Key Insights
The Canadian in-vitro diagnostics (IVD) market is poised for robust growth, projected to reach an estimated $54.08 billion by 2025. Driven by an aging population, increasing prevalence of chronic diseases such as diabetes, cancer, and cardiovascular conditions, and a heightened awareness of infectious disease outbreaks, the demand for accurate and timely diagnostic solutions is escalating. Technological advancements, including the rise of molecular diagnostics and the integration of artificial intelligence in laboratory automation, are further fueling market expansion. The shift towards personalized medicine and companion diagnostics also presents a significant opportunity, encouraging innovation in high-sensitivity and specific testing. Furthermore, government initiatives and increased healthcare expenditure in Canada are supporting the adoption of advanced IVD technologies, contributing to a projected Compound Annual Growth Rate (CAGR) of 5.23% during the forecast period of 2025-2033.

Canada in-vitro Diagnostics Market Market Size (In Billion)

The Canadian IVD market is characterized by a dynamic segmentation across various test types, products, usability, applications, and end-users. Clinical chemistry and immuno-diagnostics represent substantial segments, driven by routine testing and the diagnosis of a wide array of conditions. Molecular diagnostics is experiencing rapid growth due to its pivotal role in infectious disease detection, genetic testing, and oncology. In terms of products, reagents and instruments are the primary revenue generators, with a growing demand for advanced and automated systems. The usability segment sees a rising preference for disposable IVD devices, offering convenience and reducing cross-contamination risks, particularly in point-of-care settings. The application landscape is dominated by infectious diseases, diabetes, and cancer diagnostics, reflecting the major health burdens in Canada. Diagnostic laboratories and hospitals and clinics are the principal end-users, with an increasing number of decentralized testing sites and home-based diagnostic solutions emerging. Key players like Abbott Laboratories, Roche Diagnostics, Thermo Fisher Scientific, and Siemens Healthineers are actively investing in research and development, strategic partnerships, and market expansion within Canada to capture this growing market.

Canada in-vitro Diagnostics Market Company Market Share

Comprehensive Canada In-Vitro Diagnostics Market Report: Trends, Opportunities, and Forecasts (2019-2033)
This in-depth report offers an exhaustive analysis of the Canadian in-vitro diagnostics (IVD) market, providing critical insights for stakeholders. Covering the historical period from 2019-2024, the base year of 2025, and a comprehensive forecast period from 2025-2033, this research delves into market structure, competitive dynamics, emerging trends, dominant segments, product innovations, and strategic outlook. Leverage high-ranking keywords such as "Canada IVD market," "in-vitro diagnostics Canada," "clinical diagnostics Canada," "molecular diagnostics Canada," and "medical diagnostics market Canada" to gain a competitive edge. The market is projected to reach billions in value by 2033.
Canada In-Vitro Diagnostics Market Market Structure & Competitive Dynamics
The Canadian in-vitro diagnostics (IVD) market exhibits a moderately concentrated structure, with a significant portion of market share held by a few major international players, including Abbott Laboratories, Thermo Fisher Scientific Inc., F Hoffmann-La Roche AG, and Siemens AG. These dominant entities leverage robust R&D capabilities, extensive product portfolios, and strong distribution networks to maintain their competitive positions. Innovation ecosystems are driven by collaborative efforts between IVD manufacturers, academic institutions, and research centers, fostering the development of novel diagnostic solutions. The regulatory framework, primarily governed by Health Canada, plays a crucial role in ensuring product safety and efficacy, influencing market entry and product approvals. Product substitutes are evolving, particularly with the rise of point-of-care testing (POCT) and advanced analytical techniques that offer faster and more accurate results. End-user trends indicate a growing demand for personalized medicine, companion diagnostics, and real-time disease monitoring, pushing IVD manufacturers to develop more sophisticated and integrated diagnostic platforms. Mergers and acquisitions (M&A) activities remain a strategic imperative for market expansion and technological advancement. Key M&A deals have focused on acquiring niche technologies or expanding market reach within specific application areas, contributing to market consolidation. The overall market value is estimated to be in the billions.
Canada In-Vitro Diagnostics Market Industry Trends & Insights
The Canadian in-vitro diagnostics (IVD) market is experiencing robust growth, driven by several pivotal industry trends and insights. A significant growth driver is the increasing prevalence of chronic diseases such as diabetes, cancer, and cardiovascular conditions, necessitating early and accurate diagnosis through IVD tests. The growing aging population in Canada further amplifies the demand for diagnostic solutions to manage age-related health issues. Technological advancements are revolutionizing the IVD landscape. Molecular diagnostics, in particular, is witnessing an unprecedented surge due to its precision in identifying infectious agents, genetic predispositions, and cancer biomarkers. Innovations in next-generation sequencing (NGS), PCR-based assays, and liquid biopsies are transforming disease detection and management. The rise of automation and artificial intelligence (AI) in diagnostic laboratories is enhancing efficiency, reducing turnaround times, and improving diagnostic accuracy, leading to better patient outcomes. Consumer preferences are shifting towards personalized medicine, where IVD plays a crucial role in tailoring treatment plans based on individual genetic makeup and disease profiles. This trend is fueling the demand for companion diagnostics that predict patient response to specific therapies. The competitive dynamics are characterized by intense R&D investments and strategic partnerships aimed at developing innovative and cost-effective diagnostic solutions. The market penetration of advanced IVD technologies is steadily increasing, supported by favorable reimbursement policies and growing healthcare expenditure. The overall CAGR for the Canadian IVD market is projected to be strong throughout the forecast period. The market value is anticipated to reach several billions by the end of the study period.
Dominant Markets & Segments in Canada In-Vitro Diagnostics Market
The Canadian in-vitro diagnostics (IVD) market is segmented across various test types, products, usability, applications, and end-users, each exhibiting distinct growth trajectories and market dominance.
Test Type Dominance:
- Molecular Diagnostics: This segment stands out as a major growth engine. The increasing demand for genetic testing, infectious disease detection (especially post-pandemic), and oncology diagnostics fuels its dominance. Key drivers include rapid advancements in PCR and NGS technologies, along with the growing adoption of companion diagnostics.
- Immuno Diagnostics: This segment remains a cornerstone of the IVD market, driven by its extensive use in detecting infectious diseases, autoimmune disorders, and hormonal imbalances. Automation and the development of highly sensitive immunoassays continue to bolster its position.
- Clinical Chemistry: While a mature segment, clinical chemistry tests remain indispensable for routine health monitoring and the diagnosis of metabolic disorders. The widespread availability of automated analyzers and a broad test menu contribute to its sustained market presence.
- Hematology: This segment is crucial for diagnosing and monitoring blood disorders, anemias, and infections. Advancements in automated hematology analyzers with improved cell counting and classification capabilities are driving its growth.
- Other Tests: This category encompasses a diverse range of specialized diagnostic tests, including toxicology, urinalysis, and microbiology, which contribute to the overall market volume.
Product Dominance:
- Reagents: Reagents form the largest segment by value, as they are consumed with each IVD test performed. Their dominance is directly proportional to the volume of diagnostic tests conducted. The development of novel, highly specific, and sensitive reagents is a continuous focus for manufacturers.
- Instruments: Sophisticated IVD instruments, such as automated analyzers and sequencers, are essential for performing complex diagnostic tests. Their initial cost is high, but they enable high-throughput testing and are critical for modern diagnostic laboratories and hospitals.
- Other Products: This includes consumables, software, and accessories that support IVD testing, contributing to the overall market infrastructure.
Usability Dominance:
- Disposable IVD Devices: The convenience, sterility, and reduced risk of contamination associated with disposable devices make them highly prevalent, particularly in point-of-care settings and for rapid diagnostic tests.
- Reusable IVD Devices: While requiring sterilization and maintenance, reusable instruments offer a lower cost per test in high-volume laboratory settings, contributing significantly to the market.
Application Dominance:
- Infectious Disease: This application segment has witnessed exponential growth, particularly in the wake of recent global health crises. Rapid diagnostic tests and molecular assays for identifying a wide array of pathogens are in high demand.
- Cancer/Oncology: The increasing incidence of cancer and the growing emphasis on early detection, diagnosis, and personalized treatment drive the demand for advanced IVD solutions in oncology. Companion diagnostics are a key growth driver here.
- Diabetes: With the rising global burden of diabetes, IVD tests for blood glucose monitoring, HbA1c measurement, and early detection of complications remain a significant market segment.
- Cardiology: IVD plays a vital role in the diagnosis and management of cardiovascular diseases through the detection of biomarkers for heart attacks, arrhythmias, and other cardiac conditions.
- Autoimmune Disease: The increasing understanding and diagnosis of autoimmune disorders are fueling the demand for specific immunoassays and molecular tests.
- Nephrology: IVD is crucial for monitoring kidney function and diagnosing kidney diseases.
- Other Applications: This broad category includes diagnostics for neurological disorders, gastrointestinal diseases, and other specialized medical fields.
End User Dominance:
- Diagnostic Laboratories: These independent and hospital-affiliated laboratories are the primary consumers of IVD products and services, owing to their high volume of testing.
- Hospitals and Clinics: In-house laboratories within hospitals and clinics also represent a substantial end-user segment, particularly for emergency and critical care diagnostics.
- Other End Users: This includes research institutions, academic centers, and government agencies that utilize IVD technologies for research and public health initiatives.
The economic policies promoting healthcare infrastructure development, coupled with advancements in medical technology, are key drivers for the dominance of these segments. The market size for each of these segments is in the billions.
Canada In-Vitro Diagnostics Market Product Innovations
Product innovations in the Canadian IVD market are primarily focused on enhancing accuracy, speed, and accessibility of diagnostic testing. There is a significant trend towards the development of highly multiplexed assays capable of detecting multiple biomarkers or pathogens simultaneously, reducing sample volume and turnaround time. Point-of-care testing (POCT) devices are evolving with increased sophistication, offering laboratory-grade accuracy in a portable format, enabling rapid diagnosis at the patient's bedside or in remote settings. Molecular diagnostics are witnessing innovations in isothermal amplification techniques and liquid biopsy technologies for non-invasive cancer detection and monitoring. Furthermore, the integration of AI and machine learning algorithms into IVD platforms is improving data analysis, interpretation, and predictive capabilities. These innovations offer significant competitive advantages by addressing unmet clinical needs, improving patient outcomes, and reducing healthcare costs, thus driving market growth and adoption. The market value for these innovations is projected to be in the billions.
Report Segmentation & Scope
This report provides a comprehensive analysis of the Canada in-vitro diagnostics (IVD) market, segmented by:
- Test Type: The market is segmented into Clinical Chemistry, Molecular Diagnostics, Hematology, Immuno Diagnostics, and Other Tests. Molecular Diagnostics is projected to exhibit the highest growth rate, driven by advancements in genetic testing and infectious disease diagnostics. The market size for each segment is estimated to be in the billions.
- Product: The market is analyzed across Instruments, Reagents, and Other Products. Reagents are expected to maintain the largest market share due to their consumable nature. Instruments, while having a higher initial cost, are crucial for advanced testing.
- Usability: The segmentation includes Disposable IVD Devices and Reusable IVD Devices. Disposable devices are anticipated to grow at a faster pace due to their convenience and adoption in POCT.
- Application: Key applications covered include Infectious Disease, Diabetes, Cancer/Oncology, Cardiology, Autoimmune Disease, Nephrology, and Other Applications. Infectious Disease and Cancer/Oncology are projected to be the fastest-growing application segments, driven by global health concerns and advancements in personalized medicine.
- End Users: The market is categorized into Diagnostic Laboratories, Hospitals and Clinics, and Other End Users. Diagnostic Laboratories are expected to remain the dominant end-user segment, followed closely by Hospitals and Clinics.
The scope of this report covers detailed market size estimations, growth projections, competitive landscape analysis, and key industry trends for each segment within the Canadian IVD market from 2019 to 2033, with a base year of 2025. The total market value is projected to reach billions.
Key Drivers of Canada In-Vitro Diagnostics Market Growth
The Canadian in-vitro diagnostics (IVD) market growth is propelled by several key drivers. Technological advancements in molecular diagnostics, automation, and AI are enabling more accurate, faster, and comprehensive testing. The increasing prevalence of chronic and infectious diseases, coupled with an aging population, necessitates advanced diagnostic solutions for early detection and effective management. Growing healthcare expenditure and government initiatives focused on improving public health infrastructure further stimulate market expansion. The rise of personalized medicine and companion diagnostics creates demand for specialized IVD tests that guide targeted therapies. Furthermore, supportive regulatory frameworks that facilitate the approval of innovative diagnostic technologies contribute to market dynamism. The market value is expected to be in the billions.
Challenges in the Canada In-Vitro Diagnostics Market Sector
Despite its growth potential, the Canadian in-vitro diagnostics (IVD) market faces several challenges. Stringent regulatory hurdles imposed by Health Canada can lead to lengthy approval processes for new diagnostic products, impacting time-to-market. Reimbursement policies for novel IVD tests can be complex and may not always cover the full cost, potentially limiting widespread adoption. Supply chain disruptions, as observed in recent global events, can impact the availability of critical reagents and components. High initial investment costs for advanced diagnostic instrumentation can be a barrier for smaller laboratories and clinics. Interoperability issues between different laboratory information systems and diagnostic platforms can hinder seamless data integration. Intense competition from established players and emerging innovators also necessitates continuous investment in R&D and market penetration strategies. The market value is in the billions.
Leading Players in the Canada In-Vitro Diagnostics Market Market
- Becton Dickinson and Company
- Bio-Rad Laboratories Inc
- Hologic Inc
- Abbott Laboratories
- Qiagen N V
- Siemens AG
- Danaher Corporation
- F Hoffmann-La Roche AG
- Thermo Fischer Scientific Inc
- Arkray Inc
Key Developments in Canada In-Vitro Diagnostics Market Sector
- May 2022: BioMérieux received Health Canada approval for the BioFire Blood Culture Identification 2 (BCID2) panel. This panel is designed for the rapid identification of bloodstream infections, offering an expanded range of pathogens and antimicrobial resistance genes compared to its predecessor, significantly enhancing sepsis diagnostics.
- January 2022: Yourgene, the international molecular diagnostics group, launched its new expanded facility named "Yourgene Health Canada Inc." in Canada. This expansion signifies the company's commitment to the Canadian market and is expected to bolster its presence and offerings in molecular diagnostics within the country.
Strategic Canada In-Vitro Diagnostics Market Market Outlook
The strategic outlook for the Canadian in-vitro diagnostics (IVD) market is highly positive, driven by continued innovation and evolving healthcare needs. The increasing focus on early disease detection, personalized medicine, and infectious disease surveillance will fuel demand for advanced diagnostic technologies, particularly in molecular diagnostics and immunoassay segments. Strategic opportunities lie in the development and adoption of point-of-care testing (POCT) solutions to improve patient access and reduce healthcare costs. Collaborations between IVD manufacturers, healthcare providers, and research institutions will be crucial for driving innovation and market penetration. The growing adoption of digital health solutions and AI in diagnostics presents a significant growth accelerator. Furthermore, the Canadian government's commitment to strengthening healthcare infrastructure and investing in life sciences will provide a conducive environment for market expansion, projecting a future market value in the billions.
Canada in-vitro Diagnostics Market Segmentation
-
1. Test Type
- 1.1. Clinical Chemistry
- 1.2. Molecular Diagnostics
- 1.3. Hematology
- 1.4. Immuno Diagnostics
- 1.5. Other Tests
-
2. Product
- 2.1. Instrument
- 2.2. Reagent
- 2.3. Other Products
-
3. Usability
- 3.1. Disposable IVD Devices
- 3.2. Reusable IVD Devices
-
4. Application
- 4.1. Infectious Disease
- 4.2. Diabetes
- 4.3. Cancer/Oncology
- 4.4. Cardiology
- 4.5. Autoimmune Disease
- 4.6. Nephrology
- 4.7. Other Applications
-
5. End Users
- 5.1. Diagnostic Laboratories
- 5.2. Hospitals and Clinics
- 5.3. Other End Users
Canada in-vitro Diagnostics Market Segmentation By Geography
- 1. Canada

Canada in-vitro Diagnostics Market Regional Market Share

Geographic Coverage of Canada in-vitro Diagnostics Market
Canada in-vitro Diagnostics Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.23% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Test Type
- 5.1.1. Clinical Chemistry
- 5.1.2. Molecular Diagnostics
- 5.1.3. Hematology
- 5.1.4. Immuno Diagnostics
- 5.1.5. Other Tests
- 5.2. Market Analysis, Insights and Forecast - by Product
- 5.2.1. Instrument
- 5.2.2. Reagent
- 5.2.3. Other Products
- 5.3. Market Analysis, Insights and Forecast - by Usability
- 5.3.1. Disposable IVD Devices
- 5.3.2. Reusable IVD Devices
- 5.4. Market Analysis, Insights and Forecast - by Application
- 5.4.1. Infectious Disease
- 5.4.2. Diabetes
- 5.4.3. Cancer/Oncology
- 5.4.4. Cardiology
- 5.4.5. Autoimmune Disease
- 5.4.6. Nephrology
- 5.4.7. Other Applications
- 5.5. Market Analysis, Insights and Forecast - by End Users
- 5.5.1. Diagnostic Laboratories
- 5.5.2. Hospitals and Clinics
- 5.5.3. Other End Users
- 5.6. Market Analysis, Insights and Forecast - by Region
- 5.6.1. Canada
- 5.1. Market Analysis, Insights and Forecast - by Test Type
- 6. Canada in-vitro Diagnostics Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Test Type
- 6.1.1. Clinical Chemistry
- 6.1.2. Molecular Diagnostics
- 6.1.3. Hematology
- 6.1.4. Immuno Diagnostics
- 6.1.5. Other Tests
- 6.2. Market Analysis, Insights and Forecast - by Product
- 6.2.1. Instrument
- 6.2.2. Reagent
- 6.2.3. Other Products
- 6.3. Market Analysis, Insights and Forecast - by Usability
- 6.3.1. Disposable IVD Devices
- 6.3.2. Reusable IVD Devices
- 6.4. Market Analysis, Insights and Forecast - by Application
- 6.4.1. Infectious Disease
- 6.4.2. Diabetes
- 6.4.3. Cancer/Oncology
- 6.4.4. Cardiology
- 6.4.5. Autoimmune Disease
- 6.4.6. Nephrology
- 6.4.7. Other Applications
- 6.5. Market Analysis, Insights and Forecast - by End Users
- 6.5.1. Diagnostic Laboratories
- 6.5.2. Hospitals and Clinics
- 6.5.3. Other End Users
- 6.1. Market Analysis, Insights and Forecast - by Test Type
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Becton Dickinson and Company
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Bio-Rad Laboratories Inc
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Hologic Inc
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Abbott Laboratories
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Qiagen N V
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Siemens AG
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Danaher Corporation
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 F Hoffmann-La Roche AG
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Thermo Fischer Scientific Inc
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 Arkray Inc
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.1 Becton Dickinson and Company
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: Canada in-vitro Diagnostics Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Canada in-vitro Diagnostics Market Share (%) by Company 2025
List of Tables
- Table 1: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Test Type 2020 & 2033
- Table 2: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Product 2020 & 2033
- Table 3: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Usability 2020 & 2033
- Table 4: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Canada in-vitro Diagnostics Market Revenue billion Forecast, by End Users 2020 & 2033
- Table 6: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Region 2020 & 2033
- Table 7: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Test Type 2020 & 2033
- Table 8: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Product 2020 & 2033
- Table 9: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Usability 2020 & 2033
- Table 10: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Canada in-vitro Diagnostics Market Revenue billion Forecast, by End Users 2020 & 2033
- Table 12: Canada in-vitro Diagnostics Market Revenue billion Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Canada in-vitro Diagnostics Market?
The projected CAGR is approximately 5.23%.
2. Which companies are prominent players in the Canada in-vitro Diagnostics Market?
Key companies in the market include Becton Dickinson and Company, Bio-Rad Laboratories Inc, Hologic Inc, Abbott Laboratories, Qiagen N V, Siemens AG, Danaher Corporation, F Hoffmann-La Roche AG, Thermo Fischer Scientific Inc, Arkray Inc.
3. What are the main segments of the Canada in-vitro Diagnostics Market?
The market segments include Test Type, Product, Usability, Application, End Users.
4. Can you provide details about the market size?
The market size is estimated to be USD 54.08 billion as of 2022.
5. What are some drivers contributing to market growth?
Increasing Use of Point-of-Care (POC) Diagnostics and Advancements in Technology; Increasing Awareness and Acceptance of Personalized Medicine and Companion Diagnostics.
6. What are the notable trends driving market growth?
Reagent Segment is Expected to hold the Significant Market Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Stringent Regulations and Cumbersome Reimbursement Procedures.
8. Can you provide examples of recent developments in the market?
In May 2022, BioMérieux received Health Canada approval for the BioFire Blood Culture Identification 2 (BCID2) panel for rapid identification of bloodstream infections. The BCID2 panel includes additional pathogens, an expanded list of antimicrobial resistance genes, and revised targets compared to the original BCID panel.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Canada in-vitro Diagnostics Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Canada in-vitro Diagnostics Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Canada in-vitro Diagnostics Market?
To stay informed about further developments, trends, and reports in the Canada in-vitro Diagnostics Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
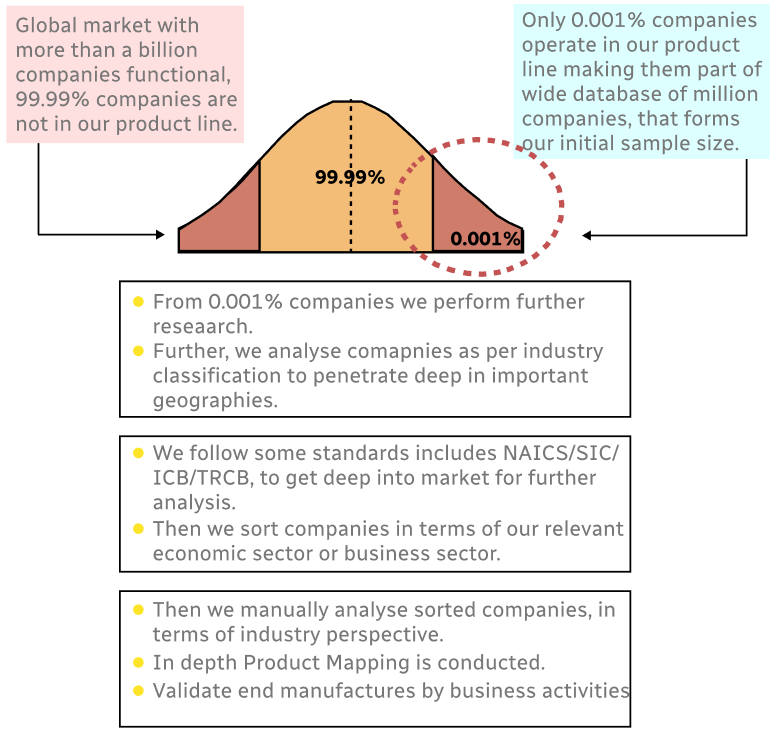
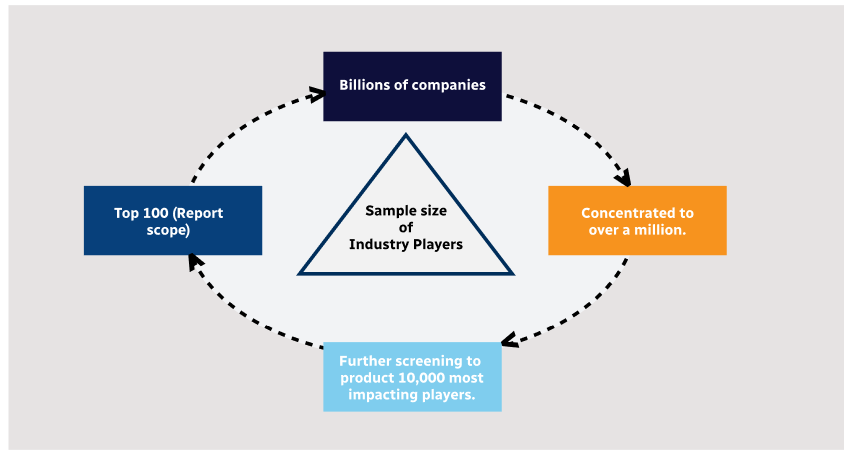
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
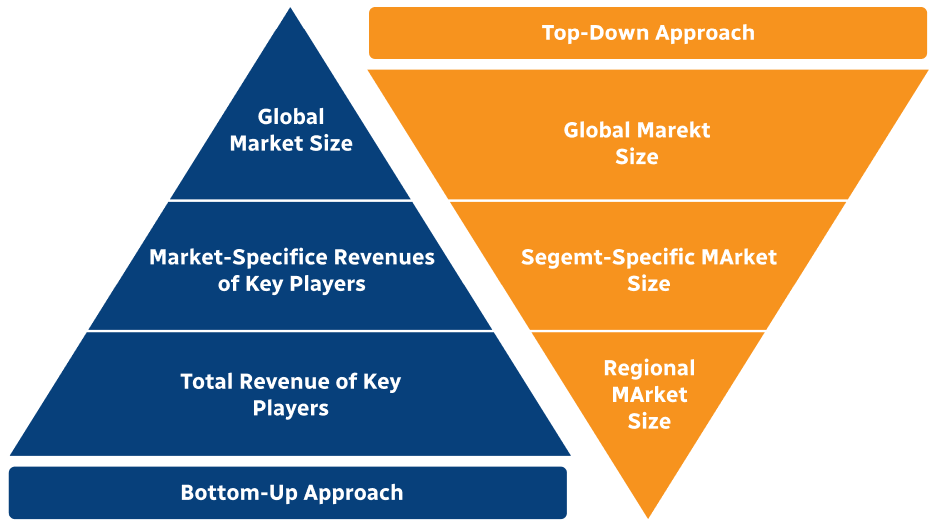
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


