Key Insights
The global Biologics Ampoule Filling Services market is poised for significant expansion, projected to reach $12.19 billion by 2025, with a robust Compound Annual Growth Rate (CAGR) of 10.42%. This remarkable growth is primarily driven by the escalating demand for biologics and biosimilars, coupled with an increasing preference for single-dose, sterile packaging solutions offered by ampoules. The pharmaceutical industry's continuous investment in research and development of novel biologic drugs, particularly in therapeutic areas like oncology, autoimmune diseases, and rare genetic disorders, fuels the need for specialized contract manufacturing organizations (CMOs) capable of providing aseptic filling services. Furthermore, the growing trend towards personalized medicine and the development of complex biologic formulations necessitate advanced filling and packaging technologies, thereby bolstering market growth. The stringent regulatory landscape and the need for high-quality, sterile manufacturing processes also favor established and specialized service providers.
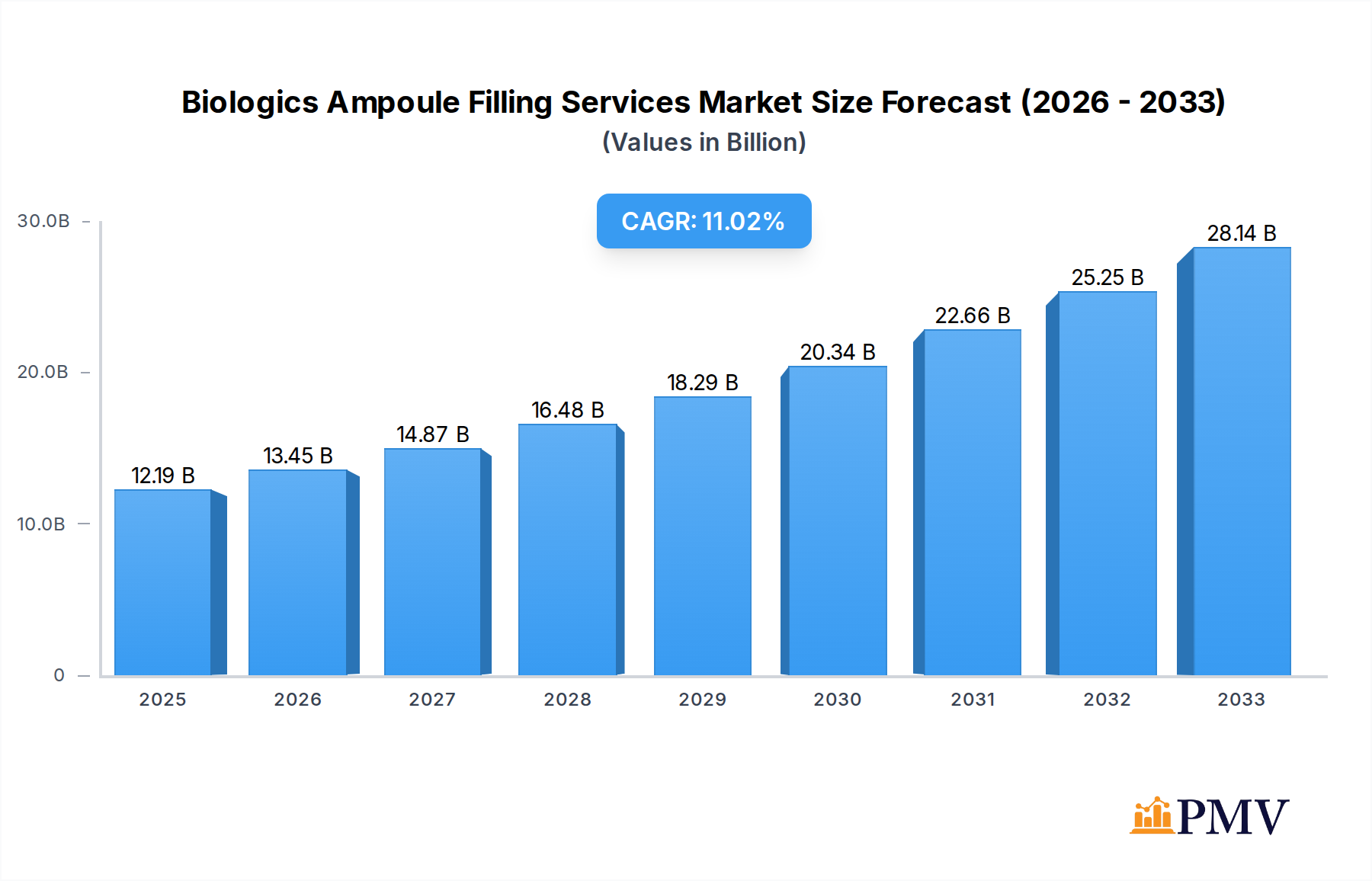
Biologics Ampoule Filling Services Market Size (In Billion)

The market's trajectory is further shaped by several key trends. The increasing outsourcing of drug manufacturing by pharmaceutical companies, seeking cost efficiencies and specialized expertise, is a significant contributor. Advancements in aseptic filling technologies, including automation and advanced containment solutions, are enhancing efficiency and product integrity, meeting the growing demand for high-volume production of sensitive biologic drugs. The expansion of biologics manufacturing capabilities in emerging economies, particularly in Asia Pacific, is also influencing market dynamics. However, challenges such as the high cost of specialized equipment, the need for highly skilled labor, and the complex regulatory compliance can pose restraints. Despite these challenges, the unwavering focus on drug quality, patient safety, and the expanding pipeline of biologics are expected to sustain the market's upward trajectory throughout the forecast period.
Biologics Ampoule Filling Services Company Market Share

This in-depth report provides an unparalleled view of the global Biologics Ampoule Filling Services market, forecasting its trajectory from 2019 to 2033. Leveraging robust data and expert analysis, this study delivers actionable insights for stakeholders seeking to navigate this dynamic and rapidly expanding sector. With a base year of 2025, the report offers granular detail across historical periods (2019-2024), the estimated year (2025), and an extensive forecast period (2025-2033). We delve into market structure, competitive landscapes, emerging trends, dominant segments, product innovations, key growth drivers, inherent challenges, leading industry players, significant developments, and strategic outlooks, all crucial for understanding the multi-billion dollar biologics contract manufacturing and fill-finish landscape. The estimated market size for biologics ampoule filling services is projected to reach xx billion USD by 2033, with a Compound Annual Growth Rate (CAGR) of xx% during the forecast period.
Biologics Ampoule Filling Services Market Structure & Competitive Dynamics
The Biologics Ampoule Filling Services market exhibits a moderately concentrated structure, with a significant presence of both large, established contract development and manufacturing organizations (CDMOs) and niche players. Innovation ecosystems are thriving, driven by advancements in aseptic processing technologies, isolator systems, and advanced aseptic filling lines. Regulatory frameworks, primarily governed by agencies like the FDA and EMA, are stringent, emphasizing sterile manufacturing and cGMP compliance, which acts as a barrier to entry for new players but ensures high quality for existing ones. Product substitutes, such as vial filling or pre-filled syringes, exist but are often application-specific, with ampoule filling remaining critical for certain sensitive biologics and high-potency drugs. End-user trends are dominated by the increasing demand for complex biologics, biosimilars, and vaccines, necessitating specialized fill-finish capabilities. Merger and acquisition (M&A) activities are notable, with deal values often reaching hundreds of millions to over a billion USD, as larger players seek to expand their service offerings and geographical reach. Key M&A targets include companies with specialized capabilities in sterile fill-finish for challenging molecules.
- Market Concentration: Moderate, with a blend of large CDMOs and specialized service providers.
- Innovation Ecosystems: Driven by sterile processing, isolator technology, and advanced filling lines.
- Regulatory Frameworks: Stringent cGMP, FDA, and EMA compliance are paramount.
- Product Substitutes: Vial filling and pre-filled syringes, but ampoules retain niche importance.
- End-User Trends: Growing demand for complex biologics, biosimilars, and vaccines.
- M&A Activities: Significant, with deal values reaching hundreds of millions to over a billion USD.
Biologics Ampoule Filling Services Industry Trends & Insights
The global Biologics Ampoule Filling Services market is experiencing robust growth, fueled by several interconnected trends. A primary driver is the escalating demand for biologics, including monoclonal antibodies, therapeutic proteins, vaccines, and advanced cell and gene therapies. The increasing prevalence of chronic diseases and the continuous development of novel biological drugs necessitate sophisticated and reliable fill-finish solutions. The rise of biosimilars further contributes to market expansion, as manufacturers seek cost-effective and high-quality manufacturing partners. Technological advancements in aseptic filling technology are revolutionizing the sector. Innovations such as advanced isolator technology, robotic filling systems, and integrated inspection and packaging solutions are enhancing accuracy, reducing contamination risks, and improving efficiency. The adoption of single-use technologies is also gaining traction, offering flexibility and minimizing cross-contamination risks.
Consumer preferences, or rather, the demands of pharmaceutical and biotechnology companies, are increasingly leaning towards integrated CDMO services that encompass not only fill-finish but also drug product formulation, lyophilization, and terminal sterilization. This holistic approach simplifies supply chains and accelerates time-to-market. The competitive landscape is characterized by intense rivalry among established players and emerging CDMOs, all striving to differentiate themselves through specialized expertise, regulatory compliance, and investment in advanced infrastructure. Strategic partnerships and collaborations are becoming more common as companies seek to leverage each other's strengths and expand their service portfolios. The market penetration of specialized biologics ampoule filling services is steadily increasing, driven by the growing complexity of drug molecules and the stringent quality requirements associated with parenteral drug products. The estimated market penetration is projected to exceed xx% by 2033.
Furthermore, the global shift towards personalized medicine and the development of smaller batch sizes for rare diseases or clinical trial materials are creating opportunities for flexible and scalable fill-finish solutions. CDMOs that can offer customized solutions and rapid turnaround times are well-positioned for success. The overall market growth is also influenced by investments in new manufacturing facilities and the expansion of existing ones to meet the growing pipeline of biologic drugs. The total market revenue is projected to grow from xx billion USD in 2019 to an estimated xx billion USD by 2033, showcasing a significant upward trend.
Dominant Markets & Segments in Biologics Ampoule Filling Services
The Biologics Ampoule Filling Services market demonstrates significant regional dominance, with North America and Europe currently leading in terms of market share and investment. This is attributed to the presence of major pharmaceutical and biotechnology hubs, a robust R&D pipeline, and favorable regulatory environments that encourage innovation and outsourcing. Within these regions, countries like the United States and Germany are particularly influential due to their advanced healthcare infrastructure and the high concentration of biologics manufacturers. The Asia-Pacific region is emerging as a rapid growth area, driven by increasing investments in biopharmaceutical manufacturing capabilities, growing domestic demand for biologics, and favorable cost structures for contract manufacturing.
In terms of Application, the Biologics and Biosimilar segment holds the largest market share. This is a direct consequence of the substantial pipeline of biologic drugs under development, including monoclonal antibodies, recombinant proteins, and enzymes, as well as the growing market for biosimilars that offer more affordable alternatives to originator biologics. The Vaccines segment is also a significant contributor, particularly in light of global health events and ongoing efforts to develop and manufacture new vaccines. The 'Others' segment, encompassing specialized biologics, cell and gene therapies, and autologous treatments, is a rapidly expanding niche with high growth potential, albeit from a smaller base.
Analyzing by Type, the 1-5ml segment is currently the dominant category for ampoule filling services. This size range is widely used for many standard biologic drug formulations. However, the >5ml segment is experiencing substantial growth, reflecting the development of higher-volume biologic therapeutics and the increasing trend of drug product concentration. The 0.2-1ml segment, while smaller, remains important for specific applications, such as high-potency active pharmaceutical ingredients (APIs) and certain peptide-based therapeutics, and also sees demand for pediatric formulations.
- Leading Region: North America, followed closely by Europe.
- Emerging Region: Asia-Pacific, driven by investment and domestic demand.
- Dominant Application: Biologics and Biosimilar, due to extensive drug pipelines.
- Growing Application: Vaccines, driven by global health initiatives.
- Niche & High-Growth Application: Others (Cell & Gene Therapies, rare disease treatments).
- Dominant Type: 1-5ml, catering to a broad range of biologics.
- Rapidly Growing Type: >5ml, accommodating larger volume therapeutics.
- Important Type: 0.2-1ml, for high-potency and specialized formulations.
Biologics Ampoule Filling Services Product Innovations
Product innovations in Biologics Ampoule Filling Services are primarily focused on enhancing sterility assurance, improving filling accuracy, and increasing process efficiency. Advancements in isolator technology, equipped with integrated robotic arms, allow for aseptic filling with minimal human intervention, significantly reducing the risk of microbial contamination. New types of filling machines offer higher throughput and precision, enabling the accurate dispensing of small volumes of sensitive biologics. Innovations in container closure integrity testing and particulate detection systems further bolster product quality and safety. The development of advanced lyophilization techniques optimized for biologics, integrated directly with ampoule filling lines, is another key innovation, preserving the stability of sensitive molecules. These technological advancements translate into competitive advantages by enabling CDMOs to handle more complex and sensitive drug products, meet stringent regulatory requirements, and offer faster turnaround times, thereby attracting a broader client base.
Report Segmentation & Scope
This report segments the Biologics Ampoule Filling Services market based on key criteria to provide granular insights. The segmentation is categorized by Application, including Vaccines, Biologics and Biosimilar, and Others. The Biologics and Biosimilar segment is projected to hold the largest market share, estimated at xx billion USD in 2025, and is expected to grow at a CAGR of xx% during the forecast period. The Vaccines segment, while influenced by seasonal demands and global health initiatives, is anticipated to reach xx billion USD by 2033. The Others segment, encompassing cell and gene therapies and other novel biological entities, is expected to exhibit the highest growth rate, projected to expand from xx billion USD in 2025 to xx billion USD by 2033.
The market is further segmented by Type of ampoule fill volume: 0.2-1ml, 1-5ml, and >5ml. The 1-5ml segment currently dominates, estimated at xx billion USD in 2025, with a steady CAGR of xx%. The >5ml segment is the fastest-growing, driven by larger biologic formulations, with projections to reach xx billion USD by 2033. The 0.2-1ml segment, crucial for specialized applications, is expected to grow at a CAGR of xx%, reaching xx billion USD by 2033. The scope of this report encompasses a comprehensive analysis of these segments, their growth drivers, market sizes, and competitive dynamics.
Key Drivers of Biologics Ampoule Filling Services Growth
The growth of the Biologics Ampoule Filling Services market is propelled by several critical factors. The escalating demand for biologics, driven by the increasing prevalence of chronic diseases and the continuous innovation in drug discovery, is a primary catalyst. Advancements in biotechnology have led to the development of complex therapeutic proteins, monoclonal antibodies, and vaccines, all of which require specialized aseptic fill-finish capabilities. Furthermore, the expanding pipeline of biosimilars, offering more affordable alternatives to expensive biologics, is stimulating demand for contract manufacturing services. Technological innovations in sterile fill-finish, such as advanced isolator technology and automated filling lines, are enhancing efficiency and safety, making outsourcing more attractive. Favorable regulatory landscapes in key markets that support the development and commercialization of biologics also contribute significantly to market growth. The trend towards outsourcing by pharmaceutical companies, seeking to focus on core competencies and leverage specialized expertise, further fuels the demand for CDMOs offering biologics ampoule filling services.
Challenges in the Biologics Ampoule Filling Services Sector
Despite robust growth, the Biologics Ampoule Filling Services sector faces several significant challenges. Stringent and evolving regulatory requirements, particularly concerning aseptic processing and sterile manufacturing, necessitate continuous investment in compliance and quality control, acting as a barrier for smaller players. Supply chain disruptions, including the availability of specialized raw materials and components for sterile packaging, can impact production schedules and costs. The highly competitive landscape, with numerous established and emerging CDMOs, leads to pricing pressures and the need for continuous differentiation through service offerings and technological capabilities. The high capital investment required for state-of-the-art aseptic filling facilities and the specialized workforce needed to operate them also present a financial hurdle. Furthermore, the risk of product recalls due to contamination or quality issues, while rare, can have severe financial and reputational consequences for CDMOs. The estimated financial impact of a major recall can range from tens to hundreds of millions of USD.
Leading Players in the Biologics Ampoule Filling Services Market
The Biologics Ampoule Filling Services market is characterized by the presence of several key players, including:
- Baxter BioPharma Solutions
- Boehringer Ingelheim
- Vetter Pharma
- Fresenius Kabi
- Pfizer CentreOne
- Aenova
- WuXi Biologics
- Jubilant HollisterStier
- Bushu Pharmaceuticals
- LSNE Contract Manufacturing
- Ajinomoto Bio-Pharma Services
- CMIC CMO
- GRAM (Grand River Aseptic Manufacturing)
- TAIYO Pharma Tech
- HALIX
- Cognate BioServices
- Afton Scientific
- Novasep
- Emergent BioSolutions
- Seikagaku
- Jiangshu YAOHAI Bio-pharmaceutical
- Akron Biotech
- Symbiosis Pharmaceutical Services
- Techdow
- Vigene Biosciences
Key Developments in Biologics Ampoule Filling Services Sector
- 2023: WuXi Biologics announced a significant expansion of its fill-finish capacity with new isolator lines, enhancing its ability to handle high-demand biologics.
- 2022: Vetter Pharma invested heavily in advanced robotic filling technology to further automate its aseptic fill-finish operations, improving efficiency and reducing human intervention.
- 2021: Baxter BioPharma Solutions acquired a new sterile manufacturing facility, significantly increasing its ampoule filling capabilities for vaccines and other biologics.
- 2020: Boehringer Ingelheim expanded its sterile filling capacity for biologics, anticipating a surge in demand for therapeutic proteins and antibodies.
- 2019: Pfizer CentreOne invested in state-of-the-art isolator technology, strengthening its position in the high-potency biologics fill-finish market.
- Ongoing: Continuous advancements in single-use technologies for sterile filling are being adopted by many CDMOs to offer greater flexibility and reduce cross-contamination risks.
- Ongoing: Increased focus on end-to-end supply chain solutions, integrating fill-finish with lyophilization, labeling, and packaging services.
Strategic Biologics Ampoule Filling Services Market Outlook
The strategic outlook for the Biologics Ampoule Filling Services market remains highly optimistic, driven by sustained innovation in the biopharmaceutical industry and an increasing reliance on specialized CDMOs. Growth accelerators include the continued development of novel biologics, the expanding market for biosimilars, and the rising demand for vaccines. The increasing complexity of therapeutic molecules and the stringent quality requirements for parenteral drugs will further solidify the need for advanced aseptic fill-finish capabilities. Strategic opportunities lie in investing in cutting-edge technologies such as advanced isolator systems, single-use technologies, and AI-driven process optimization. Companies that can offer comprehensive, integrated services, from drug product formulation to final packaging, are poised for significant growth. Expanding geographical reach, particularly into emerging markets with growing biopharmaceutical sectors, also presents a key strategic avenue. Collaboration and partnerships between CDMOs and biopharmaceutical companies will continue to be crucial for navigating the evolving market landscape and ensuring efficient delivery of life-saving biologics. The projected market size of xx billion USD by 2033 underscores the immense potential and strategic importance of this sector.
Biologics Ampoule Filling Services Segmentation
-
1. Application
- 1.1. Vaccines
- 1.2. Biologics and Biosimilar
- 1.3. Others
-
2. Type
- 2.1. 0.2-1ml
- 2.2. 1-5ml
- 2.3. >5ml
Biologics Ampoule Filling Services Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
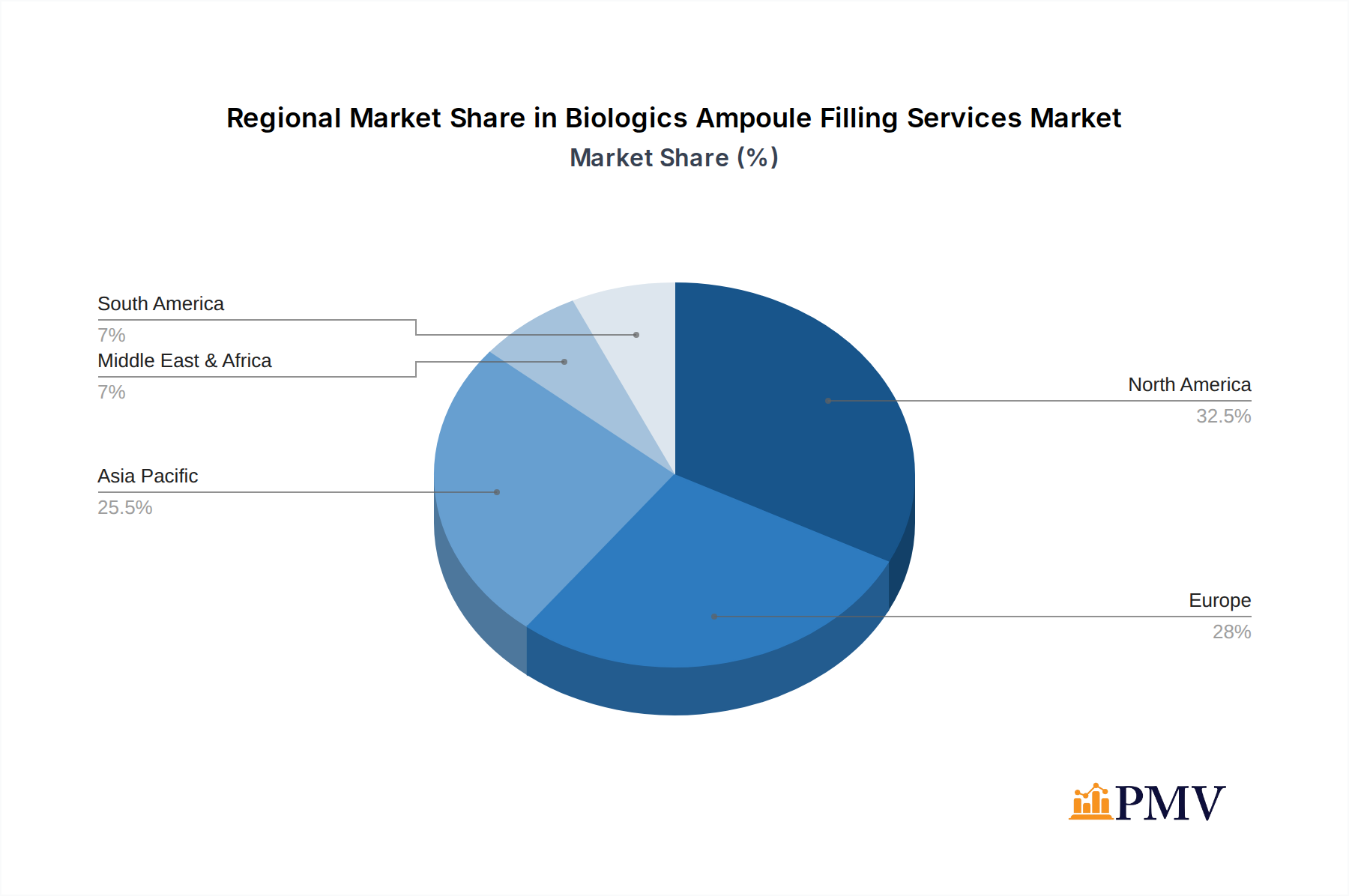
Biologics Ampoule Filling Services Regional Market Share

Geographic Coverage of Biologics Ampoule Filling Services
Biologics Ampoule Filling Services REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.42% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. PMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Vaccines
- 5.1.2. Biologics and Biosimilar
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. 0.2-1ml
- 5.2.2. 1-5ml
- 5.2.3. >5ml
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Biologics Ampoule Filling Services Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Vaccines
- 6.1.2. Biologics and Biosimilar
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. 0.2-1ml
- 6.2.2. 1-5ml
- 6.2.3. >5ml
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Biologics Ampoule Filling Services Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Vaccines
- 7.1.2. Biologics and Biosimilar
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. 0.2-1ml
- 7.2.2. 1-5ml
- 7.2.3. >5ml
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Biologics Ampoule Filling Services Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Vaccines
- 8.1.2. Biologics and Biosimilar
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. 0.2-1ml
- 8.2.2. 1-5ml
- 8.2.3. >5ml
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Biologics Ampoule Filling Services Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Vaccines
- 9.1.2. Biologics and Biosimilar
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. 0.2-1ml
- 9.2.2. 1-5ml
- 9.2.3. >5ml
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Biologics Ampoule Filling Services Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Vaccines
- 10.1.2. Biologics and Biosimilar
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. 0.2-1ml
- 10.2.2. 1-5ml
- 10.2.3. >5ml
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Biologics Ampoule Filling Services Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Vaccines
- 11.1.2. Biologics and Biosimilar
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Type
- 11.2.1. 0.2-1ml
- 11.2.2. 1-5ml
- 11.2.3. >5ml
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Baxter BioPharma Solutions
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Boehringer Ingelheim
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Vetter Pharma
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Fresenius Kabi
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Pfizer CentreOne
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Aenova
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 WuXi Biologics
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Jubilant HollisterStier
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Bushu Pharmaceuticals
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 LSNE Contract Manufacturing
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Ajinomoto Bio-Pharma Services
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 CMIC CMO
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 GRAM (Grand River Aseptic Manufacturing)
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 TAIYO Pharma Tech
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 HALIX
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.16 Cognate BioServices
- 12.1.16.1. Company Overview
- 12.1.16.2. Products
- 12.1.16.3. Company Financials
- 12.1.16.4. SWOT Analysis
- 12.1.17 Afton Scientific
- 12.1.17.1. Company Overview
- 12.1.17.2. Products
- 12.1.17.3. Company Financials
- 12.1.17.4. SWOT Analysis
- 12.1.18 Novasep
- 12.1.18.1. Company Overview
- 12.1.18.2. Products
- 12.1.18.3. Company Financials
- 12.1.18.4. SWOT Analysis
- 12.1.19 Emergent BioSolutions
- 12.1.19.1. Company Overview
- 12.1.19.2. Products
- 12.1.19.3. Company Financials
- 12.1.19.4. SWOT Analysis
- 12.1.20 Seikagaku
- 12.1.20.1. Company Overview
- 12.1.20.2. Products
- 12.1.20.3. Company Financials
- 12.1.20.4. SWOT Analysis
- 12.1.21 Jiangshu YAOHAI Bio-pharmaceutical
- 12.1.21.1. Company Overview
- 12.1.21.2. Products
- 12.1.21.3. Company Financials
- 12.1.21.4. SWOT Analysis
- 12.1.22 Akron Biotech
- 12.1.22.1. Company Overview
- 12.1.22.2. Products
- 12.1.22.3. Company Financials
- 12.1.22.4. SWOT Analysis
- 12.1.23 Symbiosis Pharmaceutical Services
- 12.1.23.1. Company Overview
- 12.1.23.2. Products
- 12.1.23.3. Company Financials
- 12.1.23.4. SWOT Analysis
- 12.1.24 Techdow
- 12.1.24.1. Company Overview
- 12.1.24.2. Products
- 12.1.24.3. Company Financials
- 12.1.24.4. SWOT Analysis
- 12.1.25 Vigene Biosciences
- 12.1.25.1. Company Overview
- 12.1.25.2. Products
- 12.1.25.3. Company Financials
- 12.1.25.4. SWOT Analysis
- 12.1.1 Baxter BioPharma Solutions
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Biologics Ampoule Filling Services Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Biologics Ampoule Filling Services Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Biologics Ampoule Filling Services Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Biologics Ampoule Filling Services Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Biologics Ampoule Filling Services Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Biologics Ampoule Filling Services Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Biologics Ampoule Filling Services Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Biologics Ampoule Filling Services Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Biologics Ampoule Filling Services Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Biologics Ampoule Filling Services Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Biologics Ampoule Filling Services Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Biologics Ampoule Filling Services Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Biologics Ampoule Filling Services Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Biologics Ampoule Filling Services Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Biologics Ampoule Filling Services Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Biologics Ampoule Filling Services Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Biologics Ampoule Filling Services Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Biologics Ampoule Filling Services Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Biologics Ampoule Filling Services Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Biologics Ampoule Filling Services Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Biologics Ampoule Filling Services Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Biologics Ampoule Filling Services Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Biologics Ampoule Filling Services Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Biologics Ampoule Filling Services Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Biologics Ampoule Filling Services Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Biologics Ampoule Filling Services Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Biologics Ampoule Filling Services Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Biologics Ampoule Filling Services Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Biologics Ampoule Filling Services Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Biologics Ampoule Filling Services Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Biologics Ampoule Filling Services Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Biologics Ampoule Filling Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Biologics Ampoule Filling Services Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Biologics Ampoule Filling Services?
The projected CAGR is approximately 10.42%.
2. Which companies are prominent players in the Biologics Ampoule Filling Services?
Key companies in the market include Baxter BioPharma Solutions, Boehringer Ingelheim, Vetter Pharma, Fresenius Kabi, Pfizer CentreOne, Aenova, WuXi Biologics, Jubilant HollisterStier, Bushu Pharmaceuticals, LSNE Contract Manufacturing, Ajinomoto Bio-Pharma Services, CMIC CMO, GRAM (Grand River Aseptic Manufacturing), TAIYO Pharma Tech, HALIX, Cognate BioServices, Afton Scientific, Novasep, Emergent BioSolutions, Seikagaku, Jiangshu YAOHAI Bio-pharmaceutical, Akron Biotech, Symbiosis Pharmaceutical Services, Techdow, Vigene Biosciences.
3. What are the main segments of the Biologics Ampoule Filling Services?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Biologics Ampoule Filling Services," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Biologics Ampoule Filling Services report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Biologics Ampoule Filling Services?
To stay informed about further developments, trends, and reports in the Biologics Ampoule Filling Services, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
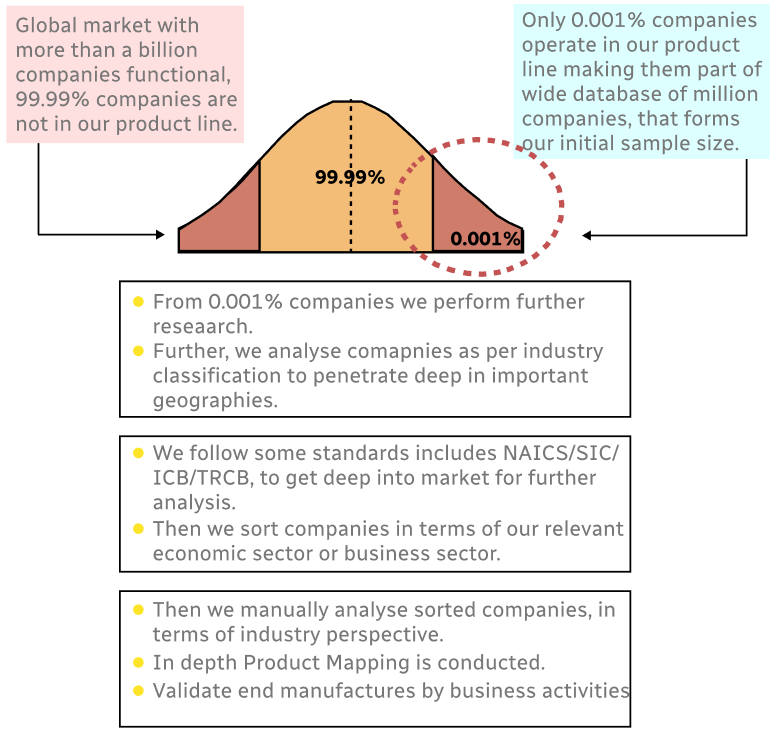
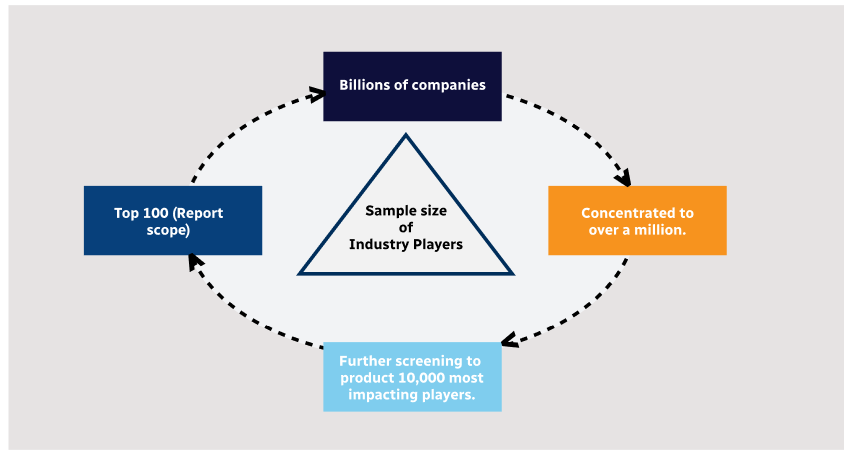
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
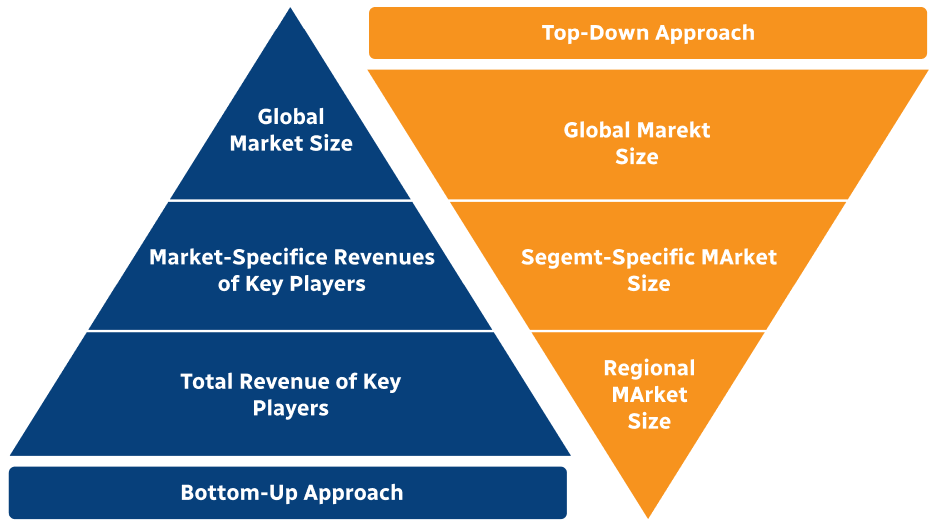
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
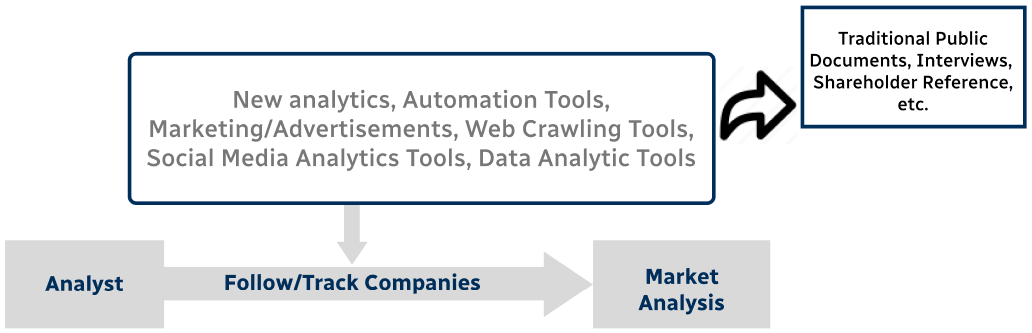
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


