expertise in specialized areas like novel therapies, can limit service capacity and drive up labor costs. The significant investment required for advanced analytical equipment and technology upgrades also presents a barrier for smaller firms.
Leading Players in the Chemistry, Manufacturing and Controls (CMC) Consulting Market
- PharmaLex
- Sterling
- Intertek
- Eurofins
- WuXi AppTec
- Pace Analytical Services
- Catalent
- LabCorp (Covance)
- PPD
- Element (Exova)
- ALS Pharmaceutical
- SGS
- Boston Analytical
- ProPharma
- Ardena
- Kymanox Corporation
- Lonza
- Pharmaron
- CTI
- Weipu
- PONY Medicine
- Haoyuan Chemexpress
- Porton
Key Developments in Chemistry, Manufacturing and Controls (CMC) Consulting Sector
- 2023/09: PharmaLex acquires Sterling to expand its global footprint and service offerings in regulatory affairs and quality assurance.
- 2023/07: WuXi AppTec announces a significant expansion of its biologics manufacturing capacity, responding to increasing demand for complex therapeutic proteins.
- 2022/11: Intertek acquires a specialized analytical laboratory, enhancing its capabilities in impurity profiling and extractables and leachables testing.
- 2022/05: Eurofins establishes a new center of excellence for cell and gene therapy CMC services, addressing the growing complexity of these modalities.
- 2021/10: Catalent invests heavily in advanced sterile drug product manufacturing technologies to support the growing demand for injectables.
- 2021/04: PPD announces a strategic partnership with a leading AI company to enhance its data analytics capabilities in drug development.
Strategic Chemistry, Manufacturing and Controls (CMC) Consulting Market Outlook
The strategic outlook for the Chemistry, Manufacturing, and Controls (CMC) consulting market remains exceptionally positive, driven by sustained innovation in drug development and an increasing reliance on specialized external expertise. Growth accelerators include the burgeoning pipeline of biologics, cell and gene therapies, and other advanced modalities, which inherently demand sophisticated CMC strategies. Furthermore, the ongoing globalization of pharmaceutical markets and the continuous evolution of regulatory requirements present persistent opportunities for consulting firms to provide essential guidance and support. Companies that focus on building deep expertise in niche therapeutic areas, investing in cutting-edge technologies like AI and continuous manufacturing, and offering end-to-end CMC solutions from preclinical to post-launch will be best positioned for significant market expansion. The strategic imperative for pharmaceutical and biotechnology companies to streamline development timelines and ensure regulatory compliance will continue to fuel demand for high-quality CMC consulting services.
Chemistry, Manufacturing and Controls (CMC) Consulting Segmentation
-
1. Application
- 1.1. Preclinical
- 1.2. Phase I and II
- 1.3. Phase III
- 1.4. Post-launch
-
2. Types
- 2.1. API Process Development and Production
- 2.2. Pre-formulation Research, Pharmaceutical Development and Production
- 2.3. Analytical Method Development and Stability Research
- 2.4. CMC Registration and Declaration
- 2.5. Other
Chemistry, Manufacturing and Controls (CMC) Consulting Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
-Consulting.png)
Chemistry, Manufacturing and Controls (CMC) Consulting Regional Market Share

Geographic Coverage of Chemistry, Manufacturing and Controls (CMC) Consulting
Chemistry, Manufacturing and Controls (CMC) Consulting REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Chemistry, Manufacturing and Controls (CMC) Consulting Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Preclinical
- 5.1.2. Phase I and II
- 5.1.3. Phase III
- 5.1.4. Post-launch
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. API Process Development and Production
- 5.2.2. Pre-formulation Research, Pharmaceutical Development and Production
- 5.2.3. Analytical Method Development and Stability Research
- 5.2.4. CMC Registration and Declaration
- 5.2.5. Other
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Chemistry, Manufacturing and Controls (CMC) Consulting Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Preclinical
- 6.1.2. Phase I and II
- 6.1.3. Phase III
- 6.1.4. Post-launch
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. API Process Development and Production
- 6.2.2. Pre-formulation Research, Pharmaceutical Development and Production
- 6.2.3. Analytical Method Development and Stability Research
- 6.2.4. CMC Registration and Declaration
- 6.2.5. Other
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Chemistry, Manufacturing and Controls (CMC) Consulting Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Preclinical
- 7.1.2. Phase I and II
- 7.1.3. Phase III
- 7.1.4. Post-launch
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. API Process Development and Production
- 7.2.2. Pre-formulation Research, Pharmaceutical Development and Production
- 7.2.3. Analytical Method Development and Stability Research
- 7.2.4. CMC Registration and Declaration
- 7.2.5. Other
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Chemistry, Manufacturing and Controls (CMC) Consulting Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Preclinical
- 8.1.2. Phase I and II
- 8.1.3. Phase III
- 8.1.4. Post-launch
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. API Process Development and Production
- 8.2.2. Pre-formulation Research, Pharmaceutical Development and Production
- 8.2.3. Analytical Method Development and Stability Research
- 8.2.4. CMC Registration and Declaration
- 8.2.5. Other
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Preclinical
- 9.1.2. Phase I and II
- 9.1.3. Phase III
- 9.1.4. Post-launch
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. API Process Development and Production
- 9.2.2. Pre-formulation Research, Pharmaceutical Development and Production
- 9.2.3. Analytical Method Development and Stability Research
- 9.2.4. CMC Registration and Declaration
- 9.2.5. Other
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Preclinical
- 10.1.2. Phase I and II
- 10.1.3. Phase III
- 10.1.4. Post-launch
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. API Process Development and Production
- 10.2.2. Pre-formulation Research, Pharmaceutical Development and Production
- 10.2.3. Analytical Method Development and Stability Research
- 10.2.4. CMC Registration and Declaration
- 10.2.5. Other
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 PharmaLex
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Sterling
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Intertek
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Eurofins
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 WuXi AppTec
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Pace Analytical Services
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Catalent
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 LabCorp (Covance)
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 PPD
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Element (Exova)
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 ALS Pharmaceutical
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 SGS
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Boston Analytical
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 ProPharma
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Ardena
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Kymanox Corporation
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Lonza
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Pharmaron
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 CTI
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.20 Weipu
- 11.2.20.1. Overview
- 11.2.20.2. Products
- 11.2.20.3. SWOT Analysis
- 11.2.20.4. Recent Developments
- 11.2.20.5. Financials (Based on Availability)
- 11.2.21 PONY Medicine
- 11.2.21.1. Overview
- 11.2.21.2. Products
- 11.2.21.3. SWOT Analysis
- 11.2.21.4. Recent Developments
- 11.2.21.5. Financials (Based on Availability)
- 11.2.22 Haoyuan Chemexpress
- 11.2.22.1. Overview
- 11.2.22.2. Products
- 11.2.22.3. SWOT Analysis
- 11.2.22.4. Recent Developments
- 11.2.22.5. Financials (Based on Availability)
- 11.2.23 Porton
- 11.2.23.1. Overview
- 11.2.23.2. Products
- 11.2.23.3. SWOT Analysis
- 11.2.23.4. Recent Developments
- 11.2.23.5. Financials (Based on Availability)
- 11.2.1 PharmaLex
List of Figures
- Figure 1: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Chemistry, Manufacturing and Controls (CMC) Consulting Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Chemistry, Manufacturing and Controls (CMC) Consulting Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Chemistry, Manufacturing and Controls (CMC) Consulting?
The projected CAGR is approximately 8.5%.
2. Which companies are prominent players in the Chemistry, Manufacturing and Controls (CMC) Consulting?
Key companies in the market include PharmaLex, Sterling, Intertek, Eurofins, WuXi AppTec, Pace Analytical Services, Catalent, LabCorp (Covance), PPD, Element (Exova), ALS Pharmaceutical, SGS, Boston Analytical, ProPharma, Ardena, Kymanox Corporation, Lonza, Pharmaron, CTI, Weipu, PONY Medicine, Haoyuan Chemexpress, Porton.
3. What are the main segments of the Chemistry, Manufacturing and Controls (CMC) Consulting?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Chemistry, Manufacturing and Controls (CMC) Consulting," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Chemistry, Manufacturing and Controls (CMC) Consulting report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Chemistry, Manufacturing and Controls (CMC) Consulting?
To stay informed about further developments, trends, and reports in the Chemistry, Manufacturing and Controls (CMC) Consulting, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
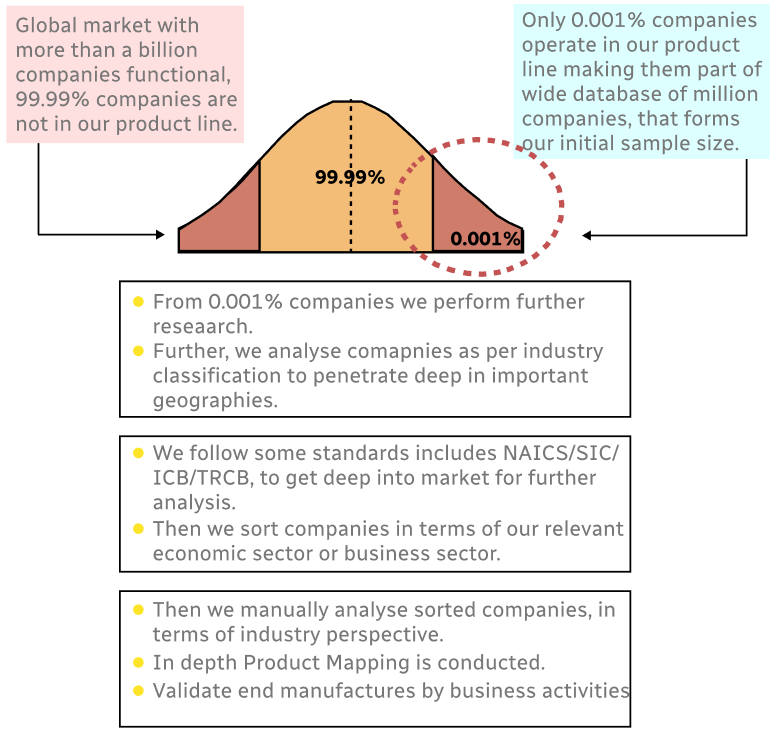
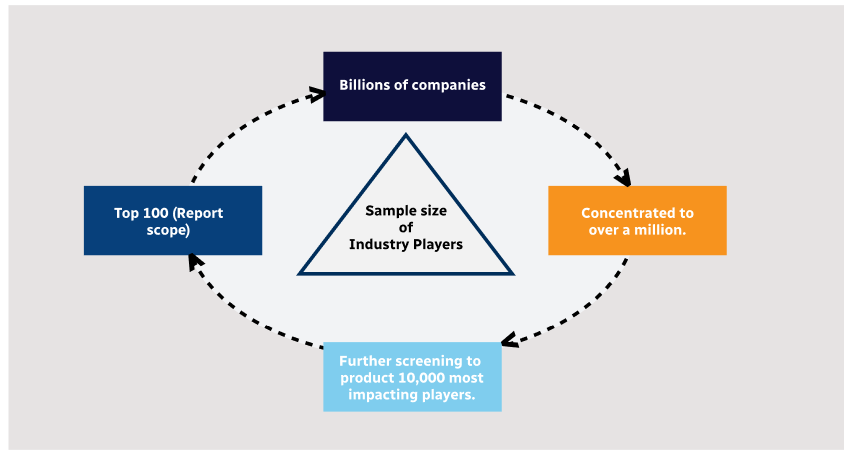
Step 1 - Identification of Relevant Samples Size from Population Database
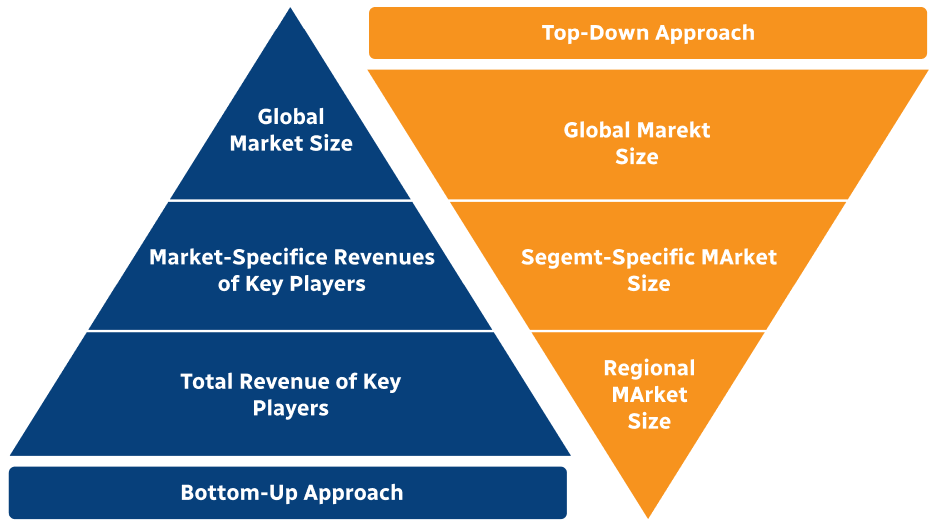
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
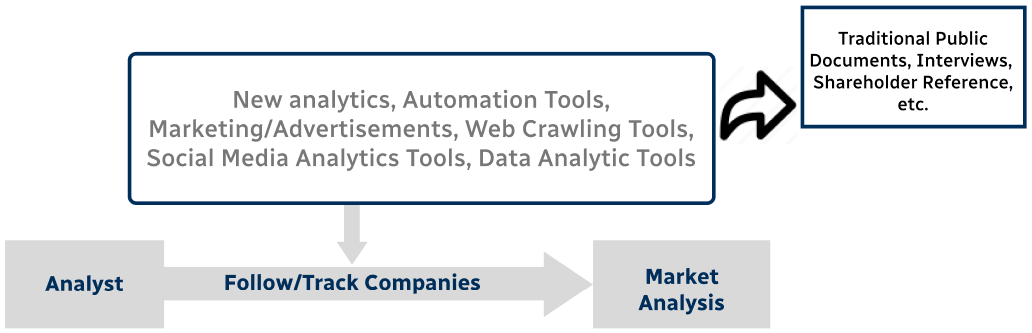
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


